Abstract
This review focuses on neonatal transient hypothyroxinaemia, a condition characterized by temporary postnatal reductions in concentrations of Total T4 or Free T4, with normal or low concentrations of thyroid stimulating hormone (TSH). There is neither an agreed quantitative definition, nor an agreed mode of measurement for the condition. Transient hypothyroxinaemia is not routinely monitored yet it is thought to affect about 50% of preterm infants; it was thought to be without long-term sequelae but observational studies indicate that neurodevelopment may be compromised. The aetiology of transient hypothyroxinaemia is complex. There are significant contributions from the withdrawal of maternal–placental thyroxine transfer, hypothalamic–pituitary–thyroid immaturity, developmental constraints on the synthesis and peripheral metabolism of iodothyronines and iodine deficiency. It is not possible to distinguish clinically, or from laboratory measurements, whether transient hypothyroxinaemia is an independent condition or simply a consequence of non-thyroidal illness and/or drug usage. An answer to this question is important because studies of thyroid hormone replacement have been instigated, with mixed results. Until the aetiology of transient hypothyroxinaemia is better understood it would seem prudent not to routinely supplement preterm infants with thyroid hormones. Iodine deficiency, non-thyroidal illness and drug usage are the most modifiable risk factors for transient hypothyroxinaemia and are the clear choices for attempts at reducing its incidence. We suggest that transient hypothyroxinaemia in preterm infants is defined as a normal or low TSH concentration in conjunction with a concentration of Total T4, that is ≤10th percentile of cord Total T4 of the equivalent gestational age had the infant remained in utero.
Introduction
Thyroxine is essential for human brain development throughout fetal life and for the first two years postnatally. Disruption or attenuation of the supply of thyroid hormones, if permanent, can result in congenital hypothyroidism and if temporary can result in conditions such as transient hypothyroidism, transient hyperthyrotropinaemia or transient hypothyroxinaemia. The diagnosis of each of these conditions is based on concentrations of specific thyroid hormones and thyroid stimulating hormone (TSH), but the causes and consequences of all of these conditions are not yet fully understood. Congenital and transient hypothyroidism are confirmed, respectively, at TSH concentrations persistently or temporarily >20 mU/L (UK Newborn Screening recommendation
Ontogeny of thyroid hormones
