Abstract
Background
The measurement of anti-Müllerian hormone (AMH) has been by two commercial enzyme-linked immunosorbent (ELISA) assays: Diagnostics Systems Laboratory (DSL 10-14400) and Immunotech (A11893 IVD EU only). Beckman Coulter has developed a new assay for AMH (AMH Gen II A79765), which uses the DSL antibodies but is standardized to the Immunotech calibration. As a result, comparative data are urgently required between the old DSL assay and its replacement AMH Gen II.
Methods
An evaluation of the AMH Gen II assay was performed at three sites, each with extensive experience of measuring circulating AMH in the adult female. Results were compared with the original DSL ELISA assay. The analysis was performed on a total of 271 patients' samples, approximately 90 at each site.
Results
Performance characteristics were evaluated for the AMHGen II assay. Linearity was acceptable with observed values close to the expected (mean recovery 106.3%). The functional sensitivity (20% coefficient of variation), calculated from precision profile data, was 1.5 pmol/L. Within- and between-batch imprecision, assessed over the concentration range of 5–70 pmol/L, were 5.3–11.4% and 3.8–17.3%, respectively. There was good agreement between assays with a Bablok-Passing regression equation AMH Gen II = 1.40 DSL–0.62 pmol/L, r = 0.96, n = 271.
Conclusions
Our results demonstrate that similar precision and excellent between-assay agreement should be obtained when laboratories change from the DSL to the AMH Gen II ELISA and they should expect an increase in AMH values of approximately 40%.
Introduction
Anti-Müllerian hormone (AMH), also known as Müllerian inhibiting substance (MIS), is strongly expressed in Sertoli cells from the time of testicular differentiation to puberty and to a much lesser degree in granulosa cells from birth to menopause. 1,2 AMH is a glycoprotein dimer composed of two 72-kDa monomers linked by disulphide bridges and is a member of the transforming growth factor-β super family. 3 Circulating AMH is detectable at birth in males and then rises dramatically during infancy before gradually declining by puberty. 4 In females, circulating concentrations are generally an order of magnitude lower than for males, but do increase around the time of puberty to maximal concentrations in early adulthood. Thereafter, a steady and unrelenting decline is seen until AMH becomes undetectable in the circulation several years prior to the menopause. The decline in AMH through reproductive life appears to mirror the non-linear decay in the number of non-growing primordial follicles – the ovarian reserve. 5
Increased understanding of the role of AMH in pathophysiology and recent availability of commercial immunoassays for its measurement has led to its increased use as a diagnostic tool in reproductive medicine. 6 The main roles include investigations into disorders of sexual differentiation, polycystic ovarian disease, female fertility and as a tumour marker in granulosa cell tumours. 6–8 The dramatic increase in AMH requesting over the last few years is mostly related to it being an accurate predictor of the extremes of ovarian response to exogenous gonadotrophins in cycles of assisted conception. 9–17 In particular, AMH can identify women at risk of a poor ovarian response 10,13–17 and also ovarian hyperstimulation. 9 Consequently, it is rapidly being adopted by clinicians in reproductive medicine to individualize stimulation strategies and improve outcome counselling for women about to undergo assisted conception. In addition, AMH has been proposed as a means to predict the age of menopause, 18 with further longitudinal studies ongoing.
Until recently, two enzyme-linked immunosorbent (ELISA) assays have been available; from Diagnostics Systems Laboratory (DSL 10-14400) and Immunotech (A11893 IVD EU only). These assays use different pairs of monoclonal antibodies and have been standardized differently so give different values for quantitation of AMH. The different values generated by the two assays for AMH have created some confusion for investigators. Following the acquisition of both DSL and Immunotech by Beckman Coulter, the assays have recently been harmonized. In the future, a new Gen II assay (AMH Gen II A79765) will be the only test available from Beckman Coulter. The new assay incorporates the antibodies previously deployed in the DSL assay and, as such, will allow the use of the test in species other than human, but is calibrated to the Immunotech assay and therefore will give similar numerical values.
In the current study we use a multicentre, simultaneous approach to assess the performance of the new AMH Gen II assay versus the original DSL ELISA assay, and make recommendations regarding appropriate clinical cut-off values especially for use in controlled ovarian stimulation (COS) programmes.
