Abstract
Background
Serum concentrations of fetuin A/α2HS-glycoprotein (AHSG) have been linked to human metabolic alterations and can serve as an indicator of liver cell function. We assayed serum levels of AHSG in patients with non-alcoholic fatty liver disease (NAFLD), a hepatic manifestation of the metabolic syndrome, and examined their association with clinical, biochemical and histological phenotypes.
Methods
Serum AHSG levels were determined by enzyme linked immunosorbent assay in 99 patients with biopsy-proven NAFLD and 75 age- and gender-matched controls.
Results
Serum AHSG levels were significantly higher in patients with NAFLD (940 ± 120 μg/mL) compared with healthy controls (800 ± 130 μg/mL, Student's t test, P < 0.001). Bivariate analyses (Spearman's rank correlation) in patients with NAFLD showed a statistically significant association between AHSG levels and insulin resistance as assessed by the HOMA (homeostasis model assessment) index (r = 0.31, P < 0.01) and the liver fibrosis score index (r = 0.36, P < 0.001). The association between AHSG and fibrosis remained statistically significant even after adjustment for potential confounders, including the HOMA index ([beta] = 1.65, t = 2.38, P < 0.05).
Conclusion
Serum AHSG levels are significantly increased in adult patients with biopsy-proven NAFLD and are associated with insulin resistance. Importantly, our pilot data indicate that serum AHSG levels may identify NAFLD patients with higher fibrosis scores.
Introduction
The development of non-alcoholic fatty liver disease (NAFLD), affecting 20–34% of Western population, is strongly associated with the presence of the metabolic syndrome. 1 The term NAFLD is used to describe a wide spectrum of fatty liver changes ranging from simple steatosis to non-alcoholic steatohepatitis (NASH). 2 Current understanding of pathogenesis of NAFLD involves a two-hit hypothesis wherein initial hepatic insulin resistance causes steatosis and the second pathogenic stimulus causes oxidative stress and inflammation, ultimately resulting in the development of NASH. 3–5 Human fetuin A/α2HS-glycoprotein (AHSG) is a 59-kDa serum glycoprotein produced exclusively by hepatocytes with a concentration around 450–600 μg/mL in healthy subjects. 6,7 AHSG is a natural antagonist of growth factors like insulin and transforming growth factor β and levels of this molecule decrease during acute inflammation. 8,9 Growing evidence has suggested a potential role for AHSG in human metabolic alterations and in insulin resistance. 6,7,10,11 AHSG knockout mice have enhanced glucose clearance and insulin sensitivity, resistance to weight gain and lower serum free fatty acid and triglyceride levels. 12 Serum levels of AHSG have been linked to the features of the metabolic syndrome in humans. 7 Increased AHSG levels have been reported in children with NAFLD and related to insulin resistance and to features of the metabolic syndrome in both cross-sectional and longitudinal analyses. 13 In a study of 106 healthy Caucasians without type 2 diabetes, serum AHSG levels were positively associated with liver fat. 6 Of note, a decrease in liver fat content due to weight loss was accompanied by a decrease in AHSG plasma concentrations. 6 Recently, AHSG has been also suggested to play a role in the mechanisms linking obesity, chronic kidney disease and fatty liver disease. 14 Although metabolic alterations and insulin resistance are main contributors to the prevalence of NAFLD, 1–3 there is a paucity of data on the association between serum AHSG levels and biopsy-proven NAFLD in adult patients. Consequently, the aim of the present study was to determine serum levels of AHSG in patients with NAFLD and to examine the association between levels of this molecule and clinical, biochemical and histological phenotypes in this patient group.
