Abstract
Background
Immunoassays are susceptible to analytical interferences including from endogenous immunoglobulin antibodies at a rate of ∼0.4% to 4%. Hundreds of millions of immunoassay tests (>10 millions in the UK alone) are performed yearly worldwide for measurements of an array of large and small moieties such as proteins, hormones, tumour markers, rheumatoid factor, troponin, small peptides, steroids and drugs.
Methods
Interference in these tests can lead to false results which when suspected, or surmised, can be analytically confirmed in most cases. Suspecting false laboratory data in the first place is not difficult when results are gross and without clinical correlates. However, when false results are subtle and/or plausible, it can be difficult to suspect with adverse clinical sequelae. This problem can be ameliorated by using a probabilistic Bayesian reasoning to flag up potentially suspect results even when laboratory data appear “not-unreasonable”.
Results
Essentially, in disorders with low prevalence, the majority of positive results caused by analytical interference are likely to be false positives. On the other hand, when the disease prevalence is high, false negative results increase and become more significant. To illustrate the scope and utility of this approach, six different examples covering wide range of analytes are given, each highlighting specific aspect/nature of interference and suggested options to reduce it.
Conclusion
Bayesian reasoning would allow laboratorians and/or clinicians to extract information about potentially false results, thus seeking follow-up confirmatory tests prior to the initiation of more expensive/invasive procedures or concluding a potentially wrong diagnosis.
False-positive result is a term generally used to indicate a test that suggests the presence of disease when there is none or vice versa for false-negative data. This may arise from the common/accepted practice of establishing a ‘statistical normal/reference’ range for an individual analyte. The quoted reference range for each test (including immunoassays) is obtained by using cut-off points (∼95%) of continuum obtained from ‘normal’ individuals. This statistical truncation causes some false-positive or false-negative calculable data, expressed as predictive false-positive and false-negative rates using the well-established contingency table.
Many hundred millions immunoassay tests, on a wide range of analytes are carried out yearly worldwide. In these tests, the terms false-positive and false-negative are additionally used in a different context, namely when endogenous immunoglobulin antibodies in some patients, unpredictably interfere in immunoassays causing false analytical results. 1–13 This underlying cause must not be confused with false results produced by statistical truncation for establishing reference ranges. A probabilistic reasoning using Bayes' theorem 14 in which both the rate of interference and disease prevalence (i.e. the number of people having the condition in a population) can help understanding the likelihood of false-positive and ‘-negative’ results occurring. Essentially, in disorders with low prevalence, the majority of positive results caused by interference are likely to be false-positives. On the other hand, when the disease has high prevalence, false-negative results increase and become more significant. This starting point, although useful, is not sufficient in its own to formulate a firm decision, namely whether or not to accept a result. Further strategies will be described to illustrate their utilities in enhancing the reliability of decision-making. This is important because clinicians tend to perceive all laboratory data in the same light. All abnormal results including false ones are considered ‘pathological’ necessitating follow-up. 5–8,11,13 Even more seriously, ‘false-negative results’ may wrongly exclude pathology, thus denying patients' necessary treatment. 9–11 The wide variation in accuracy between immunoassays and other common routine laboratory tests such as urea and electrolyte (U&E), liver function test (LFT), full blood count (FBC), lipids, glucose, etc. is not commonly appreciated by general practitioners as well as hospital clinicians.
Interference can lead to falsely high or falsely low results, depending on the immunoassay's format. For uniformity, the incidence of interference in immunoassays from endogenous antibodies in all examples will be assumed to be ±0.4% (literature quoted figures ranged from 0.4% to 4% 1,6,8,12 ). This may also help highlight the impact of analytical error on augmenting clinical error rate which additionally takes disease prevalence/incidence into account. Needless to say that this figure may vary being method dependent both in trend and magnitude.
Thyroid-stimulating hormone in subclinical hypothyroidism
Subclinical hypothyroidism is a disorder with a prevalence ranging from 1% in young adults and children to 17% in elderly women.
