Abstract
Background
Current guidelines define acute myocardial infarction (AMI) by the rise and/or fall of cardiac troponin with ≥1 value above the 99th percentile. Past troponin assays have been unreliable at the lower end of the range. Highly sensitive assays have therefore been developed to increase the clinical sensitivity for detection of myocardial injury.
Methods
Three hundred and thirty-two patients with chest pain suggestive of AMI were prospectively recruited between November 2006 and April 2007. Serial blood samples were analysed to compare Roche Elecsys high sensitivity troponin T (hsTnT), Abbott Architect troponin I 3rd generation (TnI 3) and Roche Elecsys troponin T (TnT) for the diagnosis of AMI.
Results
One hundred and ten (33.1%) patients were diagnosed with AMI. Test performance for the diagnosis of AMI, as quantified by receiver operating characteristic area under the curve (95% confidence intervals) for baseline/follow-up troponins were as follows: hsTnT 0.90 (0.87–0.94)/0.94 (0.91–0.97), TnI 3 0.88 (0.84–0.92)/0.93 (0.90–0.96) and TnT 0.80 (0.74–0.85)/0.89 (0.85–0.94). hsTnT was superior to TnT (P < 0.001/0.013 at baseline/follow-up) but equivalent to TnI 3. For patients with a final diagnosis of AMI, baseline troponins were raised in more patients for hsTnT (83.6%) than TnI 3 (74.5%) and TnT (62.7%). A delta troponin of ≥20% increased the specificity of hsTnT from 80.6% to 93.7% but reduced sensitivity from 90.9% to 71.8%.
Conclusion
hsTnT was superior to TnT but equivalent to TnI 3 for the diagnosis of AMI. Serial troponin measurement increased test performance. hsTnT was the most likely to be raised at baseline in those with AMI. A delta troponin increases specificity but reduces sensitivity.
Introduction
The global taskforce for the redefinition of acute myocardial infarction (AMI) characterizes AMI according to three criteria:
Evidence of myocardial ischaemia; A rise and/or fall of troponin; At least one troponin ≥99th percentile of the upper reference limit.
1
Prior and even some contemporary troponin assays have been relatively insensitive, in part due to increased imprecision at low values and limits of detection that are comparatively high.
2–8
There has been development of high-sensitivity troponin assays to increase the analytical, and thus clinical, sensitivity for detection of myocardial injury.
Recent studies using contemporary assays with enhanced analytical sensitivity 5,6,9–11 and with specialized high-sensitivity assays 11–13 have demonstrated that such an approach significantly improves sensitivity for the diagnosis of AMI and also permits earlier diagnosis. 2–13 Earlier detection of troponin elevation may enable a change in hospital chest pain pathway algorithms. This may allow more rapid triage to intensive and invasive treatment strategies and may allow those with normal early troponin values to undergo earlier stress testing or even be discharged without such testing. 4,14
There are concerns that the specificity will be adversely affected by increasing the analytical sensitivity since many other insults, both acute and chronic, can damage the myocardium. 8 Although it has been the practice of some to diagnose AMI based on a single measurement of troponin ≥99th percentile, the formal definition requires a rise and/or fall 1 to help distinguish chronic from acute elevations, although the degree to which troponin must change has not been explicitly defined. The National Academy of Clinical Biochemistry Laboratory Medicine Practice Guidelines has recommended changes in troponin of >20% from elevated baseline values but they too are not explicit on the change that must occur when levels are lower. 15 This is because the appropriate change to define either analytical or biological variability, or to define clinical efficacy, is assay dependent. 12 If this dynamic change in troponin is not present, non-acute cardiac conditions such as cardiomyopathy and valvular disease should be considered. 4,8,10,16,17 However, even a rising pattern does not confirm the diagnosis of AMI unless the presentation is suggestive of acute coronary syndrome (ACS) because other acute illnesses such as pulmonary embolism and sepsis have also been associated with acute cardiac injury.