Materials and methods
Measurement of AMH
Two commercially available ELISAs for measuring serum AMH were compared. The DSL Active MIS/AMH Elisa (reference DSL-10-14400) and the re-standardized newer version AMH Gen II Elisa (reference A79765) were supplied by Beckman Coulter (High Wycombe, UK). Kit quality control material (QC I and II) was used as supplied. Further human serum QC material was obtained from the Assay and Control Company Ltd (Upper Heyford, Oxon, UK) (QC A and B). To enable assessment of assay performance around a critical decision-making concentration (5 pmol/L), an ‘in house’ serum pool was also examined.
Study design and analyses
An evaluation of the AMH Gen II assay was performed at three sites, each with extensive experience in measuring circulating AMH in the adult human female (Department of Clinical Biochemistry, Glasgow Royal Infirmary, the Centre for Population and Health Sciences, University of Glasgow and Glasgow Centre for Reproductive Medicine). A total of 271 patients' samples were analysed in duplicate (approximately 90 at each of the three sites) to provide comparative data, with additional comparison of a series of common pooled samples at critical clinical levels. The use of patients' samples for this purpose was approved by the local ethics committee. Statistical analysis was performed using SAS v9.2 (SAS Institute, Cary, NC, USA), Analyse-it software for Microsoft® Excel (Analyse-it Software Ltd, Leeds, UK) and Variance Function Program 9.0 (B Sadler, Nuclear Medicine Department, Christchurch Hospital, Christchurch, New Zealand).
Results
AMH Gen II assay characteristics
Linearity
Six serum pools containing AMH concentrations varying between 25 and 62 pmol/L were diluted two-, four-, eight- and 16-fold, volume for volume in serum samples from postmenopausal women (no detectable AMH) and analysed at one laboratory (Glasgow Royal Infirmary). For all samples, dilution was linear with an overall % recovery of 106 ± 15%.
Functional sensitivity
The functional sensitivity was determined as the concentration at which the imprecision of duplicate determinations is 20%. It was calculated from the precision profile using patients' samples (n = 250) measured at each of the sites in the study on four consecutive assays and found to be 1.5 pmol/L.
Imprecision
Imprecision was established using the two supplied kit controls, an ‘in house’ control and two commercial controls. For intra-assay performance the kit controls were assayed eight times and the other controls five times for each laboratory. Inter-assay performance was assessed in a total of 15 assays (5 performed in each laboratory) across a period of 20 d. Mean results (coefficient of variation [cv]) for the three laboratories are reported in Table 1.
Imprecision data of Gen II AMH assay
AMH, anti-Müllerian hormone; CV, coefficient of variation
Results are reported as mean for the three participating laboratories
*Commercial control material
Comparative study
A total of 271 patients' samples were analysed in duplicate (∼90 at each of the three sites) to provide comparative data. To allow assessment of assay agreement a Bland–Altman difference plot was constructed (Figure 1). Passing–Bablok regression analysis was also performed and using the complete data-set a regression equation derived, where y = AMH Gen II and x = AMH DSL, was y = 1.39x − 0.62 (r = 0.96). Given the suggested crossover in bias at 20 pmol/L, analysis of AMH concentration range less than and greater 20 pmol/L were undertaken separately; however, the derived equations were not statistically different. Full regression analysis, including the 95% confidence limits (95% CL) and standard errors (SE) are shown in Table 2.
