Abstract
Background
Primary hyperparathyroidism (PHP) is the most common cause of hypercalcaemia, and often requires surgical treatment. Familial hypocalciuric hypercalcaemia (FHH) has similar biochemical features to PHP, but requires no treatment. The most common biochemical method used to distinguish between PHP and FHH is the urine calcium-to-creatinine ratio (UCCR). Vitamin D deficiency may alter the renal excretion of calcium, but it is unclear how vitamin D deficiency affects the diagnostic performance of UCCR.
Aim
To examine the reliability of UCCR to detect PHP in patients presenting with asymptomatic hypercalcaemia, in the presence or absence of vitamin D deficiency.
Methods
One hundred and eighteen UCCR measurements from 97 asymptomatic hypercalcaemic patients diagnosed with PHP presenting to a single specialist endocrine unit were analysed retrospectively.
Results
A significantly higher proportion of UCCR measurements were <0.010 in patients with serum vitamin D <25 nmol/L when compared with patients with serum vitamin D >25 nmol/L, thus incorrectly suggesting the presence of FHH (proportion of measurements with UCCR >0.010: 11/48 [22.9%], vitamin D <25 nmol/L; 4/70 [5.7%], vitamin D >25 nmol/L; P < 0.001). Urine calcium concentration was 26% lower and serum parathyroid hormone (PTH) was 27% higher in patients with vitamin D deficiency when compared with patients without vitamin D deficiency.
Conclusions
These data suggest that the presence of vitamin D deficiency is associated with worsened PTH hypersecretion, impairment of urinary calcium excretion and reduced sensitivity of UCCR measurement with respect to the detection of PHP. These data have important clinical implications for the investigation and management of patients with asymptomatic hypercalcaemia.
Introduction
Hypercalcaemia is a common biochemical abnormality usually arising in the absence of clinical symptoms. The most common cause of hypercalcaemia in asymptomatic healthy patients is primary hyperparathyroidism (PHP), which arises from the neoplastic or hyperplastic growth of one or more parathyroid glands. 1 Parathyroidectomy restores normal serum calcium levels in patients with PHP, and reduces the risk of long-term complications such as nephrolithiasis and oesteodystrophy. However, parathyroid surgery is associated with a risk of vocal cord palsy and postsurgical hypocalcaemia.
Other important causes of hypercalcaemia must be considered in asymptomatic individuals. Familial hypocalciuric hypercalcaemia (FHH) is characterized by mutations of the calcium sensing receptor leading to reduced renal calcium clearance and hypercalcaemia. 1 Unlike PHP, FHH is a benign condition without associated end-organ complications; hence, affected individuals do not require treatment. It is imperative to identify accurately which asymptomatic hypercalcaemic patients have PHP. Patients with PHP and FHH may have similar biochemical serum measurements of calcium, phosphate, magnesium and parathyroid hormone (PTH). The most common biochemical method used to distinguish between PHP and FHH is the urine calcium-to-creatinine clearance ratio (UCCR). 1,2 Previous studies suggest that a UCCR < 0.010 indicates FHH, and makes the diagnosis of PHP unlikely. 3
Vitamin D is a regulatory hormone that promotes bone mineralization and gut calcium absorption. Vitamin D deficiency is common in Northern Europe and the USA, 4 and has a higher prevalence in patients with PHP when compared with normocalcaemic controls. 5,6 Renal excretion of calcium may become reduced during vitamin D deficiency. Therefore, the presence of vitamin D deficiency in a patient with hypercalcaemia may affect UCCR measurement. However, it is unclear to what extent the presence of vitamin D deficiency affects the diagnostic performance of UCCR. We examined the reliability of UCCR to detect PHP in patients presenting with asymptomatic hypercalcaemia, in the presence or absence of vitamin D deficiency.
Methods
One hundred and eighteen UCCR measurements from 97 hypercalcaemic patients diagnosed with PHP presenting to a single specialist endocrine unit were analysed retrospectively. Seventy patients had parathyroidectomy with histological features of PHP and normocalcaemia postsurgery. The remaining 27 patients had an elevated serum PTH, hypophosphatemia and ultrasound or 99mtechnetium-sestamibi localization of parathyroid adenoma. Four additional patients were excluded from the study; three patients were on lithium or thiazide medication and one patient had an estimated glomerular filtration rate below 30 mL/min/1.73 m2. The mean age was 65 y (range 24–92 y) and 26 patients were men. Paired blood and 24 h urine collections were performed in all patients and used to calculate the UCCR as follows: [urine Ca (mmol/L)× serum creatinine (μmol/L)/1000]/[adjusted serum Ca (mmol/L)× urine creatinine (mmol/L)]. 1,2 Hypercalcaemia was defined as albumin-adjusted serum calcium above 2.6 mmol/L. The range of serum albumin observed in patients with PHP was 25–48 g/L. Serum 25-hydroxy (25OH) vitamin D was measured using Diasorin Liason Automated Immunoassay (Diasorin S.p.A, Crescentino, Italy). Plasma PTH was measured using Siemens Immulite immunoassay (Siemens Healthcare Diagnostics, Deerfield, IL, USA). A P value < 0.05 indicated statistical significance. Prism 5.0 software (Graphpad Inc, La Jolla, CA, USA) was used to calculate Pearson's correlation coefficient, compare mean values using unpaired t-tests and perform Fisher's exact test in order to analyse contingency tables.