Subjects and methods
Study participants
In this observational case-control study, a total of 99 patients with NAFLD (46 men and 53 women, mean age, 47 ± 9 years) and 75 healthy comparison subjects (38 men and 37 women, mean age, 47 ± 8 years) were enrolled. Patients with NAFLD were consecutively seen at our hospital-based specialized outpatient clinics over the past 12 months. All showed ultrasonographic evidence of steatosis grade 1 or higher. Patients with viral hepatitis, haemochromatosis, Wilson's disease, autoimmune hepatitis, primary biliary cirrhosis, sclerosing cholangitis, biliary obstruction, alpha-1 antitrypsin deficiency, ischaemic cardiac or cerebrovascular disease, impaired renal function or malignancies were carefully excluded from the present study. Subjects using oestrogens, amiodarone, steroids, tamoxifen and lipid-lowering agents were not eligible for this study. Patients with daily alcohol intake exceeding 20 g/d or previous abdominal surgery were also excluded. Seventy-five healthy age- and gender-matched volunteers were recruited as controls. All subjects included in the control group were judged to be in good health, with normal results on liver function tests and confirmed as having normal liver by ultrasound. Subjects with a consumption of alcohol >20 g/d or who were taking any medication were not included in the control group. The control group did not include relatives of NAFLD patients. All patients and controls were of Turkish descent. The study protocol was approved by the Ethics Committee of the Marmara University School of Medicine and all subjects gave their written informed consent to participate in the study.
Clinical and biochemical characterization
All subjects underwent physical examination, anthropometric measurements and biochemical screening. Body mass index was calculated from measurements of height and weight. Diabetes mellitus was diagnosed according to American Diabetes Association (ADA) criteria. 15 The metabolic syndrome was diagnosed using the Adult Treatment Panel III (ATP III) criteria. 16 The estimate of insulin resistance was calculated using the HOMA-IR (homeostasis model assessment) index, with the following formula: insulin resistance = fasting plasma insulin (in microunits/mL) × fasting plasma glucose (FPG) (in mmol/L)/22.5. Blood pressure was measured using a mercury sphygmomanometer in a quiet room after ≥10-min rest. Korotkoff 1 and 5 were taken for systolic blood pressure and diastolic blood pressure, respectively. Routine blood chemistry analyses were performed at the central laboratory of clinical chemistry of the university hospital. Serum high-sensitivity C-reactive protein (hs-CRP) levels were measured in duplicate, random order and in a blinded fashion using a commercially available method (Dade Behring, Marburg, Germany). The intra-assay and the inter-assay coefficients of variation for hs-CRP were 4.6% and 6.1%, respectively, and the lower detection limit was 0.19 mg/dL.
Liver histology
Ultrasonography-guided liver biopsies were performed under conscious sedation using a 16-gauge Hepafix needle. All biopsy specimens were placed in formalin solution for fixation and embedded in paraffin blocks. Serial sections (sectioned at 4 mm intervals) were stained with haematoxylin–eosin, Masson's trichrome. An experienced pathologist blinded to clinical data scored the liver biopsies according to the National Insurance of Diabetes and Digestion and Kidney Diseases (NIDDK) NASH Clinical Research Network scoring system. 17 Steatosis was scored from 0 to 3 with a four-grade scoring system from S0 to S3: S0: no steatosis or less than 5%, S1: 5–33%, S2: 33–66%, S3: >66%. Lobular inflammation was graded as follows: stage 0, no foci; stage 1: <2 foci.per.200× field; stage 2: 2–4 foci.per 200× field; stage 3: >4 foci.per 200× field. Ballooning degeneration of liver cells was evaluated as: grade 0, absent; grade 1, few cells; grade 2, many cells. The histological NASH score was defined as the unweighted sum of the scores for steatosis (0–3), lobular inflammation (0–3) and ballooning (0–2); thus ranging from 0 to 8. Cases with scores of 0–2 were considered as having simple steatosis; on the other hand, cases with scores of 5 or greater were diagnosed as definitive NASH. Cases with activity scores of 3 and 4 were considered as borderline NASH. 17 Fibrosis was staged as follows: stage 0: no fibrosis; stage 1: perisinusoidal or periportal fibrosis with 3 different patterns: 1A: mild, zone 3, perisinusoidal; 1B: moderate, zone 3, perisinusoidal fibrosis and 1C portal/periportal fibrosis; stage 2: perisinusoidal and portal/periportal fibrosis; stage 3: bridging fibrosis; stage 4: cirrhosis.