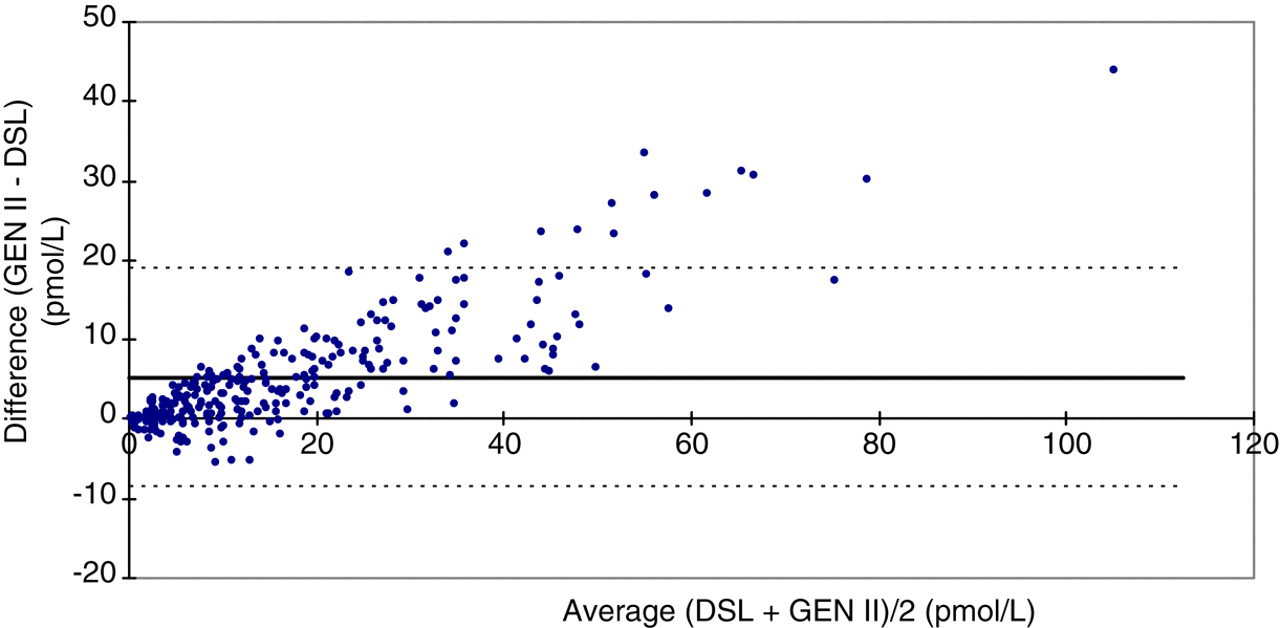
Bland and Altman difference plot comparing the anti-Müllerian hormone (AMH) Gen II and the DSL (Diagnostics Systems Laboratory) AMH enzyme-linked immunoassays. The bold solid line denotes bias and the dotted line denotes 95% limits of agreement of the difference. Values are in pmol/L
Passing-Bablok regression analysis of AMH Gen II (y) versus DSL (x) AMH assays
DSL, Diagnostics Systems Laboratory; AMH, anti-Müllerian hormone; CL, confidence limits; SE, standard error
*The equations for concentrations below and above 20 pmol/L did not differ significantly
Discussion
This study clearly demonstrates that AMH results produced using the AMH Gen II assay agree well with the original DSL ELISA assay, but gives results that are 40% higher due to harmonization with the Beckman Coulter Immunotech assay. A close association between Gen II AMH and egg yield in COS, as previously demonstrated for the AMH DSL assay, 5 is therefore to be expected. As illustrated by the Bland–Altman difference plot, higher AMH values are obtained by the Gen II assay and the extent of this increase, increases with AMH concentration. To accommodate this, regression analysis was performed not only on the whole data-set but on two subsets representing the lower and upper half of values obtained in adult females (<20 and >20 pmol/L). Although the gradient of the regression equation increased from 1.32 to 1.42 in the subsets representing lower and higher AMH concentrations, they were not statistically different.
The specific interest of the Glasgow group is related to the use of AMH measurements to aid the strategic approach to COS. 9,19 We have previously demonstrated that the two most important diagnostic cut-offs were at 5 and 15 pmol/L (DSL assay), which demarcate the poor responder at the bottom and the potentially excessive responder at the top. Concentrations between 5 and 15 pmol/L identified women who were described as ‘normal’ responders, but who may be better declared as ‘safe’ responders, as the incidence of excessive responses and ovarian hyperstimulation syndrome were negligible in these women. Using the linear regression equation for the full data-set, the corresponding cut-offs for the Gen II assay, are 6.4 and 20.4 pmol/L. Confirmation of the reliability of these values and development of new age-related normative ranges will be required in prospective studies. 5 Acceptable inter-assay imprecision with CVs <11.9% were obtained for the higher QC pools, which had a concentration range of 15.5–70 pmol/L. Intra- and inter-assay impression (%CV) of the lowest QC pool (5.1 pmol/L) was 11.4% and 17.3%, respectively, well above the functional sensitivity of 1.5 pmol/L. This information confirms that the assay will perform adequately at the important diagnostic cut-off points of 6.4 and 20.4 pmol/L. However, subtle clinical interpretation of results below this, given that it is approaching the limits of assay functional sensitivity, is not possible apart from indicating that the ovarian reserve is low. Overall our results demonstrate that similar precision and excellent agreement should be obtained by laboratories transitioning from the DSL to the AMH Gen II ELISA, although they will observe a 40% increase in results due to harmonization with the Immunotech assay.
DECLARATIONS