When interpreting the clinical significance of concentrations of thyroid hormones, TSH and thyroxine binding globulin (TBG) it is necessary to have knowledge of their ontogeny as many authors have demonstrated that concentrations are gestationally age related. 2–11 Much of the earlier work was based on relatively limited gestational ages, included samples obtained from different procedures such as serum from cardiocentesis, 2,5,7 from cordocentesis 3,4 or from cord at delivery, 6,9–11 and used different assays. The largest single series of infants describing the ontogeny of iodothyronines, TSH and TBG comprised 617 term and preterm infants and 22 fetuses (15–20 weeks gestation). 11 That study found that concentrations of TT4, TBG and total triiodothyronine (TT3) increase linearly from 15 to 42 weeks gestation; concentrations of FT4 increase linearly until late second trimester, when they peak, before falling very slightly to term; concentrations of reverse triiodothyronine (rT3) increase linearly and peak at around 27 weeks and then fall slightly to term; concentrations of TT4 sulphate (T4S) are similar in pattern to rT3 but the fall towards term is more marked; and TSH concentrations are fairly constant throughout gestation (Figure 1). 11 (Sulphation helps to regulate the amount of active thyroid hormone; T4S is exclusively metabolized to the biologically inactive reverse T3S (rT3S). 12 ) The TT4/TBG ratio increases with gestational age until late second trimester and thereafter remains constant; the TSH/FT4 ratio decreases appreciably with gestation until the end of the second trimester, thereafter it very slightly decreases to term. 11
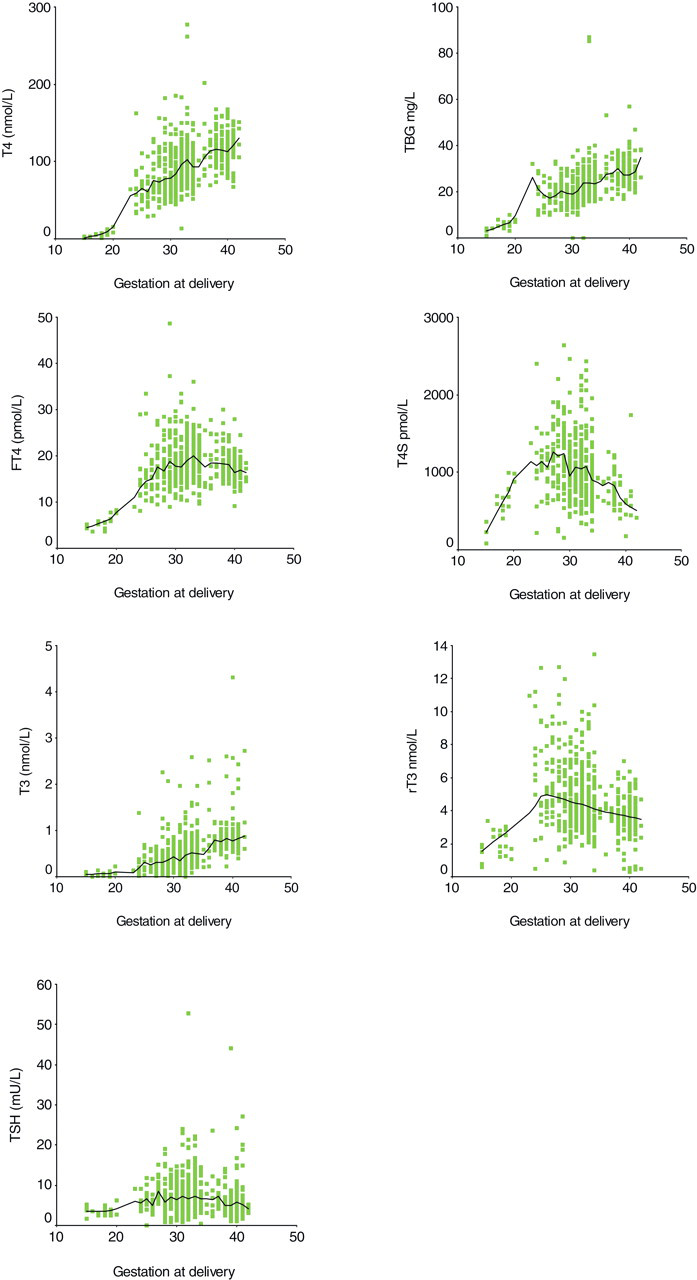
Levels of iodothyronines, TSH and thyroxine binding globulin (TBG) by gestational age (in weeks) (modified from: Hume R, Simpson J, Delahunty C, van Toor H, Wu SY, Williams FLR, VisserTJ Human fetal and cord serum thyroid hormones: developmental trends and inter-relationships. J Clin Endocrinol Metab 2004;
Measurement of thyroid hormones for classification of transient hypothyroxinaemia
Currently, routine neonatal thyroid hormone measurement in the UK is restricted to congenital hypothyroidism screening around postnatal days 5 and 6; the recommendation, at present, is that this is repeated in preterm infants at 36 corrected weeks gestation. Thereafter, thyroid hormones are measured in infants in response to clinical signs and symptoms or for research purposes. When the measurements are made is critical to interpretation, as there are very marked changes in the concentrations of these hormones both gestationally and postnatally. 13
The cooling of the infant at delivery, along with other birth stresses are natural stimulants of hypothalamic thyrotropin releasing hormone production. Such cooling evokes a surge of TSH at around 30 min of age, which in turn stimulates TT3 and TT4 secretion from the thyroid; concentrations of serum TT3 and TT4 peak at about 24–36 h postpartum. 14 Within the first 24 h and within restricted gestational age groups there are marked differences in concentrations of thyroid hormones, TSH and TBG between premature infants ≤27 weeks gestation and infants ≥28 weeks gestation. The mean TSH surge in infants ≤27 weeks gestation is markedly attenuated at 8 mU/L, compared with 20 mU/L in infants of 28–30 weeks and 23 mU/L in infants of 31–34 weeks. Mean TT4 concentrations in infants ≤27 weeks decline from cord values over the first 24 h (from 65 to 59 nmol/L) whereas concentrations in 28–30 weeks and 31–34 weeks increase (70–84 nmol/L and 98–125 nmol/L, respectively). FT4 concentrations increase slightly in infants ≤27 weeks, but more so in the other gestational groups. TBG concentrations in all groups are relatively consistent over the first 24 h although absolute concentrations are attenuated in infants ≤27 weeks (Figure 2). 15