Results
The range of UCCR observed in patients with PHP was 0.0028–0.0624 (Figure 1). Mean serum creatinine was 86 μmol/L (range 55–161 μmol/L). The mean age of patients was similar in those with a UCCR >0.010 and those with a UCCR <0.010: 64 ± 1.5 y versus 68 ± 3.1 y; P = 0.31.
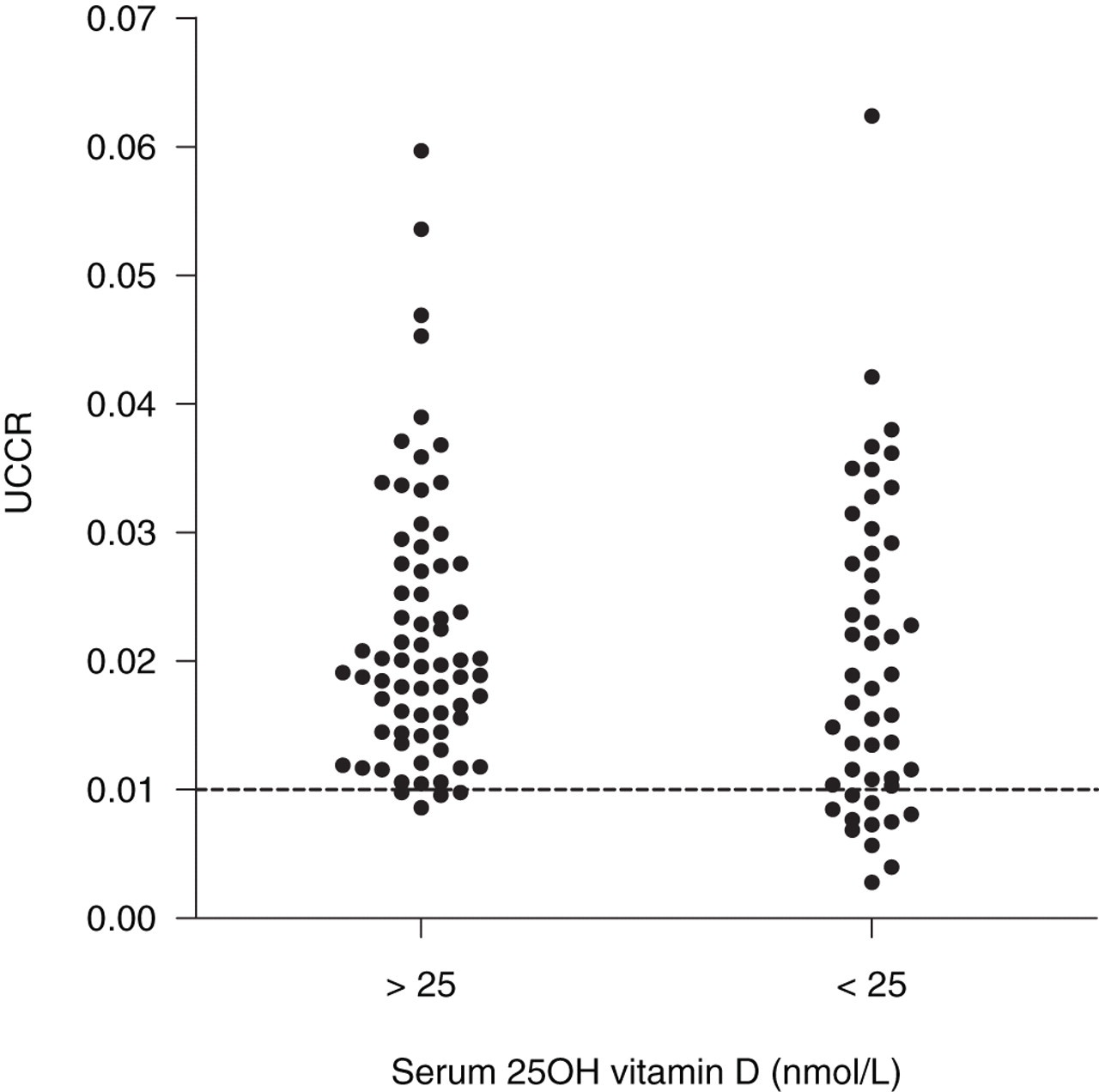
Scatter plot graph comparing urine calcium-to-creatinine clearance ratio (UCCR) for patients diagnosed with primary hyperparathyroidism with serum 25OH vitamin D levels either above 25 nmol/L or below 25 nmol/L. A cut-off line is displayed for UCCR = 0.010
Forty-eight of the 118 UCCR measurements were taken from PHP patients with serum 25OH vitamin D <25 nmol/L. Patients with serum 25OH vitamin D <25 nmol/L had a significantly higher serum PTH when compared with patients with serum 25OH vitamin D >25 nmol/L (21.6 ± 2.39 ng/L, 25OH vitamin D <25 nmol/L versus 15.7 ± 1.58 ng/L, 25OH vitamin D > 25 nmol/L; P < 0.05) (Table 1). A significantly higher proportion of UCCR measurements incorrectly suggested the presence of FHH (UCCR <0.010) in patients with serum 25OH vitamin D <25 nmol/L when compared with patients with serum 25OH vitamin D >25 nmol/L (proportion of measurements with UCCR <0.010: 11/48 [22.9%], 25OH vitamin D <25 nmol/L; 4/70 [5.7%], 25OH vitamin D >25 nmol/L; P < 0.001) (Figure 1). Pearson's correlation coefficient between serum 25OH vitamin D and UCCR was 0.42 (P = 0.75).