Serum AHSG assay
All blood samples were collected from an antecubital vein between 8:00 and 9.00 h after an overnight fasting. Samples were centrifuged at 2500
Data analysis
The study power was calculated using the StatMate software version 2.0 (GraphPad, San Diego, CA, USA) for Windows. Our experiment had a 95% power to detect a difference in mean fetuin levels of 69 μg/mL between patients and controls with a significance level (α) of 0.05 (two-tailed).The Kolmogorov–Smirnov test was performed in all continuous variables to define the presence of normality. Gaussian variables are expressed as mean ± standard deviation (SD), skewed data are reported as medians and interquartile ranges, and categorical variables are expressed as counts. The Student's t-test was used to evaluate differences between the two study groups in normally distributed continuous variables. When normality was not confirmed, the Mann-Whitney U test was used. Skewed data were log-transformed for further analysis. Correlations among the study variables were tested by the Spearman's correlation coefficient. Predictors of fibrosis scores in NAFLD patients were assessed using multiple stepwise linear regression analysis. All variables listed in Table 1 were entered into the model as potential predictors (independent variables). All calculations were performed using SPSS version 17.0 for Windows (SPSS Inc, Chicago, IL, USA). A value of P < 0.05 (two-sided) was considered statistically significant.
General characteristics of the study participants
HOMA-IR, homeostasis model of insulin resistance; AST, aspartate aminotransferase; ALT, alanine aminotransferase; HDL, high-density lipoprotein; LDL, low-density lipoprotein; hs-CRP, high sensitivity C-reactive protein; NASH, non-alcoholic steatohepatitis