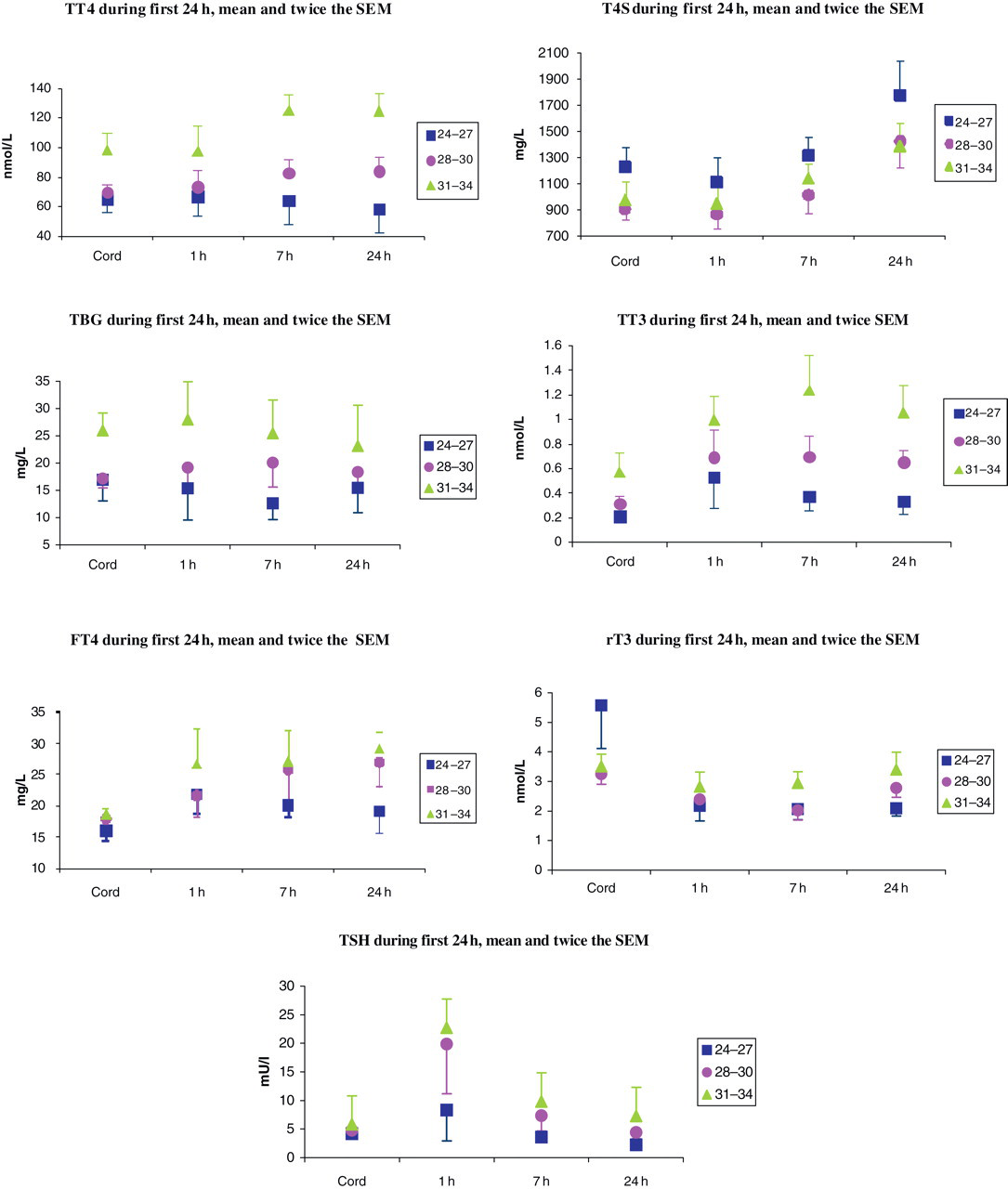
Means and ×2 standard error of means (SEM) of levels of iodothyronines, TSH and thyroxine binding globulin (TBG) (during the first 24 h of life by gestational age group (24–27, 28–30 31–34 weeks) (modified from Murphy N, Hume R, van Toor H, Matthews TG, Ogston SA, Wu SY, Visser TJ, Williams FLR The hypothalamic–pituitary–thyroid axis in preterm infants: changes in the first 24 h of postnatal life. J Clin Endocrinol Metab 2004;
Over the first month of life the concentrations of thyroid hormones, TSH and TBG continue to vary. The largest series of infants to be followed-up included 101 infants ≤27 weeks, 196 between 28 and 30 weeks and 253 between 31 and 34 weeks gestation. 16 In all preterm groups, the concentrations of TSH decrease appreciably within the first week and remain constant thereafter; TBG concentrations in term infants peak initially and thereafter fall to and remain at around cord concentrations; in 28–30 and 31–34 week infants the TBG concentrations remain at more or less cord concentrations throughout the first month, but in infants ≤27 weeks after an initial drop in concentrations TBG increases steadily thereafter. 16 Compared with term infants, who show a postnatal rise in concentrations of TT4, the TT4 concentration is attenuated in 31–34 week infants, absent in 28–30 week infants and reversed in infants ≤27 weeks. 16 The pattern of FT4 concentrations in term infants mirrors that of TT4, showing an increase at day 7 with a gradual decline to day 14 and 28; this is similar but attenuated in 31–34 and 28–30 week infants. The postnatal increase is absent in infants ≤27 weeks 16 but does not mirror the nadir evident in TT4 concentrations in this gestational range (Figure 3).
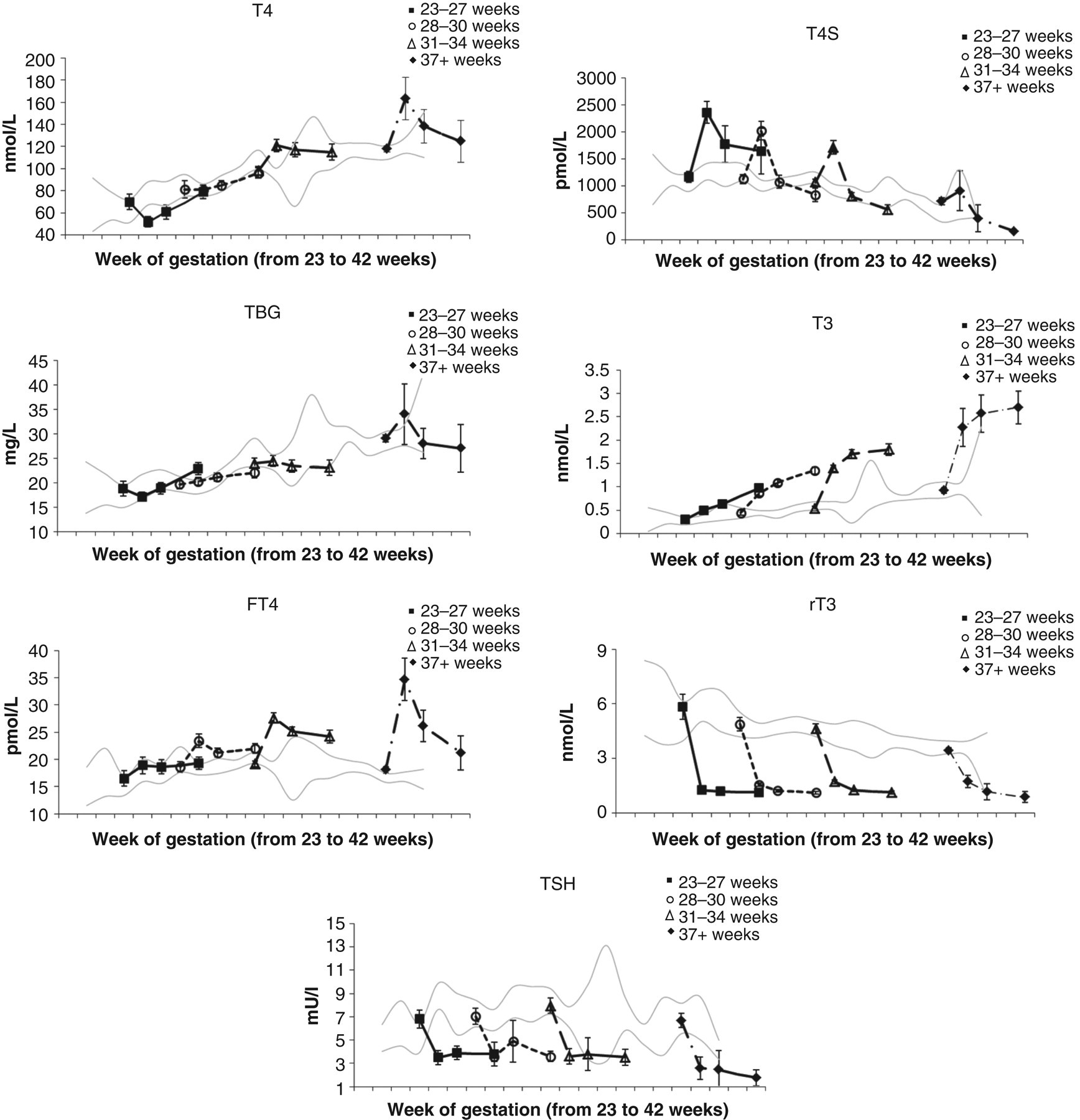
Means and ×2 standard error of means of levels of iodothyronines, TSH and thyroxine binding globulin (TBG) in cord and at day 7,14 and 28 postpartum by gestational age group (modified from Williams FLR, Simpson J, Delahunty C, Ogston SA, Bongers-Schokking JJ, Murphy N, van Toor H, WU SY, Visser TJ, Hume R Developmental trends in cord and postpartum serum thyroid hormones in preterm infants. J Clin Endocrinol Metab 2004;
Interpretation of postnatal concentrations in preterm infants requires knowledge of what is normal. Reference ranges for thyroid function tests in premature infants exist, but there are limitations. Some authors provide data for groups of gestational ages, 2,17,18 some restrict data to ‘healthy’ premature infants, 17,18 while others use unselected infants; 13 and some data are based on measurements taken at any point within the first week of life (although excluding the first 24 h) 17 (Table 1).