Comparison of patient age, serum and urinary biochemical markers between cases of primary hyperparathyroidism (PHP) associated with a serum 25OH vitamin D <25 nmol/L versus cases of PHP associated with serum 25OH vitamin D >25 nmol/L
PTH, parathyroid hormone
Values are expressed as mean ± SEM
We next examined individual serum or urinary biochemical parameters in the presence or absence of serum vitamin D deficiency (Table 1). Patients with serum 25OH vitamin D <25 nmol/L when compared with patients with serum 25OH vitamin D >25 nmol/L had significantly lower urine calcium concentrations but higher serum PTH and serum calcium concentrations. There were no significant differences between the two groups in 24 h urine calcium excretion, serum phosphate and serum magnesium concentrations. Patients with serum 25OH vitamin D <25 nmol/L were on average 6.1 y older but had similar levels of serum and urine creatinine when compared with patients with serum 25OH vitamin D >25 nmol/L.
Discussion
Clinical guidelines recommend that the UCCR cut-off 0.010 is highly sensitive for the detection of PHP, and that UCCR measurement should be performed in all hypercalcaemic patients. 1,2 Indeed, a recent systematic review suggested that a UCCR <0.010 excluded PHP in 88% of cases. 3 However, some of the studies included in this meta-analysis included patients with coexistent vitamin D deficiency, which might have affected UCCR through a reduction in renal calcium excretion. It is therefore important to examine if the diagnostic performance of UCCR measurement is affected by coexistent vitamin D deficiency.
We observed that UCCR measurement incorrectly suggested the diagnosis of FHH at a fourfold higher rate (22.9% versus 5.7%) in patients with vitamin D deficiency (serum 25OH vitamin D <25 nmol/L) when compared with patients without vitamin D deficiency (serum 25OH vitamin D >25 nmol/L). Furthermore, we observed that urine calcium concentration was 26% lower and serum PTH was 27% higher in patients with vitamin D deficiency when compared with patients without vitamin D deficiency. These data suggest that the presence of vitamin D deficiency reduces the sensitivity of UCCR measurement with respect to the detection of PHP. Furthermore, vitamin D deficiency may worsen the hypersecretion of PTH associated with PHP, which may in turn suppress urinary calcium excretion. It is therefore possible that the impaired diagnostic performance of UCCR measurement observed in vitamin D deficiency with respect to detection of PHP is a consequence of suppressed urinary calcium excretion. A lower UCCR threshold than 0.010 may improve its sensitivity for the detection of PHP in patients with vitamin D deficiency; however, this might risk the misclassification of patients with FHH and exposing them to the risk of unnecessary parathyroidectomy.
It is important to recognize that we observed significant overlap between UCCR values in patients with a serum 25OH vitamin D <25 and >25 nmol/L. Furthermore, we observed no overall correlation between serum 25OH vitamin D and UCCR. No significant differences in serum creatinine and age were observed between patients with a UCCR <0.010 and patients with a UCCR >0.010. However, it is likely that UCCR measurement is influenced by factors other than serum 25OH vitamin D.
Patients with PHP and vitamin D deficiency have significantly higher serum PTH and serum alkaline phosphatase, and significantly lower femoral neck and forearm bone mineral density when compared with patients with PHP and no coexistent vitamin D deficiency. 5,7,8 It has therefore been argued that vitamin D replacement may reduce the bone disease associated with PHP. 6 Recent evidence suggests that vitamin D replacement does not increase mean serum calcium in patients with PHP-induced hypercalcaemia, and is safe under medical supervision. 6,9 Furthermore, our results suggest that serum calcium was marginally higher in patients with vitamin D deficiency, which has been observed previously. 10
In conclusion, our results suggest that diagnostic sensitivity of the UCCR with respect to detection of PHP is higher in asymptomatic hypercalcaemic patients with a serum 25OH vitamin D >25 nmol/L when compared with asymptomatic hypercalcaemic patients with a serum 25OH vitamin D <25 nmol/L. These observations may be explained by worsening of PTH hypersecretion and impairment of urinary calcium excretion in patients with PHP and coexistent vitamin D deficiency. Vitamin D supplementation may therefore improve the diagnostic sensitivity of the UCCR with respect to detection of PHP, in asymptomatic hypercalcaemic patients with serum 25OH vitamin D <25 nmol/L. These data have important clinical implications for the investigation and management of patients with asymptomatic hypercalcaemia.
DECLARATIONS