Data are presented as means and SD, counts or medians and interquartile ranges, as appropriate
Results
Table 1 depicts the general characteristics of the study participants. The two study groups did not differ in terms of age, gender, systolic and diastolic blood pressure, smoking history and HDL cholesterol. Between-group comparisons identified a number of variables to be different in patients with histology-proven NAFLD compared with controls without NAFLD, including body mass index, HOMA-IR, aspartate aminotransferase, alanine aminotransferase, total cholesterol, low-density lipoprotein cholesterol and triglycerides. The prevalence of diabetes and the metabolic syndrome was higher in patients with NAFLD than in controls. Serum AHSG levels were significantly higher in patients with NAFLD (940 ± 120 μg/mL) compared with healthy controls (800 ± 130 μg/mL, Student's t-test, P < 0.001, Figure 1).
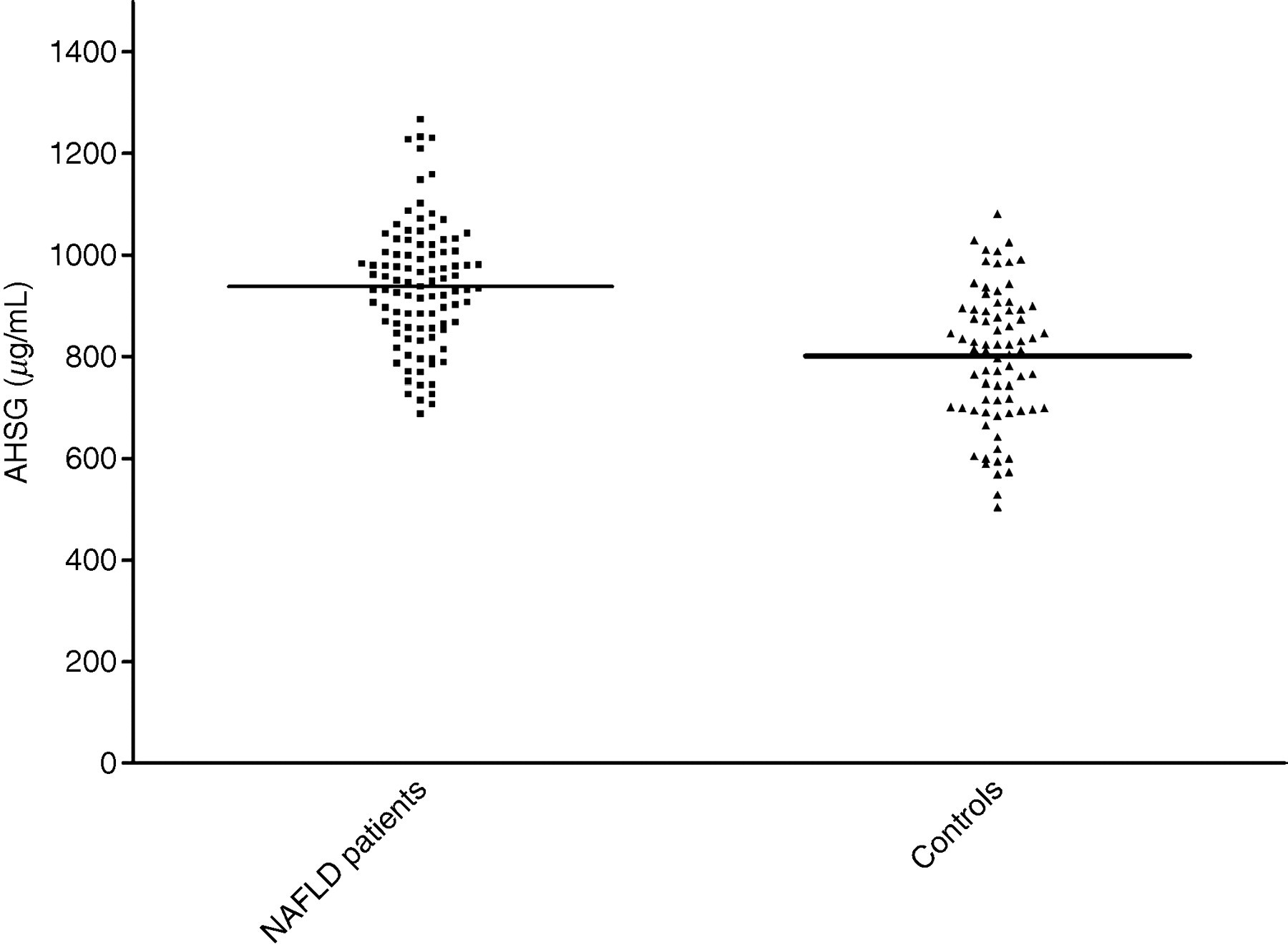
Scatter diagram for serum A/α2HS-glycoprotein (AHSG) levels in non-alcoholic fatty liver disease (NAFLD) patients and healthy controls. Horizontal lines across the scatter diagram represent mean values
Bivariate analyses in patients with NAFLD showed a statistically significant association between AHSG levels and insulin resistance as assessed by the HOMA index (r = 0.31, P < 0.01, Figure 2) and the liver fibrosis score index (r = 0.36, P < 0.001). There was a stepwise increase in the mean serum AHSG concentrations with each liver fibrosis score (P for trend < 0.001, Figure 3) in patients with NAFLD. However, we found no association between the AHSG and NASH scores (data not shown) and this molecule did not discriminate between simple steatosis and definite NASH.