Reference ranges for neonatal concentrations of TT4, FT4 and TSH, according to method of assay
SD, standard deviation
*FT4 ng/dL to pmol/L multiply by 12.9
†TT4 μg/dL to pmol/L multiply by 12.9
So when should thyroid hormones, TSH and TBG be measured? Cord concentrations most likely reflect the true in utero hormone requirement for the developing fetal brain. Because of the marked gestational and postnatal changes in thyroid hormone, TBG and TSH concentrations, we recommend that in preterm infants their concentrations should be corrected to the equivalent gestational age had the infant remained in utero.
The concept of equivalence to gestational age
The brain develops along a pre-programmed path and it is not unreasonable to assume that preterm infants require the same concentrations of TT4, the preferred brain substrate, whether in utero or ex utero. Concentrations of iodothyronines, TSH and TBG in cord blood (or from cardio- or cordocentesis) are assumed reflective of the concentrations required by the fetus in utero. This assumes that prenatal and intrapartum factors do not have a significant impact on cord concentrations, which on current evidence 13 holds valid. The very different developmental expression of concentrations of iodothyronines, TBG and, to a lesser extent TSH means that interpretation of their concentrations from cord blood should be related to gestational age. Such knowledge is essential for the management of premature infants, as it provides the clinician and researcher with a reference range for expected normal concentrations. Furthermore, because of the influence of gestational age, the postnatal concentrations of preterm infants should be interpreted corrected to cord concentrations of the equivalent gestational age had the fetus remained in utero. This approach is the best available; it was used in part by Ares et al. 19 and also by Clark et al. 18 but the first large series of data published, which allowed application of the approach for preterm infants, was published by Williams et al. 13 The original dataset had comparatively few infants recruited between 35 and 37 weeks gestation and consequently the standard deviations associated with the mean values were large. For this review we include additional infants between 35 and 37 weeks gestation (Table 2).
Mean ± standard deviation (number) for cord serum concentrations of iodothyronines, TSH and TBG by gestation in unselected infants
TBG, thyroxine binding globulin
How should thyroid hormones be measured? The research perspective
Most thyroid tests are comparative, i.e. the patient sample is compared with a laboratory standard solution; the underlying assumption is that the two solutions are similar in all respects except for the compound being measured. Under many circumstances associated with prematurity this assumption does not hold true. Interpretation and comparison of concentrations is made even more difficult for research purposes when the thyroid hormone concentrations have been assayed using different methodologies. Researchers need to consider which of the assays are important to the definition of transient hypothyroxinaemia, and which of the assays are best suited for future research in this area.
TSH is the preferred screening marker for thyroid disease in adults but it is also needed for the classification of transient hypothyroxinaemia in preterm infants, and to distinguish this from transient and congenital hypothyroidism where TSH concentrations are elevated. TSH is commonly assayed using automated immunometric methods, and one study 20 found good concordance between four assays (Immulite TSH-3G, Elecsys TSH, Vitros ECi TSH and ADVIA Centaur TSH-3) in samples of patients undergoing bone marrow transplant. However, another overview of laboratory tests reported ‘less-than-ideal’ agreement between assays for TSH. 21 Several factors, some of which are frequent occurrences in the preterm infant, can alter TSH. Acute and severe illness, and certain drug usage also alter TSH concentrations and interpretation and comparison of concentrations should take these factors into account. In addition, TSH measurement is affected by interference from heterophile antibodies, which falsely increases TSH concentrations. 21 TSH autoantibodies can also cross the placenta and may lead to an artefactual diagnosis of hypothyroidism in the neonate. 22
TT4 assays have reasonable concordance between manufacturers 23 and reasonable test reproducibility although with poor clinical precision 24 and because over 99% of TT4 is bound to binding proteins, with 50–70% bound to TBG, 15–20% bound to albumin and the remainder to pre-albumin, any changes in binding proteins or binding affinity, can affect the reliability and accuracy of TT4 measurements. TBG concentrations can be affected by drugs and acute illness, with TT4 concentrations subsequently affected 21 factors which again have to be considered when interpreting concentrations in preterm infants.