The aim of our investigation was to validate that a high-sensitivity assay for troponin T provided more rapid diagnosis of AMI, looking in particular at those who present early after symptom onset, and also to determine the effects of increased analytical sensitivity on specificity when compared with contemporary troponin assays, further investigating the role of dynamic changes in troponin to aid diagnosis.
Methods
Consecutive patients attending the Emergency Department at Christchurch Hospital, New Zealand, with chest pain, were prospectively investigated between November 2006 and April 2007. Christchurch hospital is an academic district general hospital, with the only emergency department for a regional population of 450,000. Patients were eligible for inclusion if the attending clinician had sufficient suspicion of ACS that serial troponins and electrocardiograms (ECGs) were deemed necessary to objectively investigate this diagnosis. Exclusion criteria were age less than 18 years and if sample for storage had not been obtained at both time points. Serial cardiac troponins, 2nd generation Abbott Architect troponin I (TnI 2) were measured at baseline (0 h) and follow-up (6–24 h). After routine testing, the remaining sample was frozen at −80°C and stored for later measurement of the assays under investigation, Roche high sensitivity troponin T (hsTnT), Abbott Architect troponin I 3rd generation (TnI 3), Roche troponin T (TnT) and Abbott CKMB mass (CKMB).
The patients were investigated and treated as per standard care and as such the study was classified as an audit according to institutional guidelines. The study protocol was approved by the Upper South A Regional Ethics Committee of the New Zealand Ministry of Health who did not explicitly deem it necessary to obtain informed consent. The TnI 3 assay is the current in-house troponin assay; reagents for the TnT and hsTnT assays were donated by the manufacturer (Roche Elecsys), who had no role in the design of the study, the analysis of the data, the preparation of or decision to submit the manuscript.
Reference standard (adjudication)
Investigation was at the discretion of the attending physician. ECGs and results of other investigations including stress testing (exercise tolerance testing or dobutamine stress echo) and coronary angiography were reported independently by cardiologists (authors SJA, IGC, JE, JGL) blinded to patient history or any investigation result. Final diagnoses were made independently by a cardiologist (author SJA) and a second cardiologist (author IGC) in a subgroup of 50 patients (100% agreement) with knowledge of the TnI 2 and the above investigation reports but independently of the troponin assays under investigation.
All investigations and diagnoses were reported using a predefined structured adjudication process. Patient risk factors and diagnoses were largely based on ACC definitions 2001 18 and the redefinition of AMI in 2007 1 amended to include results of coronary angiography and to contend with patients seen in operational practice (Table 1). The dynamic change in TnI 2, in those with elevated values to denote a rise and/or fall in TnI 2 as per definition of AMI, 1 was actively considered; however, if no rise or fall existed but no clear alternative cause of the troponin elevation was apparent, then an adjudication of AMI was made.
Definitions
AMI, acute myocardial infarction; ACS, acute coronary syndrome; ECG, electrocardiogram
Biochemical assays
Blood samples were collected in lithium heparin tubes at 0 and 6–24 h after presentation and sent for immediate analysis of TnI 2. Remaining sample was later analysed for the index assays. The assays are as follows with cut-points as specified by the manufacturer:
TnI 2 (2K41–30, 2000 test kit)/TnI 3 (2K41-38, 500 test kit)
hsTnT (05092744190 Troponin T hs Elecsy – 200 test kit) – Roche Diagnostics, Indianapolis, IN, USA. Elecsys system. LOD 0.005 μg/L, 99th percentile 0.014 μg/L, CV <10% at 0.013 μg/L (reported to 3 decimal places).