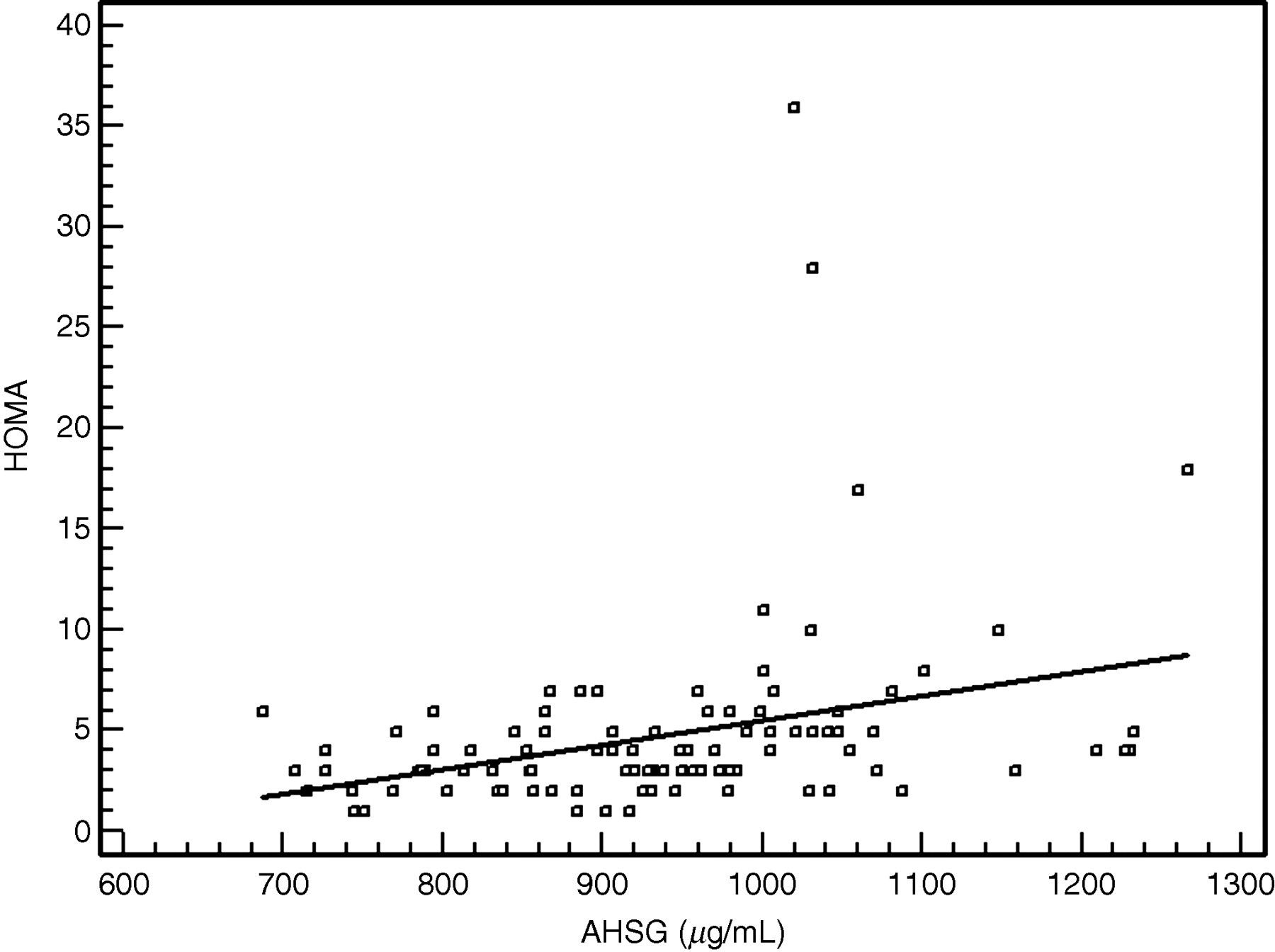
Scattergram and regression line showing a significant positive relationship between serum A/α2HS-glycoprotein (AHSG) and homeostasis model of insulin resistance (HOMA) in non-alcoholic fatty liver disease (NAFLD) patients
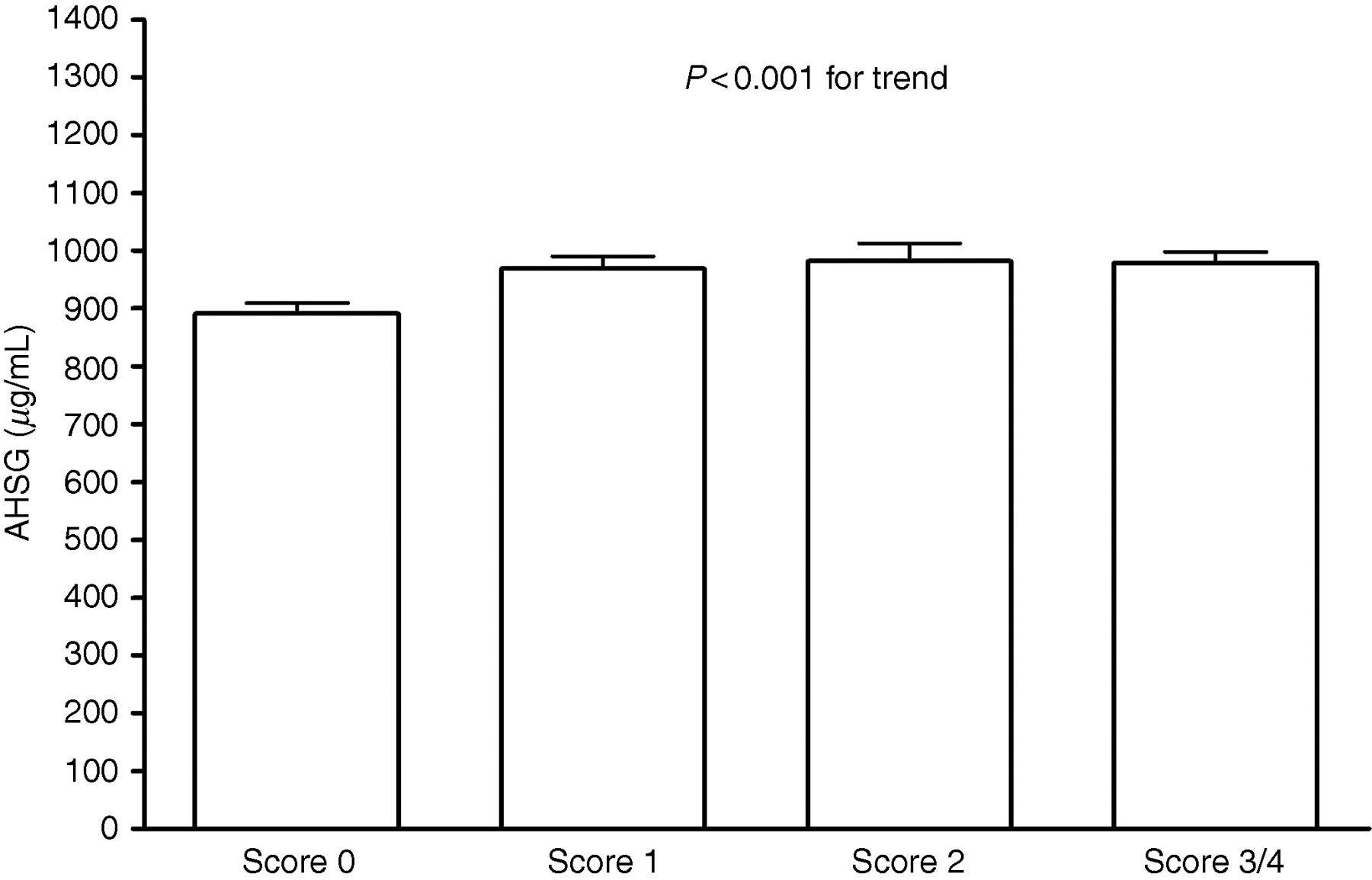
Stepwise increase in serum A/α2HS-glycoprotein (AHSG) levels with increasing severity of hepatic fibrosis
To determine the extent of confounding by other factors on the association between AHSG concentrations and fibrosis among patients with NAFLD, we performed multiple linear regression analysis with a model adjusting for age, sex, HOMA and all parameters listed in Table 1. When these potential confounders were entered into the multivariate model, we found that the association between AHSG and fibrosis ([beta] = 1.65, t = 2.38, P < 0.05) was not affected by other potential confounding factors.