For assay of FT4, with the caveat of measurement under conditions of low-binding proteins, direct equilibrium dialysis methods are considered the ‘gold standard’ 25 as other immunoassays can show bias according to protein-bound TT4, to serum TT4 binding capacity, 26,27 to inherent methodological variation 28 or to comparison to inappropriate reference ranges. 28 To determine the impact of serum TT4 binding capacity on the FT4 measurements, a French study 27 compared the FT4 measured by direct equilibrium dialysis with nine immunoassays. Serum binding capacity was determined by dividing the TT4 concentration (Elecsys) by the FT4 concentration determined by direct equilibrium dialysis and the patients divided into three groups: high, medium or low serum binding capacity. 27 Overall, although there was variation within groups, the biases between each of the immunoassays and direct equilibrium dialysis according to serum binding capacity were significant, although generally small for the normal and high serum binding capacity groups, but significant and appreciable with the low serum binding capacity group. Similarly, the concordance between FT4 measurements with direct equilibrium dialysis and each of the binding capacity groups; for normal and high serum binding capacity groups the range of concordance was between 59–96% and 66–90%, respectively, but for the low serum binding capacity group the range was 31–69%. Low serum binding capacity is frequently found in sick hospitalized patients, and because FT4 concentrations can be either increased or decreased simply due to the assay method, it is essential to consider the method used to correctly interpret the FT4 results. 27
FT4 concentrations can also be affected by drugs, 29,30 free fatty acids, 30,31 cleavage of TBG 32 and heparin use; 29 conditions which are particularly prevalent in premature infants. Free fatty acids can displace protein-bound TT4 which elevates FT4; 30,33 the parenteral nutrition of premature infants typically includes triglyceride emulsions at concentrations which are known to generate average ratios of free fatty acids to albumin of 12 or higher, which is four times higher than the ratio needed to inhibit protein binding in vitro. 33,34 The artefactual rise in FT4 is potentially compounded by the use of heparin which, even at low doses, can lead to in vitro activation of lipoprotein lipase with the subsequent generation of free fatty acids. 35 Albumin is a major component of total fetal serum protein concentrations, but TBG is the preferred carrier for TT4 even in the mid-trimester fetus. 5 During sepsis and inflammation TT4 may also be cleaved from TBG by polymorphonuclear leukocyte elastase, which again would artificially increase FT4 concentrations. 36 Finally, the assays should be performed at 37°C rather than at room temperature, for at room temperature the results in serum samples with abnormal TBG concentrations may show bias due to differences in TT4 dissociation between the standard and samples. 29,37
In infants ≤27 weeks gestation apparent hypothyroxinaemia (low TT4) with concomitantly unchanged or slightly raised FT4 concentrations is contrary to expectation, but has been described. 16,38,39 Whether this pattern is artefactual or not is unclear, but any one of the mechanisms described previously could be responsible.
In summary, direct equilibrium dialysis measurement of FT4 is the preferred analytical method as it is not biased by protein binding of TT4 nor by changes in serum proteins. The immunoassays produce variable results with a tendency to overestimate FT4 at high protein concentrations and underestimate FT4 at low protein concentrations. 37 Because of the vagaries of measuring FT4, it has been suggested that TT4 measurement is probably more accurate for critically ill patients (such as extremely premature infants) than FT4 measurements. 29 However, because of the complex interplay between acute illness and drug usage and thyroid hormone, TSH and TBG levels, which is compounded by method of assay, full interpretation of clinical condition can probably only be fully understood with review of TSH, TBG, TT4 and FT4 within the context of the current medical condition. Such an approach is only achievable currently in the UK within a research setting, as most National Health Service clinical biochemistry laboratories are unable to measure TBG and TT4 and most do not have access to direct equilibrium dialysis for analysis of FT4.
Which hormone(s) should be used in the classification of hypothyroxinaemia?
It has been suggested 40 that TT4 concentrations measured during the first two weeks of life in low birth weight infants (very low birth weight: <1500 g and 30 weeks gestation; extremely low birth weight: <1000 g and 27 weeks gestation) are inappropriate for identifying hypothyroxinaemia, as TBG concentrations in such infants are low at this time. However, in the largest series of infants followed to date concentrations of both TT4 and TBG increase in cord serum between 15 and 42 weeks gestation. 11 Cord log TT4:TBG ratios are low (<2:1) during the first trimester; 11 increase during the second trimester, 5,11 which reflects a proportionally greater rise in TT4 concentrations; and plateau at a ratio of around 4:1 in the third trimester (Figure 1). 11 During the first 28 days of postnatal life, the log TT4:TBG ratios decrease appreciably from cord ratios in infants ≤27 weeks, but increase in infants >30 weeks. 11 In these premature infants, the change in the logTT4:TBG ratios is caused by a greater change in concentration of TT4 relative to TBG; thus low concentrations of TBG per se do not seem to account for the hypothyroxinaemia.
Measurement of TSH is essential for classifying hypothyroxinaemia; and although the usual definition of transient hypothyroxinaemia typically refers to FT4 concentrations, in reality studies report TT4 concentrations. There are major problems with FT4 assays in the preterm population, 25–37 and classification of transient hypothyroxinaemia is best made from measurement of TT4 and TSH and TBG.
What blood media should be used for measuring concentrations of thyroid hormone for research?
Transient hypothyroxinaemia may be diagnosed from capillary, venous or arterial, plasma or serum samples or from whole blood on filter paper such as Guthrie cards. The gold standard for research is measurement in serum (usually venous), with the less accurate filter paper measurement used for population sampling and retrospective studies.