TnT (04660307190 Troponin T STAT Gen – 100 test kit)
CKMB mass (2K42-20, 400 test kit)
Statistical analysis
Troponin results were compared for their diagnostic utility using cut-points of the 99th percentile, 10% CV, LOD and optimum cut-points as demonstrated by receiver operating characteristic (ROC) curve analysis (=minimum of [((1 − sensitivity)
2
+ specificity
2
)0.5]). Sensitivities and specificities were calculated in those with troponins ≥99th percentile for a ‘delta’ defined as a % change in level of troponin from baseline to follow-up sample of 20% and 50%. Sensitivities and specificities were compared using the McNemar test. Continuous variables are presented as medians (interquartile range) and categorical variables as numbers and percentages. ROC curves were constructed to assess diagnostic accuracy for the diagnosis of AMI. Areas under the ROC curve (AUC) were then compared. All hypothesis testing was two-tailed and P values of less than 0.05 were considered to indicate statistical significance. All statistical analyses were performed using SPSS for windows (
Results
A total of 1479 patients with chest pain presented in that time period, of whom 332 eligible patients with sufficient sample to measure all four assays at both time points were investigated (Figure 1). The patients were identified consecutively but because of the high exclusion rate, they were not considered consecutive by final analysis. Baseline characteristics are shown in Table 2.
Recruitment ED, emergency department; ACS, acute coronary syndrome
Patient characteristics
AMI, acute myocardial infarction; IHD, ischaemic heart disease; NA, not applicable
The adjudicated index diagnosis was AMI in 110 (33.1%), definite unstable angina in 42 (12.7%), presumed unstable angina in 15 (4.5%), other cardiovascular diagnoses in 30 (9.0%), non-cardiac chest pain in 88 (26.5%) and undifferentiated chest pain in 47 (14.2%).
Diagnostic accuracy for baseline troponins