15,16
Assuming a false-positive interference rate of 0.4%, the probability of an accurately raised and truly diagnostic thyroid-stimulating hormone (TSH) result (i.e. the positive result indicates that the patient has subclinical hypothyroidism) can be calculated according to Bayes' theorem as follows:
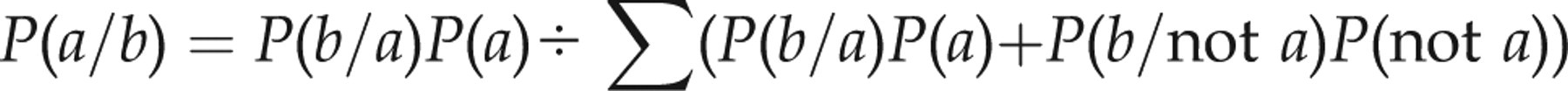
Or simply
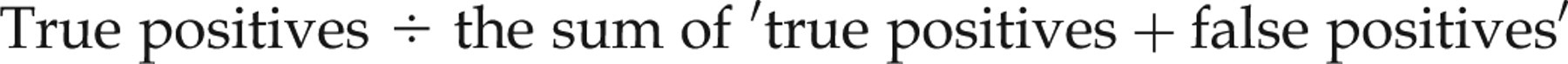
Examples of follow-up confirmatory tests commonly used in detecting interference in immunoassays from immunoglobulins antibodies
Examples of follow-up confirmatory tests commonly used in detecting interference in immunoassays from immunoglobulins antibodies
The pros and cons of screening for prostate cancer by total or free prostate-specific antigen (PSA) or their ratio are outside the scope of this note. 17–21 Screening may be carried out on middle-aged men (∼50 y old) who do not have symptoms but have a high risk of developing the disease (e.g. Africans, African-Caribbean, African-Americans, first-degree relative of a patient diagnosed with prostate cancer at an early age <65 y). Even men with low/average risk may sometimes participate in ‘health/wellness screen’, which includes PSA as a prostate cancer test because early treatment of localized tumour is curative and has a favourable disease-free outcome. In 50-y-old men, prostate cancer is generally rare with a prevalence of ∼1 in 5000 (i.e. 0.02%). Assuming a false-positive analytical rate at 0.4%, the probability that a positive test is false in this ∼50 year's old group is ∼95%. Even in the at-risk group in whom the prevalence may be one order of magnitude higher, e.g. 1 in 500, two-thirds of the positive results are likely to be false-positives. In a recent PSA-based trial involving 126,462 men (mean age 60.8 y; range 50–74 y) from seven European countries, 75.9% of men who underwent biopsy (>13,000) had false elevated PSA results. 21 This figure is surprisingly consistent with the higher Bayesian projection of ∼95% in asymptomatic 50-y-old men in whom the incidence of prostate cancer is much lower being <20% of that found in the ∼61 y olds in the multicentres European study. 21
Because of the low specificity of PSA test, it would be reasonable to authenticate all PSA elevations in asymptomatic middle-aged men free from inflammation and urinary tract infection by further affirmative tests 13,22,23 and before invasive and expensive investigations or actions are scheduled. Refined tests that may help identifying benign prostatic hyperplasia, e.g. circulating free PSA and its ratio to protein-bound PSA may also be false and misleading because antibodies can interfere with both the free analyte and/or its protein-bound moiety. Abnormal outcome in one or more of affirmative tests would be indicative of a false PSA result. Needless to say that in symptomatic/older patients the disease prevalence rises significantly reducing the rate of false-positive results and/or the need to perform follow-up affirmative tests.
hCG as a tumour marker
hCG was the first modern tumour marker used in the detection and management of cancers that produce hCG such as gestational trophoblastic disease (molar pregnancy) and some germ cell tumours (testicular, ovarian and intracranial tumours, e.g. pineal). β-hCG is more specific than intact hCG moiety and can be a valuable and highly sensitive marker for these low-incidence cancers in children, men and non-pregnant women. However, when β-hCG/hCG test is clinically indicated in differential diagnosis, care is needed because a significant proportion of positive β-hCG/hCG results are likely to be false-positive. 24 False-negative results also occur, albeit at a much reduced rate. 24
β-hCG/hCG is normally present in both blood and urine with parallel changes in concentrations. Because serum concentrations are mirrored in urine, a sensitive ‘pregnancy test’ can be simply used to authenticate a raised serum hCG data. Unlike serum, there are essentially no immunoglobulins/antibodies in urine from individuals with normal renal function (the bulk of protein in the urine of normal individuals is a trace amount of albumin with its much lower molecular weight [MW] than immunoglobulins). For this reason, simultaneous measurement of the same analyte in both serum and urine should produce concordant results. On the other hand, dis-concordance with positive serum concentration but none in urine (or vice versa) indicates interference and confirms a false serum result. 24 In a single report, failure to authenticate raised serum β-hCG/hCG in six non-pregnant women has resulted in unnecessary treatment and surgery with significant morbidity. 25,26 In the experience of one of us, simultaneous measurement of hCG in serum and urine was simple, inexpensive and useful tool.