Concentrations of thyroid hormones, TSH and TBG differ, quite markedly, according to whether they are measured in serum, plasma or whole blood. Clinically, thyroid hormones, TSH and TBG are likely to be assayed from the same blood component within the same laboratory, so between-laboratory variation is not a confounding factor. However, for research the choice of blood component is dictated, for example, by the availability of a reference range/normal data for interpretation, the ultimate use of the research and the likely logistics of the research process. Reference ranges appropriate for preterm infants until they reach the equivalent of term are not so common but are available albeit limited (Tables 1 and 2). For research, the differences in concentrations reported by the different blood components assayed are problematical. For instance, there are statistical differences between mean serum concentrations of TT4, TSH and TBG in paired samples of venous and capillary blood in healthy term infants. 41 Concentrations in venous serum are lower than those in capillary serum; but the differences are small and uniform over the range of samples. No differences were found between concentrations of TT3, rT3 and FT4 but linear regression analysis showed that the relationship was skewed. 41 The venous serum estimates were lower than the capillary serum estimates when TT3 and rT3 were low, and when FT4 was high. By contrast, venous serum estimates were higher than capillary serum estimates when TT3 and rT3 were high, and when FT4 concentrations were low. The variety of blood components and assays used in research makes detailed comparison of concentrations between research groups almost impossible.
Although serum measurement of a range of thyroid hormones, TSH and TBG is the best option for research purposes, it may not always be achievable. If repeated measurements are needed on preterm infants the amount of blood required for the assay becomes an important consideration. The amount of blood required by experienced laboratories for analysis in serum of the range of thyroid hormones and TBG and TSH is approximately 1.3 mL. 11 Serum analysis requires blood obtained from in-dwelling catheter, venepuncture or capillary sampling. These procedures in preterm infants must be performed by trained staff and in the case of venepuncture, the patency of the veins is sub-optimal often resulting in repeated attempts or insufficient blood collection. By contrast, a Guthrie card sample of whole blood takes only 0.2 mL to fill and the blood can be collected from in situ catheters, venepuncture or capillary sampling from heel prick. The smaller quantity of blood required from a Guthrie card is significant considering the blood volume of preterm infants is only about 90–100 mL/kg 42,43 and that blood loss due to phlebotomy is a cause of anaemia in preterm infants. 43
Disadvantages of using Guthrie card samples are the current lack of suitable reference data to interpret preterm infants' postnatal concentrations of thyroid hormones, TSH and TBG which are corrected to equivalent gestational age, and the lack of information about the concordance of concentrations of TT4, TSH and TBG in serum and in whole blood.
Definition of transient hypothyroxinaemia
Transient hypothyroxinaemia is characterized by temporary postnatal reductions in whole blood concentrations of thyroxine, 1 or serum FT42 with normal or low concentrations of TSH. 1,2 There is no consensus about the concentration to use to define ‘low’, which thyroid hormone to use in the classification nor when the measurements should be made. Consequently, studies have measured either TT4 44–46 or TT3 47 concentrations; collected blood at variable postnatal ages; 44–49 and defined transient hypothyroxinaemia variously as standard deviations from mean TT4 concentrations, with 13 and without 44–46 adjustment for gestational age and day of testing or simply as cut-off values for blood TT4 49,50 (Table 3).
Definition and incidence of transient hypothyroxinaemia reported by various authors
TBG, thyroxine binding globulin
*Concentrations of thyroxine estimated from Guthrie cards which are part of neonatal screening programmes are expressed as a standard deviation about the mean on a daily basis and the denominator thus includes preterm and term infants
†Levels of thyroxine estimated from serum and expressed as a standard deviation, which is appropriate to equivalent gestational age at day of testing
The use of a quantitative criterion for defining transient hypothyroxinaemia is important as it enables infants to be objectively classified as hypothyroxinaemic, and identifies those who would be suitable to enter trials of thyroxine supplementation. Previous work has used −1, 13 −2, 13 −2.6, 44 −3.0 46 or −3.1 45 standard deviations for the definition of hypothyroxinaemia. Use of a variable standard deviation creates a variable proportion of infants identified as hypothyroxinaemic; for instance in the studies represented in Table 3 the proportion of infants classified as hypothyroxinaemic born ≤27 weeks gestation varied from 7–100%. Is this variation simply a reflection of the measurement method? The use of standard deviations to provide a cut-off for classification of hypothyroxinaemia has arisen through studies which use neonatal screening laboratories, based on Guthrie card estimates of TT4 concentrations, as such laboratories typically express the TT4 as a standard deviation about the mean TT4 concentration on a daily basis. Crucially these standard deviations include term and preterm populations and as infants born over 37 weeks gestation constitute 92% of all (UK) births, the mean and standard deviations are biased towards such births. TT4 concentrations are gestationally age related, with term infants having approximately double the concentrations of TT4 in cord serum than infants born <28 weeks gestation. 16 Extreme preterm infants are therefore more likely to be classified as hypothyroxinaemic when they are actually euthyroid for corrected gestational age. Previously we defined transient hypothyroxinaemia as a gestationally age adjusted TT4 concentration, taken on postnatal day 7, which fell below mean −1 standard deviation of cord TT4 concentration corrected to an equivalent gestational age had the fetuses remained in utero. 13 The logic of this is sound but small numbers in some gestational age groups led to fairly large standard deviations; until numbers in each week can be increased, we propose as an alternative the gestationally age adjusted 10th percentile of TT4 concentrations as an appropriate cut-off value for distinguishing hypothyroxinaemic and euthyroid infants.