ROC curves for the diagnosis of AMI are shown in Figure 2. The test performance for the diagnosis of AMI, as quantified by the AUC, is shown in Table 3. For comparison of the three assays, hsTnT and TnI 3 outperformed TnT (P < 0.001 for both) but were equivalent to each other (P > 0.1). The AUC for the TnI 2 assay (and that used for adjudication) at baseline was 0.93 (0.90–0.96).
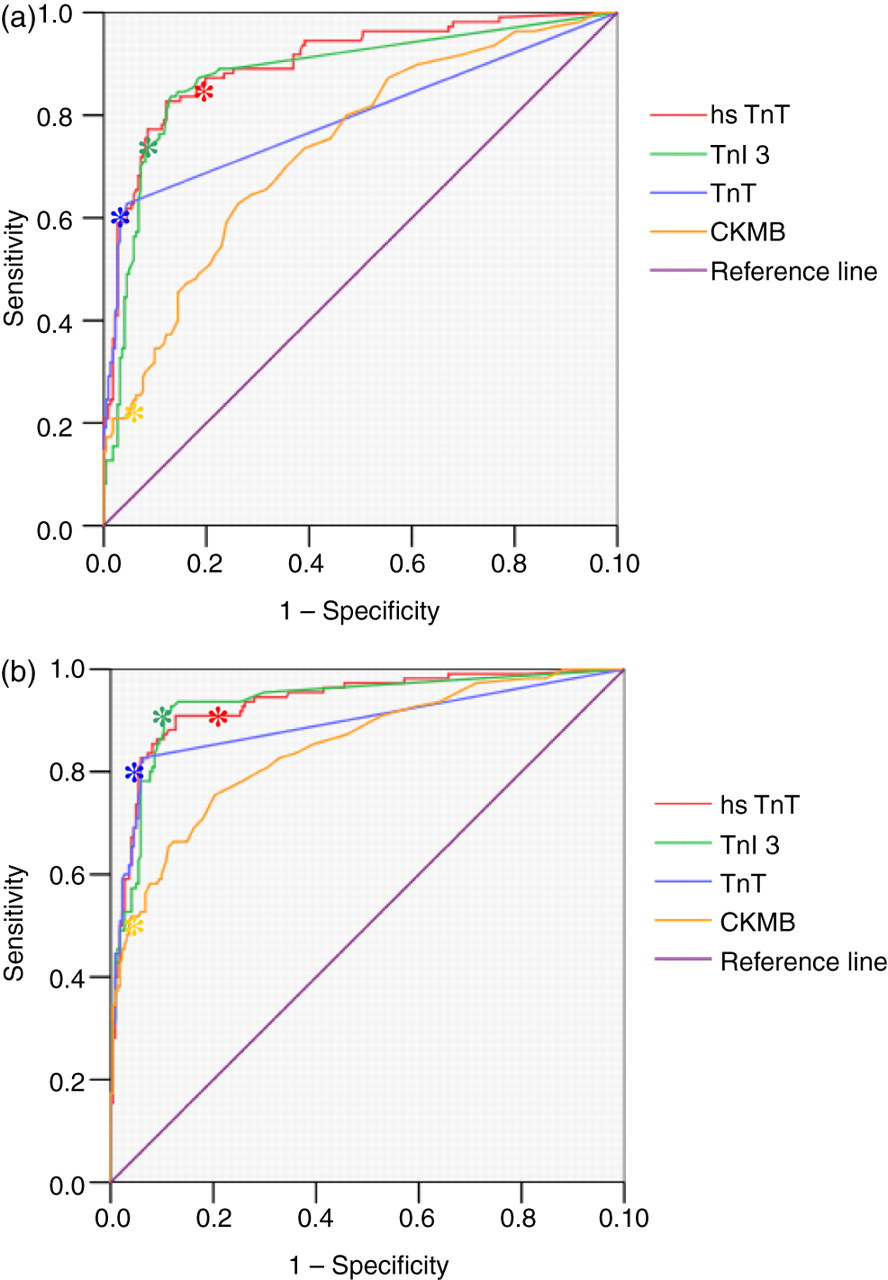
ROC curves showing the diagnostic accuracy of cardiac markers for AMI. Corresponding areas under the curve (AUC) are shown in Table 2. (a) Baseline markers. (b) Follow-up markers. ROC, receiver operating characteristics; AMI, acute myocardial infarction *99th percentile
ROC curve generated AUC for baseline and follow-up troponins
AUC, area under the curve; ROC, receiver operating characteristic; CI, confidence interval
Sensitivities and specificities of the baseline troponins using different cut-points including the 99th percentile, LOD, 10% CV and ROC curve-derived optimums are depicted in Table 4. Comparing sensitivities using the 99th percentile at baseline, hsTnT was more sensitive than both TnI 3 (P = 0.031) and TnT (P < 0.001) and TnI 3 was more sensitive than TnT (P = 0.004). Comparing specificities, TnI 3 (P = 0.020) and TnT (P < 0.001) were both more specific than hsTnT and TnT was more specific than TnI 3 (P = 0.019). The sensitivity of TnI 2 at baseline was 78.7% (75.6–80.0).
Sensitivities and specificities using different cut-points of troponin assays at baseline
CV, coefficient of variation; LOD, level of detection; ROC, receiver operating characteristic; PPV, positive predictive value; NPV, negative predictive value
Data were available for time of symptom onset for 88 of the 110 patients with AMI. The baseline sample was taken a median of 4.0 h (interquartile range 2.0–8.6) from symptom onset. A breakdown of these results into two-hour intervals of time from symptom onset to baseline sample, shown in Figure 3a, suggests how hsTnT is raised in patients with AMI at earlier time points than both TnT and TnI 3.