The same approach is applicable to other analytes whose concentrations in serum are reflected in urine, e.g. steroid hormones, drugs of abuse, small peptide, proteins with lower or comparable MW to albumins such as LH, FSH, alpha-fetoprotein (AFP), etc. For example, a perimenopausal English woman had a significantly high serum oestradiol, which was confirmed by repeat serum analysis using different immunoassay platform. This has led to hysterectomy and bilateral ovariectomy. However, serum oestradiol levels remained elevated and unchanged postoperatively. Subsequent analysis by the SAS steroid laboratory in Leeds using an extraction assay revealed undetectable oestradiol levels. Similarly, falsely elevated serum testosterone in two Norwegian women has led to unnecessary laparotomy. 27 Because of the low incidence of these diseases, it appears reasonable that excluding interference prior to invasive/expensive procedures 22,23,28 would be prudent (Table 1). Discrepant results in any of these tests confirm a false-positive result.
Rheumatoid factor in rheumatoid arthritis
Rheumatoid arthritis (RA) is more likely to affect women than men at all ages. The reported incidence of this disease (annual number of patients newly diagnosed) is higher in older women >65 y of age compared with those who are <35 y, being 0.09% versus 0.03%. The corresponding figure for men is even lower being 0.06% and 0.006%, respectively (source UK-ARC). The diagnosis of RA is largely clinical and radiological but serum rheumatoid factor (RF) has a useful role in the diagnosis and management of patients with RA. 11 However, because of the low incidence of RA in both genders and at all ages, the use of RF as a screen for RA can be misleading because a large proportion of positive results (>80%) in these patients are likely to be false-positives.
Although a negative RF is useful, a ‘health warning’ may be attached to RF-positive outcome when the test is used as a screen for patients with low index of suspicion and confirmatory tests (Table 1) may be appropriate in such cases. 22,23 Clinically, RF is the best predictor of aggressive disease in patients with RA. 11 Analytically, identifying patients with true positive RF can also be important because RF itself is a heterophilic antibody 4,10,11,12 with promiscuous binding tendency towards other different molecules/analytes. RF, if present, therefore has the potential to interfere in other tests using immunoassays. Knowledge of accurate RF results is therefore desirable both clinically and analytically.
Troponin in patients with myocardial infarction/coronary syndrome
Bayes' theorem described above for false-positive results can also be applied to assess the probability of false-negative results using the same criteria. When the disease has low prevalence the probability that a negative result is false-negative is very low, and will not be a major problem. The probability of a false-negative, however, would increase significantly when the disease prevalence rises, e.g. serum troponin measurement in patients with symptoms consistent with acute myocardial infarction (AMI)/acute coronary syndrome (ACS). Assuming an interference rate of ±0.4% and the prevalence of MI in symptomatic patients is ∼80%, the probability of a negative test being false-negative can be calculated according to the formula:
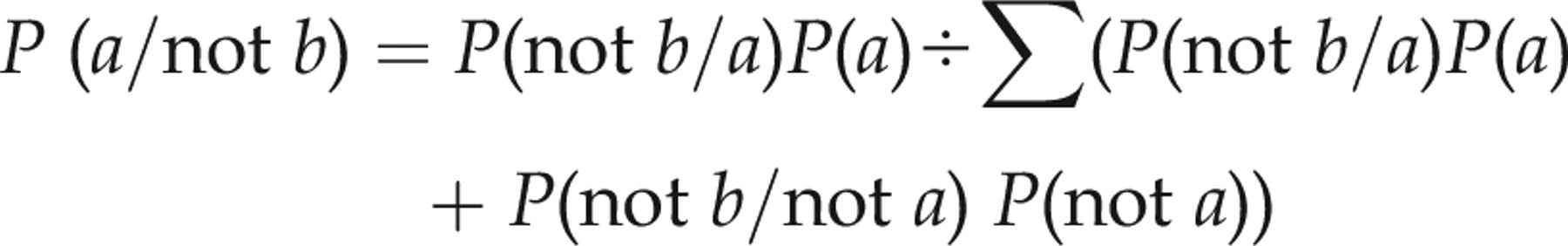

False-positive case reports have been widely reported in the recent literature though interestingly, the number of false-negative case reports is lower. 11,29 It is important also to re-emphasize that the effects of interference, either negative or positive, may persist for a long time depending on the underlying cause that triggered the production of interfering immunoglobulin antibodies. Protocols such as repeat analysis after 6–12 h or 6–18 h from onset may be unhelpful or even misleading if the initial reading is false. This is because interference causing false troponin result persists over this short period of time, reinforcing the initial reading and giving a false sense of reassurance. Falsely low serum troponin is known to occur. 30,31 However, because falsely low results are potentially more serious than the more frequently reported falsely high results, ‘low’ serum troponins in symptomatic patients should be interpreted with caution.