Discussion
The main finding of this study is that serum AHSG levels are significantly higher in patients with biopsy-proven NAFLD and may serve as a biochemical marker of fibrosis in this patients group. It is noteworthy that the association between serum AHSG levels and fibrosis was independent of potential confounders, including HOMA. Altogether, this research suggests that high serum AHSG levels in patients with NAFLD do not merely reflect the effects of insulin resistance, but also a more extensive distortion of liver architecture. If independently validated, measurements of this biomarker might potentially help identify those patients with the highest risk of liver fibrosis for liver biopsy.
In vitro data suggest that AHSG can inhibit insulin receptor autophosphorylation and subsequent downstream signalling. 18 In this regard, there is compelling evidence from human studies that AHSG is involved in insulin resistance and the metabolic syndrome. 6,7 Of note, some 6 but not all 19 previous studies have suggested a significant relationship between liver fat content and AHSG. Reinehr and Roth 13 have reported that AHSG levels were higher in children with NAFLD and were related to insulin resistance. Our results confirm and expand in adult patients the relationship between high AHSG concentrations and NAFLD. The positive relation between AHSG concentrations and HOMA as observed in our study strongly supports the original hypothesis by Stefan et al. 6 that AHSG may act as a link between fatty liver and insulin resistance. However, previous studies did not consider whether the level of AHSG in patients with NAFLD can be associated with clinical, biochemical and histological phenotypes other than insulin resistance.
One important result in this study is that serum AHSG levels were positively and independently associated with fibrosis after allowance for potential confounders including HOMA. These results suggest the existence of mechanisms by which increased AHSG can be directly associated with liver injury independently from insulin resistance. Verma-Gandhu et al. 20 have previously reported that AHSG is a natural antagonist of TGF-beta – the key mediator of hepatic fibrosis – and that administration of this molecule may be beneficial in liver fibrosis through the inhibition of hepatic stellate cell function and collagen synthesis. These data therefore seem to suggest that levels of AHSG should be lower, not higher, in patients with more severe fibrosis. Although the pathophysiological mechanisms underlying the elevation of AHSG in hepatic fibrosis observed in our patients cannot be directly inferred from our present study, we interpret our results of a raised level of this molecule in advanced fibrosis as a counter-regulatory mechanism for slowing or halting the progression of fibrosis.
Several caveats of this study merit consideration. First, the case-sectional design of the study prevents us from inferring the direction of the association between serum AHSG levels and the presence of NAFLD and fibrosis. Second, the relatively small sample size limits the generalizability of our conclusions. Third, our sample included subjects of Turkish nationality, so that results cannot be extrapolated to populations with different ethnic background. This shortcoming is common to several single-centre studies where the population is genetically and ethnically homogeneous. On the other hand, a strength of our study is that all patients with NAFLD underwent liver biopsy, the best standard to diagnose this condition. 21–23 A liver biopsy has the advantage of providing important information relating to the degree of liver damage, as well as severity of inflammatory activity and fibrosis. However, it is an invasive test, has inherent complications, is expensive and operator-dependent. 21–23 As a result, serum biomarkers have raised great expectations as a reliable non-invasive means of assessing the presence and severity of NAFLD. 23
In conclusion, the results of our study indicate that serum AHSG levels are significantly increased in adult patients with biopsy-proven NAFLD and are associated with insulin resistance. Importantly, our pilot data suggest that serum AHSG levels may identify NAFLD patients with higher fibrosis scores. The results of current study add to the current knowledge suggesting a potential role for AHSG as a biomarker in a number of different human disease conditions, including atherosclerosis, 24 diabetes, 11 the metabolic syndrome, 7 osteoporosis, 25 and dementia. 26 Future studies are warranted to assess the potential clinical utility of AHSG as a biochemical marker to identify the different conditions within the spectrum of NAFLD.
DECLARATIONS