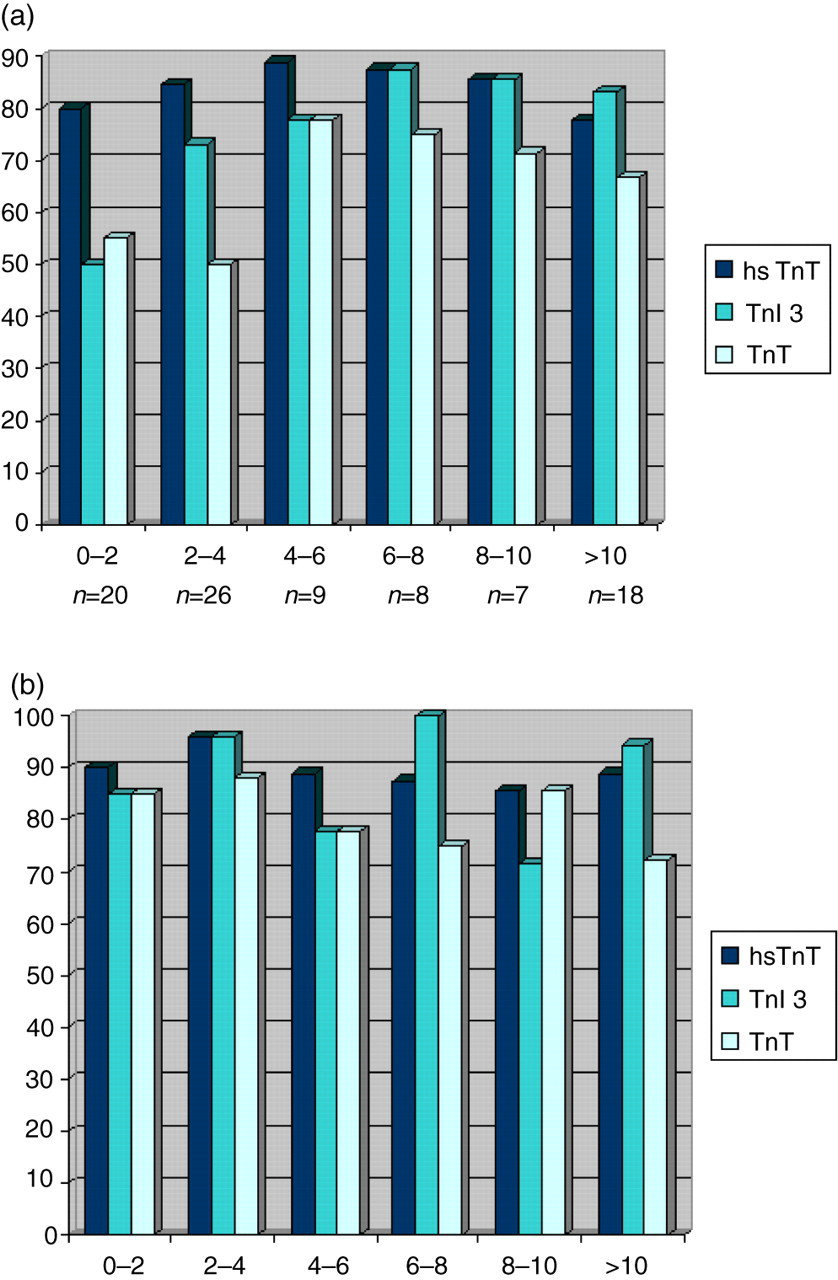
Proportion of patients with AMI having markers ≥99th percentile categorized by time from symptom onset to presentation. (a) Baseline sample. (b) Follow-up sample (6–24 h). AMI, acute myocardial infarction
Diagnostic accuracy for serial cardiac troponins
The AUC for follow-up markers are shown in Table 3. Test performance increased at the later sampling point for all assays with follow-up AUCs outperforming baseline AUCs for hsTnT (P = 0.049), TnI 3 (P = 0.005) and TnT (P < 0.001). Again hsTnT and TnI 3 (P = 0.013 for both) performed better than TnT but were statistically equivalent to each other (P > 0.1). The AUC for the TnI 2 assay (and that used for adjudication) at follow-up was 0.97 (0.95–0.99).
Sensitivities and specificities for follow-up troponins based on values ≥99th percentile are 90.9% (85.3–94.7)/81.5% (78.7–83.4) for hsTnT, 90.9% (85.5–94.6)/88.3% (85.6–90.1) for TnI 3 and 82.7% (77.3–86.8)/93.7% (91.0–95.7) for TnT. Comparing the sensitivities of the assays at follow-up, both hsTnT (P = 0.004) and TnI 3 (P = 0.012) are more sensitive than TnT but equivalent to each other. Both TnI 3 (P = 0.014) and TnT (P < 0.001) are more specific than hsTnT and TnT is also more specific than TnI 3 (P = 0.002).
The follow-up sample was taken a median of 9.4 h (interquartile range 6.3–13.3) from presentation. Patients categorized by time from onset to presentation and follow-up troponin results are shown in Figure 3b.