It may be of interest to point out that autoantibodies to cardiac troponins (cTn) were found in 3–6% of healthy control subjects, blood donors and hospitalized patients with or without a history of cardiac disease. 30,32 Further studies 33–39 have provided additional evidence for the presence of these potentially offending and interfering antibodies capable of causing false-negative immunoassay results. A number of small clinical studies 33–39 on patients with AMI/ACS have demonstrated false-negative rates of cTn I ranging from ∼1.5% to 6%; a figure not dissimilar to that projected by Bayesian theorem. In a recent multicentre study for early diagnosis of AMI involving 1818 patients, 40 the clinical sensitivity and specificity of cTn I immunoassay were 90.7% and 90.2%, respectively, regardless of the intervals between the onset of chest pain and admission/analyses. Unfortunately, in this and other similar clinical studies, no follow-up investigations were carried out to assess analytical interference in cTn I analyses and its contribution to the ∼10% loss of accuracy.
Ideally, advice to identify false-negative results for cTn I should be provided by the immunoassay's supplier. However, laboratorians can initiate some confirmatory tests (see Table 1) such as repeat analysis using blocking antibodies or measuring creatine kinase activity enzymatically (non-immunologically) as well as the doubling dilution test, which in some cases of negative interference can paradoxically produce higher results on progressive dilution 6,23 (i.e. reverse linearity). Discrepant results should raise other diagnostic possibilities including false-negative interference. 22,23
Serum insulin, proinsulin and C-peptide in hypoglycaemia
Hypoglycaemia is an uncommon clinical entity. 41 In many cases, the underlying cause may be clinically apparent, e.g. diabetics on medications, alcoholics and septic shock syndrome. In the absence of clinical and/or radiological surrogates however, biochemical investigations are necessary for diagnosis and differential diagnosis. Front-line tests commonly used are serum insulin, proinsulin and C-peptide. Pancreatic islet-cell tumours that produce insulin and/or proinsulin (insulinomas) are rare, being less than 4 per million people per year (0.0004%). Other conditions such as fictitious hypoglycaemia and autoimmune hypoglycaemia are even rarer. With such low incidence, the positive result in one or more parameters is likely to be false-positive. Excluding interference in each of these immunoassays 42 is essential when other clinical or radiological correlates are absent. This is because in this rare but complex disease, four separate antibodies may be present, and more than one may simultaneously occur in the same patient. 42–47 Furthermore, three of these interfering antibodies exhibit a dual role, namely a pathological one causing hypoglycaemia and an analytical one causing false-positive results. 42
The first potential source of interference is heterophilic antibodies that can interfere at random and in unpredictable way (positive or negative) in one or more of the immunoassays of insulin, proinsulin and C-peptide. The other three antibodies are, however, unique because they can interfere in immunoassays and also cause hypoglycaemia (autoimmune) in their own right, namely antibodies that predominantly bind to insulin, bind to proinsulin or mimic insulin action (i.e. receptor's antibodies). Their mode of interference in the immunoassays of insulin, proinsulin and C-peptide immunoassays is different (detailed discussion elsewhere 13,42 ). Confirmatory tests are therefore necessary and described in detail elsewhere 42 to detect interference from any of the other three antibodies, if present. These rare but unique clinical entities highlight the very complex nature of interference and its potential to produce false results confusing diagnosis.
Comments and conclusion
Interference from endogenous antibodies in immunologically based tests such as immunoassays remained pernicious, unpredictable and insidious. 4,9,10,12,48–53 Some endogenous antibodies may be polyreactive especially in early immune responses. 11 Polyreactive heterophile antibodies of the IgM class such as RF tend to have lower specificity, bind to many antigens, potentially causing interference in their measurements. Even more specific antibodies 11 may also exhibit molecular structures on their surface, which may by sheer chance mimic an immunological binding site of the measured analyte (i.e. similar epitope), a binding site on the reagent antibody (i.e. similar paratope), both (i.e. in idiotypic immune response) or to the bound antigen–antibody complex (i.e. metatope). In all such cases, interference may occur, leading to erroneous results (falsely high or low). Screening every sample prior to analysis for interfering antibodies is, however, neither specific nor practical. 13,48,54
Molecular mimicry and lack of mono-specificity of an antigen-antibody reaction is a feature of some autoimmune diseases. Experimentally, it was also a frequent finding since the earliest immunochemical studies. This phenomenon was regarded by molecular immunologists as an intrinsic property by which antibodies participate in degenerate binding to differing antigens to amplify their functional diversity. 13,55 Furthermore, in ‘heteroclitic’ binding reactions, antibodies can bind some of these unintended antigens/analytes with even higher affinity. 53 Studies and conceptual models/mechanisms to explain the paradox of high specificity and broader cross-reactivity remain speculative. It is therefore important to emphasize that despite mesmerizing innovations in measurements by immunoassays such as the use of multiplexed quantum dots nanoparticles to label antigens or antibodies, 56,57 interference will continue to occur, being an inherent part of the very basic antigen–antibody interaction, and hence the utility of probabilistic Bayesian reasoning as an aid to reduce false results (see Table 2).
