Abstract
Current guidance recommends titrating the dose of metyrapone against serum cortisol concentration, in patients under medical management of Cushing's syndrome. In the UK, this almost always involves measuring serum cortisol concentration by immunoassay, the performance of which is questionable in the presence of altered steroid metabolism. Sera from two patients receiving metyrapone were analysed using a liquid chromatography tandem mass spectrometry (MS) steroid assay to identify which steroids, if any, were elevated in these patients. In addition, control serum was spiked with a series of steroids to identify any potential positive interferences in a cortisol immunoassay. Serum 11-deoxycortisol concentration was elevated in both of the patients studied. One patient also had an elevated serum 17-hydroxyprogesterone concentration and the other an elevated androstenedione. In addition, the results of the interference studies indicated that the cortisol immunoassay was susceptible to interference from 11-deoxycortisol, 17-hydroxyprogesterone and 21-deoxycortisol. However, the magnitude of interference, in the serum cortisol immunoassay, due to these three steroids could not account for the discrepancy between the cortisol concentrations measured by immunoassay and those measured by MS. Both clinicians and laboratory staff should be aware of these interferences when monitoring patients undergoing treatment with metyrapone, and consequently serum should be measured in these patients by MS, not by immunoassay.
Introduction
Metyrapone is a drug that is used in the medical management of Cushing's syndrome. It acts by inhibiting adrenal beta-hydroxylation of 11-deoxycortisol (11DOC), the final step in cortisol synthesis, resulting in a reduction of serum cortisol concentration. Published clinical guidance recommends titrating the dose of metyrapone against the measured serum cortisol concentration. 1 However, serum cortisol concentration is routinely measured by immunoassay in clinical laboratories and it has been well documented that there are interferences in these assays in those patients with altered steroid metabolism due to either pathological processes or drug treatment. 2,3 For example, an increased circulating concentration of 11-deoxycortisol in patients receiving metyrapone therapy has previously been considered as a potential interferant in serum cortisol immunoassays. 4
The increasing availability of liquid chromatography tandem mass spectrometry (LC-MS/MS) assays for steroid hormones may circumvent this issue and assays that measure multiple steroids may also be used to identify the nature of any interferences observed.
In this report, we have firstly, investigated the specificity of an immunoassay for cortisol in the presence of high concentrations of structurally related steroids; and secondly, using LC-MS/MS, we have identified which steroids are raised in patients receiving metyrapone therapy.
Methods
Stock solutions of 11DOC, 21-deoxycortisol (21DOC), androstenedione (A4), 17-hydroxyprogesterone (17OHP), 21-hydroxyprogesterone (21OHP), progesterone and testosterone were made in methanol (Sigma, Poole, Dorset). These stock solutions were used to spike a single anonymized serum sample to give a concentration of each steroid of 500 nmol/L. The serum before and after spiking was analysed in singleton using a Roche electrochemiluminescent cortisol immunoassay (Roche, Manneheim, Germany). Any apparent increase in measured cortisol concentration was noted and apparent % cross-reactivity was calculated for each steroid.
Sera from two patients, undergoing treatment with metyrapone, were analysed using an LC-MS/MS steroid assay for cortisol, 11DOC, 21DOC, 17OHP and A4. This was to identify which steroids, if any, were elevated and therefore have the potential to interfere in a cortisol immunoassay. LC-MS/MS was performed on a Waters Acquity coupled to a Waters Quattro Premier, using samples prepared by protein precipitation. On-line solid-phase extraction was performed on a 4 × 2 mm Gemini cartridge followed by chromatography on a 25 × 4.6 mm Onyx C18 analytical column (Phenomenex, Macclesfield, UK). 5 Serum cortisol was also measured on these samples using the Roche electrochemiluminescent method mentioned above.
Results
The results of the LC-MS/MS assay showed an increase in serum concentration of 11DOC in both patients, consistent with them receiving metyrapone therapy. The concentration of 11DOC was much higher in one patient, and this patient also had a very low serum cortisol measured by LC-MS/MS, suggesting almost a complete block of 11beta hydroxylase (Figure 1a). A surprising finding was a significantly raised 17OHP in this patient and A4 in the other patient (Figure 1b). No 21DOC was detectable in either patient.