For hsTnT, there were nine patients with an adjudicated diagnosis of AMI who did not have any measurement ≥99th percentile. These patients had a mean peak TnI 2 of 0.059 μg/L. Two patients did not have coronary angiograms. Of the seven remaining patients, four had angiograms revealing at least one culprit stenosis (2 were revascularized) and three had angiograms with <50% stenoses. For TnI 3, there were 10 patients with an adjudicated diagnosis of AMI who did not have any measurement ≥99th percentile; these patients had a mean peak TnI 2 of 0.065 μg/L. Four patients did not have coronary angiograms. Of the six remaining patients, four had angiograms revealing at least one culprit stenosis (two were revascularized) and two had angiograms with <50% stenoses. For TnT, there were 17 patients with an adjudicated diagnosis of AMI who did not have any measurement ≥99th percentile; these patients had a mean peak TnI 2 of 0.072 μg/L. Five patients did not have coronary angiograms. Of the 12 remaining patients, six had angiograms revealing at least one culprit stenosis (four were revascularized) and six had angiograms with <50% stenoses; the diagnosis of AMI was a default diagnosis in these instances as no alternative cause for a troponin rise was found. There was only one patient with AMI but no rise and/or fall in troponin; this patient also had a negative angiogram, and again the diagnosis of AMI was a default diagnosis.
Troponin concentrations in patients with an adjudicated diagnosis of unstable angina
Of the 57 patients adjudicated as unstable angina on the basis of the TnI 2 results, 10 (17.5%) had values ≥99th percentile for hsTnT, three (5.3%) had values ≥99th percentile for TnI 3 and one (1.8%) had a value ≥99th percentile for TnT.
Troponin concentrations in patients with other diagnoses
Of the 30 patients with objective evidence of an alternative diagnosis, 15 (50%) had peak values ≥99th percentile for hsTnT; these diagnoses included seven arrhythmias, two cardiomyopathies, two heart failures, one valvular heart disease, one pericarditis, one aortic dissection and one sepsis. Fourteen (46.7%) had peak values ≥99th percentile for TnI 3, including eight arrhythmias, two heart failures, one valvular heart disease, one pericarditis, one aortic dissection and one sepsis. Nine (30%) had values ≥99th percentile for TnT, including five arrhythmias, one valvular heart disease, one heart failure, one aortic dissection and one sepsis.
Diagnostic accuracy for delta cardiac troponins
Sensitivities and specificities of the assays using a change in troponin from baseline to follow-up (delta) of 20% and 50% in conjunction with ≥1 measurement of troponin ≥99th percentile are shown in Table 5. The time from baseline to follow-up sample was variable, with follow-up samples taken a median time of 9.4 h (interquartile range 6.3–13.3) after the baseline sample. The improvement in specificity in absolute terms, using a 20% delta in those with peak values ≥99th percentile for hsTnT, identified 22 fewer patients without ACS at a cost of missing 21 patients with AMI. For TnI 3, this method identified 10 fewer patients without ACS at a cost of missing 17 patients with AMI, and for TnT it was five versus 12 patients.
Sensitivities and specificities incorporating delta troponins
Discussion
In this study, we report the diagnostic utility of a contemporary high-sensitivity troponin T assay compared with a third generation troponin I and fourth generation troponin T assay, for the evaluation of patients with chest pain and assess the implications of improving analytical sensitivity on specificity. We also demonstrate challenging data, suggesting that the use of a ‘delta’ to increase specificity of high-sensitivity assays has significant limitations.
The findings of this study are comparable with other studies, showing that when the 99th percentile was utilized, higher sensitivity assays (Abbott Architect troponin I, Roche hsTnT, Roche troponin I, Siemens troponin I Ultra, Vitros troponin I, Beckman Coulter high sensitivity troponin I) were equivalent to each other but outperform assays previously used routinely in practice, some of which may be less sensitive and some of which appear less sensitive because they are not used with a cut-point of the 99th percentile. 3,5,9,11–13,19,20 Also comparable with data from this study, Reichlin et al. 11 demonstrated that the hsTnT assay did not outperform contemporary assays with enhanced analytical sensitivities.