A definition of transient hypothyroxinaemia needs to encompass an appropriate time frame. Most definitions have used TT4 concentrations taken within the first three weeks of life (e.g. Table 3). Definitions based on cord concentrations are inappropriate to describe this postnatal condition/illness, and measurements over the first two or three postnatal days are probably inappropriate as thyroid hormone, TSH and TBG concentrations change appreciably over this period in response to extrauterine adaptation. 3,7,15,51 Likewise, until the aetiology of transient hypothyroxinaemia is clarified, a definition based on only one time point is liable to underestimate its true incidence. The duration and incidence of hypothyroxinaemia has changed over the past 20 y. In an Australian study in 1988, 100% of 23–25 week infants and 40% of the 26–27 week infants were reported as hypothyroxinaemic at 6 weeks; 50 comparable Scottish figures in 2004 were 47% and 22% at four weeks. 39 This temporal change in the concentrations of TT4 in extreme preterm infants over the past 20 y both in the severity and duration of hypothyroxinaemia may be due to assay differences but also to reduction in the severity of non-thyroidal illness with the advent of prenatal corticosteroid use and postnatal surfactant administration.
We suggest that transient neonatal hypothyroxinaemia in preterm infants is defined as a normal or low TSH concentration in conjunction with a concentration of TT4 that is ≤10th percentile of cord TT4 of the equivalent gestational age had the infant remained in utero. Using this definition (with postnatal TT4 concentrations taken on days 7, 14 and 28) we found 20% of infants born ≤34 weeks gestation were hypothyroxinaemic. 52
Aetiology of transient hypothyroxinaemia
The diagnosis of transient hypothyroxinaemia is not usually made out of the research context unless thyroid function tests have been requested for some reason. However, it is important for clinical laboratories to be aware of this condition as in studies of preterm infants which have measured thyroid hormones and TSH postnatally, the incidence of hypothyroxinaemia varies from 7–100%; although a recent paper quoted concentrations of around 50% in infants 24–28 weeks gestation. 53 A key question still to be answered is exactly what is transient hypothyroxinaemia; can it be distinguished clinically and in the laboratory from non-thyroidal illness?
Many illnesses, conditions and treatments which are prevalent in the neonatal period of preterm infants affect concentrations of thyroid hormones, TSH and TBG. For example, critical illnesses such as respiratory distress reduce TT4 51,54,55 and TT3. 56 Infection in blood or positive endotracheal cultures reduces serum concentrations of TT4, 57,58 TT3, 57,58 TBG 58 but increases concentrations of T4S. 58 Necrotizing enterocolitis reduces serum concentrations of TT4 but increases serum concentrations of rT3. 58 Persistent ductus arteriosus is associated with reduced concentrations of serum TT4 and TT3. 58 Many frequently used medications also affect concentrations of thyroid hormones in a complex manner. For example, aminophylline, which is used as a respiratory stimulant in preterm infants with recurrent apnoea, increases TSH, 59 TT4 58,59 and TT3 concentrations 58 whereas theophylline, a metabolite of aminophylline, increases plasma TT4 concentrations in adults and children, with inconsistent effects on TT3 and rT3; 60 although prolonged administration of theophylline to infants has no effect on thyroid hormone concentrations. 61 Glucocorticoids, which are used to mitigate chronic lung disease in preterm infants, have various effects on the pituitary–thyroid axis. TSH secretion in adults is reduced, 62 peripheral deiodination of TT4 to TT3 is decreased 63 in hypothyroid adults and rT3 and TBG concentrations are reduced in healthy adult volunteers. 64 Postnatal dexamethasone use has also been associated with large reductions in FT4 concentrations on day 14. 58 Dopamine, which is used to maintain blood pressure, inhibits secretion of TSH 65,66 and reduces TT4, TT3 58,67 and TBG concentrations in premature infants. 58 Diamorphine and morphine, which are used for sedation and pain relief in preterm infants, suppress secretion of TSH in rats 68 and reduces serum concentrations of TT4, TT3, FT4 and TBG in preterm infants. 58
The aetiology of transient hypothyroxinaemia is complex and non-thyroidal illness and postnatal drug use clearly play an important part. 39,57,69 But there are also significant contributions to the aetiology of transient hypothyroxinaemia from the withdrawal of maternal–placental thyroxine transfer, 70–72 hypothalamic–pituitary–thyroid immaturity, 15,73 developmental constraints on the synthesis 11,16,74 and peripheral metabolism of iodothyronines 75,76 and iodine deficiency. 19,77
Clinical consequences of transient hypothyroxinaemia
Currently, it is not possible to distinguish clinically or from laboratory measurements, whether transient hypothyroxinaemia is an independent condition or whether it is simply a consequence of non-thyroidal illness and/or drug usage. An answer to this question is important because studies of thyroid hormone replacement 78–82 in extreme preterm infants have been instigated, with mixed results. Only one of these studies included long-term neurodevelopmental outcome 78 and the results were equivocal. Benefit was evident only in subgroup analysis (underpowered) which showed that infants <27 weeks scored more highly on the Bayley Mental Development Index at two years of age if supplemented with thyroxine when compared with unsupplemented infants. By contrast, supplemented infants ≥27 weeks significantly scored 10 points lower than unsupplemented infants. Subsequent follow-up (but with different assessments and with slightly different gestational age groupings) at 5.7 79 and 10 y 80 reported similar findings. Until the aetiology of transient hypothyroxinaemia is better understood and in particular the contribution is elucidated of non-thyroidal illness and drug usage to its incidence, it would seem prudent not to routinely supplement preterm infants with thyroid hormones.
Iodine deficiency, non-thyroidal illness and drug usage are the most modifiable risk factors for transient hypothyroxinaemia and are the clear choices for attempts at reducing its incidence. Iodine is essential for the synthesis of TT4 and even mild deficiency of iodine is associated with neurodevelopmental deficits in infants and children.
83,84
Iodine balance studies on enterally fed, healthy preterm infants at one month of age concluded that an enteral intake of at least 30–40 μg/kg/d was required.
19,85
Iodine contents of a number of term and preterm formulae mostly reflect this requirement
86,87
but the most recent guidelines
88
for parenteral nutrition recommend only 1 μg iodine/kg/d. Almost all extreme preterm infants receive parenteral nutrition at some point, and one study found that it provides 95% of caloric intake of the sickest infants, 23–27 weeks gestation, on day 7 and 51% at day 28.