Take-home message
It may be important to highlight the two main aspects that affect the pattern and duration of interference in measurement carried out by immunoassays, namely the concentrations of interfering antibody (i.e. titres at the time of analysis) and its binding affinity/avidity. Both factors can vary with time; for example, interference causing false results may be transient over months (the half-life of immunoglobulin/antibody is about 1 month) arising from antibodies produced as a result of bacterial/viral infection, recent immunization, treatment with monoclonal antibodies, a blood transfusion or may be chronic in patients exposed to animal proteins (through pets or via occupational exposure), allergic diseases or those with autoimmune disorders. When the underlying cause is transient, interference tends to be variable, rising to a peak before declining or disappearing all together. On the other hand, chronic exposure to the immunogen tends to cause prolonged and persistent production of interfering antibody.
Two components underpin probabilistic Bayesian projection, namely the rate of interference in measurement carried out by immunoassays and disease prevalence/incidence. Informed guesstimates of these two parameters are used in this note to project the likelihood of false results. The figure chosen for the incidence of interference in immunoassays 6 was 0.4%. This seemed reasonable as shown by the good agreement between theoretical Bayesian projections of the false-positive rate of PSA when compared with the actual findings revealed in the largest independent clinical trial. 21 Six dissimilar examples are given to illustrate various options available to reduce false results. The extra laboratory costs of follow-up confirmatory tests are relatively inexpensive (reagents' cost few pounds or less) and this should be perceived in the wider context of avoiding expensive/invasive procedures, wrong diagnosis/management with potential damage to patients' care. It may be of interest to point out that a new law has recently been introduced in France, which views clinical laboratory tests as medical procedures in their own right. The French legislation provides a focus for immunoassay providers and laboratorians to ascertain analytical integrity and quality reporting of diagnostic tests (Journal of the French Republic, 15 January 2010).
Generally, false-positive results must be considered in conditions with low prevalence or incidence when other specific clinical or radiological correlates are absent. Furthermore, the rate of false-positive results is inversely related to disease prevalence/incidence. It is, however, paramount to point out that disease prevalence changes when other surrogate correlates (biochemical, radiological and/or clinical) strongly suggest disease presence, even if it is rare with low prevalence to start with, e.g. PTH in hyperparathyroidism/hypercalcaemia (1 in 3000); GH in acromegaly (1 in 25,000); insulin in insulinoma/hypoglycaemia (1 in 250,000); ACTH in Cushing's syndrome/hypercortisolaemia (1 in 400,000), etc. Negative immunoassay results are on the other hand more likely to be significant in diseases with high prevalence.
In conclusion, because of the high analytical error rate in immunoassays relative to other routine analyses, Bayesian reasoning provides a knowledge approach 58–61 which allows laboratorians, the providers, and clinicians, the users of immunoassays to extract information about potentially false results even when they appear ‘not-unreasonable’. Essentially, false-positive immunoassay results are most likely to occur when immunoassays are used as screen in condition with low prevalence/incidence or conditions with less specific clinical correlates. On the other hand, false-negative immunoassays results are more likely in disorders with high prevalence/incidence, associated with strong clinical/radiological correlates. In both cases, further analytical validation would be justifiable or even necessary before invasive and expensive follow-up investigations or management/diagnosis is concluded. A validation strategy incorporating probabilistic Bayesian reasoning 58–61 coupled with judicious and ascetic requests of immunoassays by clinicians as well as follow-up affirmative tests by laboratorians can ameliorate this source of error. However, this approach must not be regarded as an optimal resolution because a long-term analytical solution to enhance immunoassays' accuracy is not only feasible and desirable but long overdue. 9,10 Only immunoassays' providers can, however, introduce these radical changes, being beyond laboratorians' and clinicians' control.
DECLARATIONS