The higher sensitivity assays were more likely to be increased at baseline in those with AMI; this observation is confirmed in other studies 3,4,6,13,20,21,22 with reported sensitivities of 61.5–90.7% on admission. 3,5,6,9,13,20 Furthermore, this study demonstrates how hsTnT appeared to be positive at much earlier time points after symptom onset than the other two assays with 80% patients with AMI having a hsTnT ≥99th percentile within two hours of symptom onset (the number of patients in this subset is small however). Studies confirm that the advantage of using assays with higher sensitivity is most pronounced when comparing results from baseline samples and in those presenting early after symptom onset. 10,11 This has implications both for management of those with early positive values but also results in an improved dependability of early negative results.
The sensitivity and overall test performance (AUC) of the baseline sample for TnI 2 was high but because it was serial TnI 2 measurements that were used to adjudicate the diagnosis of AMI, this introduces incorporation bias and test performance was therefore likely overestimated.
The sensitivity of these newer assays may be even more superior than calculated in this study as many of the patients with an adjudication of AMI had coronary angiograms without stenoses of >50% or indeed had no angiogram for confirmation, and was a default diagnosis as no alternative cause for the troponin elevation was found. If these patients were recategorized as no definite AMI, the number of ‘missed’ AMIs would be smaller and the total proportion of patient with AMI identified would be greater.
Given that concordance with the assays under investigation and the older TnI 2 used for adjudication was not 100%, it is difficult to ascertain when the optimum timing for troponin measurement should be. Assuming an approximate concordance of 90%, the time to achieving a sensitivity of 90% at presentation is 4–6 h from onset of symptoms for hsTnT, 6–8 h for TnI 3 and not at all for TnT. Using the follow-up sample, which is taken at least six hours after presentation, hsTnT achieves a 90% sensitivity even if patients presented within two hours of symptom onset and TnI 3 achieved 90% sensitivity if the patients presented over four hours of symptom onset. Again TnT did not achieve a 90% sensitivity. This may suggest that samples should be taken on presentation and at 4–6 h for hsTnT and on presentation and at 8–10 h for TnI 3 and that TnT does not achieve satisfactory sensitivity. The timing of the second measurement was variable, however, and hence the suggested timings are estimates only.
There are concerns regarding reduced specificity in assays with higher analytical sensitivity and the inherent risk put to patients who undergo invasive investigations and potent treatments who then are discovered to have ‘falsely’ elevated biomarkers. Specificities for hsTnT (defined using values ≥99th percentile irrespective of dynamic changes) were low in this study, although were calculated using an older assay for adjudication and are consequently likely to be underestimates. As shown, some patients previously classified as unstable angina, have troponin values ≥99th percentile, could now be considered for reclassification to AMI. If we were to make this reclassification, the specificities would improve. For example, 17.5% patients with unstable angina had hsTnT ≥99th percentile; if these patients were now reclassified as AMI (regardless of presence or absence of a relevant change in hsTnT), the specificity of hsTnT would improve from 80.6% to 84.4%. Other authors have already considered re-categorizing these patients, for example, Casals et al. 10 found a re-categorization rate of 10%, Giannitsis et al., 13 29% and Keller et al., 9 22.1%. Troponin is also appropriately raised in other conditions, which again affects the specificity; the higher sensitivity assays are more likely to have raised values in patients with these other conditions.
