Abstract
Glucose-6-phosphate dehydrogenase (G6PD), an X-linked hereditary deficiency, is the most common of all clinically significant enzyme defects. While many drugs are responsible for haemolytic anaemia in G6PD-deficient patients, acetaminophen's imputability is still under debate, although an overdose of this drug can provoke acute haemolytic events. We report a case of a Philipino child carrying the G6PD-Vanua Lava mutation with acute haemolytic crisis related to infection in progress and acetaminophen's administration. Fever and concomitant infection, through an increment of erythrocyte glutathione depletion, sensitized the infant to the haemolytic event. In this condition, acetaminophen (or paracetamol [PCM]) was capable of inducing a haemolytic crisis in our G6PD-deficient patient although administered under standard conditions. PCM seems to have induced the haemolytic event, probably by the alteration of its catabolism due to dehydration and fever. The enzymatic G6PD instability associated to the presence of the G6PD-Vanua Lava mutation could have led to an increment of red blood cells' sensitivity to lysis; hence, it is possible that PCM toxicity may also be due to the presence of this particular mutation. Finally, we propose a new biochemical classification of this G6PD variant.
Introduction
Glucose-6-phosphate dehydrogenase (G6PD) is a critical enzyme in the redox metabolism of red blood cells (RBCs), since it catalyses the first and rate-limiting step of the pentose phosphate pathway (PPP). Since the PPP is the unique NADPH source, the alteration of this pathway does not enable the RBCs to counterbalance the oxidative stress triggered by several drugs, resulting in failed regeneration of the reduced form of glutathione (GSH) and finally determining acute haemolytic anaemia in G6PD-deficient individuals. 1
G6PD deficiency diagnosis is based on an enzymatic assay and on the search of the genetic alteration involved. The mutation identification can improve the clinical management of the patient. A reliable evaluation of the residual enzymatic activity in G6PD-deficient patients is considered of primary importance for the mutation classification according to World Health Organization (WHO) recommendations. 1
We now report the case of a Philipino child carrying the G6PD-Vanua Lava mutation and presenting with acute haemolytic crisis. At the clinical examination, his parents referred that they had administered only paracetamol (PCM) and that no other medications were given during the three previous weeks.
Although this child presented with fever due to a non-established, two-day-old latent infection, we hypothesized that the haemolysis was triggered by the PCM administered 10 h before. For this reason, we report this episode as a possible case of PCM-induced haemolysis in a G6PD-deficient individual.
Methods
Patient
A five-year-old Philipino child (proband) was referred to the emergency department with vomiting, jaundice, haematuria and fever. The results of laboratory tests, performed 10 h following PCM administration, are reported in Table 1. Clinical and laboratory findings were compatible with acute severe haemolysis, so he was given two blood transfusions followed by rehydration treatments due to evident signs of vomit-induced dehydration. Seven days after hospitalization he was in good health (Table 1).
Proband's laboratory tests assayed at DEA admittance and during PICU hospitalization
The values considered as indices of haemolysis and dehydratation are reported in bold
DEA, emergency department; PICU, intensive paediatric care unit; PCM, paracetamol
G6PD family investigation: enzymatic assay and gene analysis
Due to the fact that the child had undergone blood exchanges, we could not assay the enzymatic G6PD activity in his blood. After parental informed consent, we performed the genetic test on collected buccal cells. Further biochemical and genetic investigations of the G6PD enzyme were also performed on both his brother (eight years old) and his mother. G6PD activity was determined from a de-leucocytated blood sample by means of a commercially available kit (Trinity Biotech, Co. Wicklow, Ireland), following the WHO recommendations. 1 In addition, the G6PD/6-PGD (6-phosphogluconate dehydrogenase) ratio methodology (a recommended test for heterozygous women's identification) was performed on the mother (by means of a kit provided by Nurex, Sassari, Italy). 1
Genomic and buccal cell DNAs were obtained by means of an automatic DNA extractor (Maxwell™ 16, Promega, Madison, WI, USA) and phenol/chloroform extraction techniques, respectively. Genetic screening was performed following a specific protocol standardized in our laboratory. 2 Since this preliminary test resulted negative, we sequenced the whole G6PD gene, following the literature protocols. 3
PCM glucuronidation and sulphation activities: UGT1A1, UGT1A6 and SULT1A1 gene investigation
To exclude additional alteration of the PCM glucuronidation and sulphation system, we investigated the most common polymorphisms in the UGT1A1, UGT1A6 and SULT1A1 genes which may contribute to the interindividual variations of observed PCM toxicity in the general population. 4,5
Results
Sequencing analysis of the entire G6PD gene performed on the patient revealed the presence of the c.383T > C missense mutation, known as G6PD-Vanua Lava (p.L128P) 6 : the same mutation was found in both his brother and mother. As expected, the enzymatic assay showed G6PD deficiency in the brother (2.2 IU/gHb [normal values 7–20]). In contrast, the mother had normal G6PD activity (19.2 IU/gHb).