58
There are no current recommendations indicating the iodine requirements of extreme preterm infants, sick infants nor infants in the early neonatal period but it is very likely that their needs are no less than those of healthy preterm infants.
89
Preterm, sick infants are vulnerable to iodine deficiency and thus at an increased risk of transient hypothyroxinaemia. The (UK) Medical Research Council has recently funded a randomized trial of iodine supplementation of infants born <31 weeks gestation, with neurodevelopmental assessment at two years of age (
Transient hypothyroxinaemia is the commonest thyroid dysfunction in preterm infants. It was thought to be without clinical significance, 1,7,90 but some observational studies have reported adverse associations between transient hypothyroxinaemia and neurodevelopment, 44–46 and low postnatal concentrations of TT3 and neurodevelopment. 47 The magnitude of association between hypothyroxinaemia and neurodevelopmental outcome differs between the studies, which might be due to the choice of hormones measured (TT4 or TT3), the method of sample collection (whole blood on Guthrie card or plasma), the variable postnatal age of iodothyronine measurement and of neurodevelopmental assessment and the variable definition of transient hypothyroxinaemia. 44–47 All of the studies adjusted for a range of likely confounders of neurodevelopment but only one 44 included a measure of maternal intellect; and whether the mother had attended school for less or more than 12 years. Cause and effect cannot be elucidated from observational studies and results are notoriously difficult to interpret. While the current studies have included an impressive range of potential confounding factors some important factors are nevertheless missing, such as: a more objective measurement of contemporary parental intellect, birth order, whether the child was breast or formula fed, multiple birth and use of postnatal corticosteroids.
Comparability between the studies is impossible as they use different methodologies, different assessment scales and different analytical procedures. For the future, it would be beneficial for researchers and clinicians to decide upon an array of standard assessment tools and analyses to be used. All that can be concluded overall is that neurodevelopmental deficit associated with hypothyroxinaemia at 2 y corrected age using a global test of cognitive function is between 17.5 (unadjusted) and 6.8 points (adjusted); 44 the mean and standard deviation for such tests is 100 ± 16 points. At two years corrected age, hypothyroxinaemic infants are 3.5 times more likely to have developmental delay (adjusted odds ratio 3.5); 45 at five years hypothyroxinaemic infants were more likely to have neurological dysfunction (adjusted odds ratio 1.3) and mental retardation (adjusted odds ratio 1.1). 46 By nine years of age, hypothyroxinaemic infants were more likely to be classified as school failures (adjusted odds ratio 1.3). 46 The study using TT3 as the definition of thyroid dysfunction, found lower TT3 concentrations associated with a deficit of 8.3 points (adjusted) at 18 months using a global test of cognitive function. 47 By 7.5–8 y, the deficit was 6.6 points overall (adjusted). 91
Clinical treatment of hypothyroxinaemia
Unless clinicians have reason to suspect thyroid dysfunction, transient hypothyroxinaemia is likely to go unnoticed in the majority of infants. However, if it is recognized the question is raised of whether it should be treated. La Gamma et al. 53 recently reported the results of a phase 1 trial (to establish the appropriate dose) of four thyroid hormone regimens for the treatment of transient hypothyroxinaemia. The trial was restricted to infants <28 weeks gestation. The trial had six arms: placebo (5% dextrose), potassium iodide (30 μg/kg/d) and continuous or bolus infusions of either 4 or 8 μg/kg/d of TT4 for 42 d. TT4 was supplemented with TT3 (1 μg/kg/d for the first 14 d). TSH was suppressed in all active arms of the trial, with least suppression and some elevation of TT4 achieved with the continuous supplement of TT4 at 4 μg/kg/d. A full trial with neurodevelopment as the outcome is the essential next stage. In view of the risk of suppression of the hypothalamic–pituitary–thyroid axis, it has been suggested that only preterm infants who are hypothyroxinaemic should be entrants to further clinical trials. 92 The quantitative classification of hypothyroxinaemia is essential to avoid entering infants who are in fact biochemically euthyroid. In the absence of definitive trials of efficacy and safety of thyroid hormone supplementation in transient hypothyroxinaemia in preterm infants, it would seem prudent – on finding biochemical evidence of hypothyroxinaemia – to watch and wait. A caveat to this conservative recommendation would be if the infant with hypothyroxinaemia had any features suggestive of congenital hypopituitarism, e.g. optic atrophy, mid-line defects, hypoglycaemia, electrolyte and water disturbances, etc. and where the thyroid dysfunction was thought part of a wider pan-hypopituitarism; 93 such an infant requires further investigation and appropriate hormonal treatment of this very much rarer condition.
Conclusions
Neonatal transient hypothyroxinaemia has a complex aetiology 11,15,16,19,70–77 and it is still uncertain whether or not it is a condition in its own right, or an epiphenomenon of pre-existing neonatal illness, prematurity and certain drug usage. 39,57,69 The condition is essentially a research entity because, routinely in the UK, it is neither monitored biochemically nor directly treated. There is growing observational evidence that the condition is associated with adverse developmental outcome, 44–47,52,78–82 and trials of thyroid hormone replacement have been started but with mixed results. 78–82 A coordinated research effort is needed to determine definitively the nature and consequences of the condition. Currently, the definition and measurement of transient hypothyroxinaemia varies between researchers. For the future, it would be helpful for researchers and clinicians to decide upon an array of standard assessment tools and analyses to be used. As a start in this process, we suggest that transient neonatal hypothyroxinaemia in preterm infants is defined as a normal or low TSH concentration in conjunction with a concentration of TT4 that is ≤10th percentile of cord TT4 of the equivalent gestational age had the infant remained in utero.
DECLARATIONS