As previously stated, there have been recommendations to use the dynamic change in troponin to signify that a raised troponin is due to AMI. 4,8,16,22 A delta of greater than both the biological and analytical variability of the troponin assay is deemed the most appropriate. Wu 16 demonstrated that biological variability of a cardiac troponin I assay was as much as an increase of 46% or decrease of 32% from the previous measurement and Apple et al. 23 suggested this has reduced to 15–20% with newer assays-Giannitsis et al. 13 determined the ROC curve derived optimum delta values to be as high as 117% for hsTnT taken at zero and three hours and 243% for hsTnT taken at zero and six hours. However, other acute cardiac conditions such as arrhythmia or acute cardiac failure may also produce significant changes in troponin and therefore a dynamic change in troponin is not specific to AMI alone. 16,18 Nevertheless, the use of a delta troponin has been proposed to improve specificity for AMI in these assays. This study confirmed that a delta troponin of ≥20% increased the specificity for AMI significantly and this finding has been corroborated by Apple et al. 10 and Giannitsis et al. 13 found that specificity was increased by using a delta value above the ROC curve determined optimum. However, the use of delta troponins also reduces sensitivity, and this study shows that there are a significant number of patients with missed AMIs by the use of a delta ≥20%. The reduction in sensitivity has been confirmed in other studies; 20,22,24 Casals et al. 22 found that the ‘positive rate reduced from 38.4% (defined by any troponin ≥99th percentile) to 23.2% (using a delta of ≥20%). Giannitsis et al. 13 found that sensitivity for AMI reduced from 100% to 84.6% when using a delta of 20% and from 100% to 69.2% when using a delta calculated using ROC curve analysis.
The data calculated here assessing the value of a delta troponin have limitations. For example, patients presented at variable times from symptom onset to presentation and there were variable time intervals between the baseline and follow-up samples. The high variability of these timings in conjunction with the fact that the rise and fall in troponin may be unpredictable in those with non-ST elevation AMI, means that the degree of apparent change in troponin values may be underestimated. Most other studies evaluating a delta criterion have used follow-up troponin measurement between one and six hours after presentation. 9,12,13 Another consideration is that patients may have significant coronary artery disease but their presentation with chest pain on the index event may not be due to ischaemia; this would therefore lead to positive stress testing or coronary angiography, but low delta troponin values. Also, we have shown that some of the patients adjudicated as AMI did not have culprit stenoses found on angiography; it may be that these patients had an alternative cause for their troponin elevation that was not demonstrated and therefore a significant rise or fall would not occur. Despite these limitations, it is evident that use of a delta improves specificity and its value will likely be in patients with atypical symptoms or in those with other co-morbidities or diagnoses present; however, clinical acumen is still required to prevent those with true AMI but without significant calculated dynamic change in troponin, from being overlooked. As a rise or fall in troponin is required by definition to make the diagnosis of AMI, 1 more research is required to ascertain the optimum timing for serial measurement and the degree to which a troponin must rise or fall may well be different between assays.
Conclusions
The higher sensitivity troponin assays, hsTnT and TnI 3, outperformed the assay with the lowest analytical sensitivity, TnT, comparable with other studies. hsTnT and TnI 3 were equivalent for test performance and overall sensitivities, again consistent with other studies. The advantage of switching to a specialized high-sensitivity assay is that patients can be diagnosed earlier, even early after symptom onset and by corollary, may also better rule out patients without AMI at an earlier time. The disadvantage of switching to a specialized high-sensitivity assay is the increased uncertainty that a raised troponin is due to an AMI rather than other conditions. The use of delta troponin calculations improves specificity though at the expense of reduced sensitivity, which would suggest that further research into such an approach is warranted and that clinical acumen should not be under-valued.
Limitations
In addition to the limitations already described, this is a relatively high-risk population with AMI rates higher than that seen in our institution from audit data, where AMI rates are approximately 20–25% and ACS rates 30–35%, suggesting a selection bias although the overall frequency of the diagnosis of AMI in the excluded population is not known. This may also introduce a spectrum bias leading to an over-estimation of overall test performance. This possibility should be taken into consideration if applying our findings to a lower risk population. Although it is known that a significant proportion of patients with ACS or AMI present with symptoms other than chest pain, these patients were not included for this study, which should be taken into account if applying our findings to patients without chest pain. The timing of follow-up samples was highly variable, although this would be consistent with real-world clinical practice. The study design involved the evaluation of newer troponin assays with a gold standard that is a previously validated troponin assay. This results in a circular argument when calculating diagnostic utility of the assays under investigation. Performance characteristics would change if evaluated against similar newer assays, as discussed earlier.
DECLARATIONS