Finally, the genetic screening for UGT1A1, UGT1A6 and SULT1A1 gene polymorphisms yielded wild-type alleles, UGT1A1*1, UGT1A6*2 and SULT1A1*1, in the proband.
Discussion
G6PD-Vanua Lava is reported as a Class II mutation. This class generally shows a G6PD enzyme residual activity below 10%. In contrast, the G6PD activity assayed in the proband's brother was typical of a Class III G6PD group, with a residual value of approximately 15% (range values for Class III: from 15% to 60%). 1 In previous papers regarding the Vanua Lava mutation, 6,7 G6PD activity was assayed in umbilical cord cells by means of a semi-quantitative method, while genetic tests were performed by single-strand conformation polymorphism (SSCP) screening; thus, some methodological biases could have affected the results obtained in these reports, also considering that SSCP results were not confirmed by whole gene sequencing.
Therapeutic doses of PCM are considered as a safe antipyretic-analgesic in patients with G6PD deficiency, 8 while an overdose of this drug could be responsible for acute haemolytic crises. 9 However, conflicting data concerning the true safe therapeutic dosage and use of PCM are available since at least two cases of haemolysis in G6PD-deficient individuals, following therapeutic doses, have been reported in literature. 10,11
In humans, therapeutic doses of PCM are primarily metabolized (about 60%) by glucuronidation and sulphation, whereas only a small fraction of PCM (5%) is excreted unmodified by the kidney. The remaining fraction, usually ranging from 5% to 15%, is oxidized by liver cytochrome P450, resulting in the formation of N-acetyl-p-benzoquinoneimine (NAPQI), then scavenged by hepatic GSH. Following overdose, the rate and quantity of NAPQI formation may outstrip GSH supply and regeneration. When GSH stores are depleted below a critical value (about 30% of normal stores), free NAPQI rapidly and covalently binds critical cell proteins, inducing a series of events that may result in cell death. As long as sufficient GSH is present, the liver is protected from injury. 12
The aim of this case report was to provide additional information regarding possible adverse reactions (in this case a haemolytic crisis) produced by PCM in patients affected by peculiar acute conditions, such as fever, vomit-induced dehydration and flu. In these conditions, even therapeutic PCM administration could turn out to be toxic, particularly due to haemo-concentration subsequent to dehydration.
In addition, in order to explore other possible mechanisms underlying the PCM metabolism in our patient, we indirectly evaluated the PCM glucuronidation and sulphation activities by means of genetic polymorphism analysis. Since this screening was negative, we hypothesized that the dehydration could have altered the relative concentration of PCM, both at renal and at hepatic levels mimicking an ‘overdose’. In this way, higher amounts of toxic NAPQI molecule were present, thereby activating the liver's response, which culminated in higher GSH depletion (Figure 1). Since GSH plasmatic levels correlate with RBC number, and significant amounts of GSH are released by RBCs, we can speculate that RBCs could be considered as an important GSH tissue source capable of significantly contributing to extra and intracellular GSH homeostasis. 13