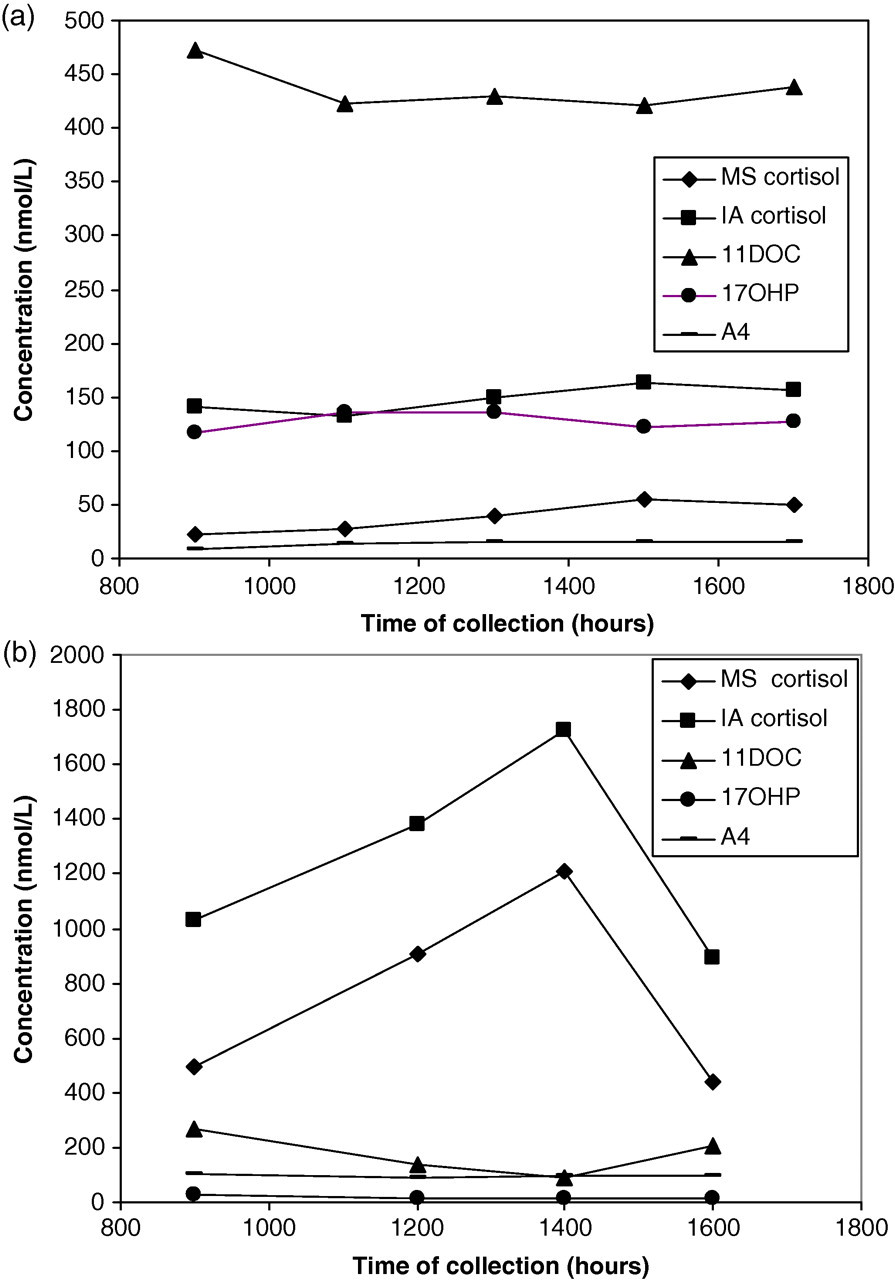
Steroid measurements for two-day curves in patients receiving metyrapone therapy. (a) A patient with an adenocorticotrophic hormone (ACTH) secreting pituitary adenoma receiving 1 g metyrapone four times daily (b) A patient with adrenocortical carcinoma receiving 1 g metyrapone four times daily. MS, liquid chromatography tandem mass spectrometry measurement; IA, immunoassay measurement
The results of the interference studies showed that 11DOC, 17OHP and 21DOC all have a positive interference in the cortisol immunoassay. Both 17OHP and 11DOC had a cross-reactivity of 28% with the serum cortisol immunoassay, with measured cortisol increasing from 369 to 507 nmol/L. However, 21DOC showed much greater cross-reactivity (76%), with measured cortisol increasing from 369 to 750 nmol/L. The immunoassay kit insert reports cross-reactivities of 4.1% for 11DOC (29 mmol/L added), 45.4% for 21DOC (2.9 mmol/L added) and 1.5% for 17OHP (28 mmol/L added).
Comparing the cortisol values obtained by immunoassay and LC-MS/MS shows a significant over-estimation of cortisol by immunoassay. The degree of over-estimation was variable both between the patients (43–541%) and between multiple samples from each patient (43–109% and 196–541%). Based on our findings, the extent of this interference cannot be explained by the interference of 11DOC alone or the combined interference of 11DOC and 17OHP. The expected immunoassay cortisol, calculated by combining the cross-reactivity of the antibody with 11DOC and 17OHP with the LC-MS/MS cortisol, shows over-estimation in one patient and under-estimation in the other.
Discussion
This study has shown that several steroids, produced as a result of metyrapone treatment, cause interference in the Roche cortisol immunoassay. It is of concern that the observed interference appeared much greater than that indicated in the kit insert. This discrepancy may be due to the supra-physiological concentrations used by the manufacturer in their interference studies. In contrast, the concentrations used in the present study approximated those typically seen in patients with altered steroid metabolism. Interferences in other manufacturers' immunoassays have also been reported, indicating this is not just a problem for a single manufacturer. 6
The level of interference seen in the sera of patients receiving metyrapone cannot be explained solely by 11DOC as was previously thought and appears to be variable within- and between-individuals. The apparent difference between the observed levels of interference in the two cases outlined in this study may have been due to different steroid production as a result of different aetiologies. One of the patients presented here demonstrated an increase in 17OHP with a near complete block of 11beta hydroxylase and this may have contributed to the interferences seen. If this is substantiated, interference by 17OHP may have implications when measuring cortisol in children with suspected congenital adrenal hyperplasia (CAH).
In conclusion, this small study has shown that care needs to be taken in interpreting serum cortisol concentrations measured by immunoassay in patients receiving metyrapone. We would recommend that LC-MS/MS be the method of choice when monitoring serum cortisol in these patients.
DECLARATIONS