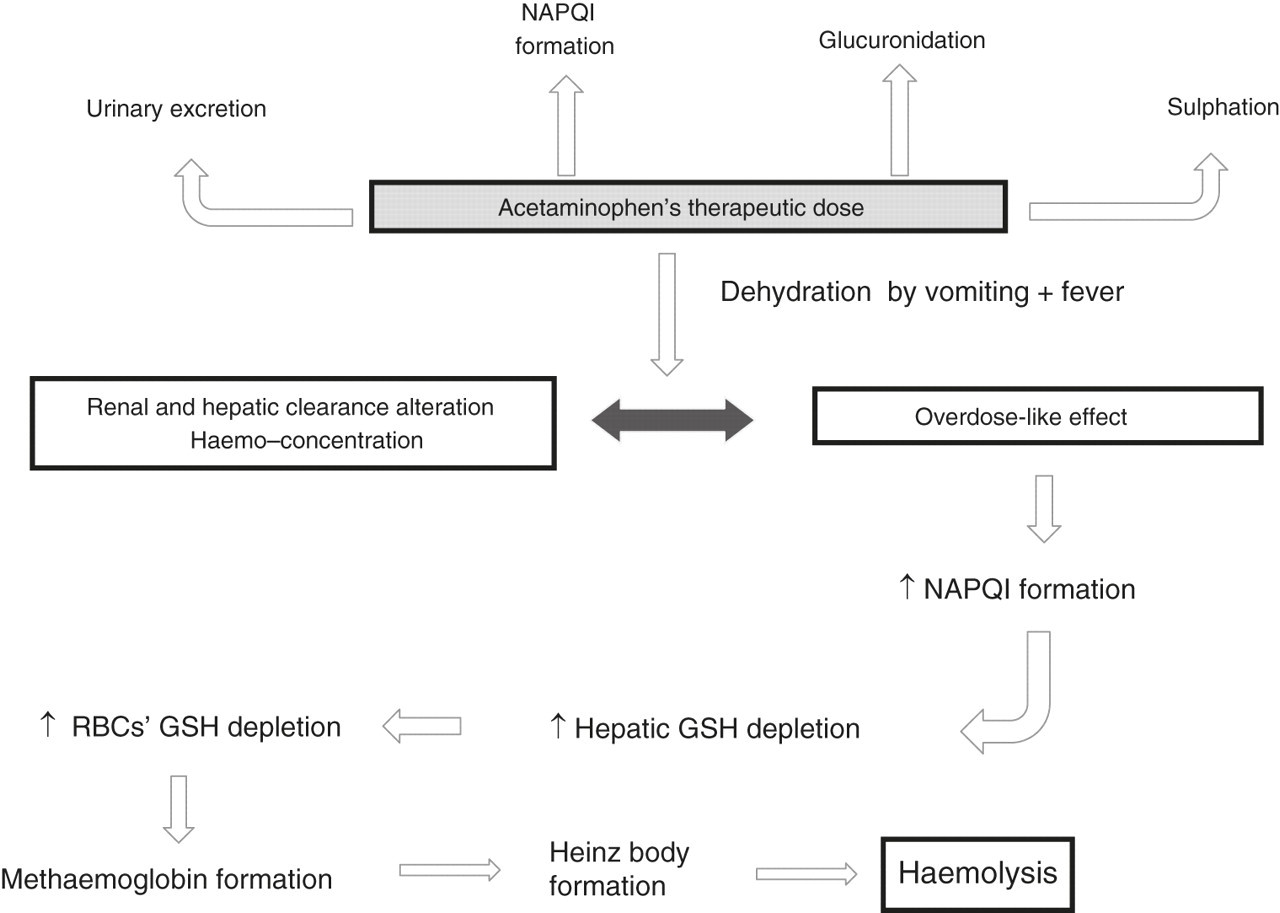
Possible mechanisms of PCM's toxicity occurred in the child carrying the G6PD-Vanua Lava mutation. Therapeutic doses of PCM are principally metabolized by glucuronidation and sulphation (60–80%), while 5% of PCM is excreted unmodified by the kidney. The remaining fraction (from 5% to 15%) is oxidized by liver cytochrome P450, producing NAPQI, which is scavenged by hepatic GSH. Following overdose, the NAPQI formation may exceed the GSH regeneration: when GSH stores are depleted below 30% of normal concentrations, the highest levels of free NAPQI may lead to RBC haemolysis via methaemoglobin formation. PCM, paracetamol; NAPQI, N-acetyl-p-benzoquinoneimine; GSH, glutathione; RBC, red blood cell
As a consequence, since both liver and RBCs present lower GSH levels in patients with G6PD deficiency, the possible RBC ‘buffer effect’ could be reduced, exposing G6PD-deficient RBCs to several oxidative insults, as occurs during high fever peaks. This hypothesis is in agreement with a previous paper 14 showing decreased erythrocyte and hepatic GSH levels in children treated with high doses of PCM.
Finally, we believe that the G6PD-Vanua Lava mutation should be considered as a Class III mutation for the following reasons: (1) in our study, the enzymatic assay was performed on a de-leucocytated peripheral blood sample by quantitative biochemical tests; (2) the proband's brother was healthy, with fully normal haemocromocytometric values and fitting with the age; (3) we performed a complete analysis on the entire G6PD gene; and (4) similar results were also obtained from a different patient carrying a mutation involving the same Vanua Lava codon, but classified as Class III mutation. 15
Conclusions
This case presents the following elements of interest:
This is the third case reported in literature showing haemolytic crises after administration of a therapeutic-dose PCM in a G6PD-deficient patient, presenting with vomit, fever and flu symptoms. Fever and possible infections may have contributed to erythrocyte GSH depletion, promoting the haemolytic event.
G6PD-Vanua Lava mutation, reported for the first time in Italy, is very frequent among the Philipino population. The enzyme impairment, caused by this mutation, determines the alteration of the enzyme stability.
6
This feature increases RBCs' sensitivity to cell lysis. Indeed, we can hypothesize a direct correlation between PCM and this peculiar mutation. Finally, considering the high rate of migratory fluxes around the world, we should reflect on the opportunity of using G6PD gene scanning as an additional approach for a more complete molecular investigation in migrants.
We emphasize that a possible limit of this paper is represented by the lack of serum PCM assay in our proband; this test was not performed for two reasons: (1) the infant was immediately transfused and (2) this assay was not required at the income in our emergency department since our protocols do not include this type of test, above all when the drug is administered under medical control and at therapeutic dosage.
Finally, we cannot exclude that other unknown biochemical or genetic factors may have also influenced the symptoms and the patient's PCM response.
DECLARATIONS
