Abstract
There are many causes of interference in immunoassays causing erratic patient results. A method-specific interference due to antiruthenium antibodies in Roche free thyroxine (fT4) and free triiodothyronine (fT3) assays has been described previously. As a result, a new generation fT4 assay has been introduced by Roche. We describe six cases of interference due to antiruthenium antibodies, where in four cases interference in the Roche thyroid-stimulating hormone (TSH) assay was found as well. This raised the question as to whether other assays on this platform would also give incorrect results in patients with antiruthenium antibodies. Interference due to antiruthenium antibodies was suspected because of discrepancies between clinical presentation and/or TSH, fT4 and fT3 results. Samples of these six patients were re-analysed in Roche Diagnostics Laboratory, where it was demonstrated that the found discrepancies were indeed caused by interfering antiruthenium antibodies. Subsequently, these patients were asked to donate some blood once more for further evaluation, and three subjects agreed to participate. Their plasma was used to assay 18 analytes on Modular E and on a ruthenium-independent platform. The results were compared taking into account the known differences between distinct methods. As expected, significant interference was found in TSH. Also, in the new generation fT4 assay, ruthenium-induced interference was still present. However, the other assays, both competitive and immunometric, did not show clear interference. We therefore conclude that although antiruthenium antibodies theoretically can interfere in all assays on the Modular E platform, this kind of interference is found in the thyroid hormone assays, without marked interference in the other assays.
Introduction
There are many causes of interference in immunoassays causing erratic patient results. 1,2 These erroneous results potentially lead to unnecessary, expensive and possibly harmful investigations and treatment. A method-specific interference due to antiruthenium antibodies in the Roche free thyroxine (fT4) and free triiodothyronine (fT3) assays has been described previously. 3–5 To overcome this problem in the fT4 assay, Roche introduced a new generation fT4 assay at the end of 2008. Here, we describe six cases of identified antiruthenium antibody interference. In four of these patients, this kind of interference was demonstrated in the Roche thyroid-stimulating hormone (TSH). The observation of interference in the TSH assay raised the question as to whether other assays on this platform would also give incorrect results in patients with antiruthenium antibodies.
Case reports
In the first patient, a 73-year-old woman, thyroid function was monitored because of atrial fibrillation in 2005. Repeated tests (Roche Modular E170) showed normal TSH of 0.53 mU/L (reference range 0.4–4.2 mU/L) with increased fT4 of 43.7 pmol/L (reference range 12.0–22.0 pmol/L) and fT3 of 17.3 pmol/L (reference range 2.8–7.2 pmol/L). Thyroid scintigraphy showed a proper uptake, appropriate with long existing Graves' hyperthyroidism or toxic multinodular goiter. These results together were interpreted as atrial fibrillation due to hyperthyroidism. Subsequently, the patient was treated with strumazol, digoxin, metoprolol and acenocoumarin. About a month later, thyroid function tests showed an elevated TSH of 11.65 mU/L with a normal fT4 of 17.9 pmol/L, but still an elevated fT3 of 13.5 pmol/L. Although these results were not completely understood, levothyroxine was added because of the increased TSH level. Almost two years later, thyroid function tests were still abnormal with TSH of 0.62 mU/L, fT4 of 28.8 pmol/L and fT3 of 16.4 pmol/L. However, the patient did not have any complaints of hyperthyroidism. Because of this discrepancy in clinical presentation and laboratory results, the possibility of an analytical interference was considered. Therefore, residual plasma was sent to another laboratory using a different method for TSH, fT4 and total T3 concentration. Completely normal fT4 and total T3 results were obtained (see Table 1, Alternative method). All thyroid medication was stopped immediately.
Thyroid hormone levels in six patients measured with different platforms
nd: not determined; TSH, thyroid-stimulating hormone; fT4, Roche free thyroxine; fT3, Roche free triiodothyronine; TT3; total triiodothyronine
*Determined by new-generation fT4 assay
†Determined by Iuminescence immunometric assay (ILMA; Brahms)
‡Determined by fluoroimmunoassay (FIA; Delfia)
§Determined by in-house radioimmunoassay (RIA)
#Using modified research conjugate preventing interference by ruthenium antibodies
The second case concerns a 56-year-old man without any history of thyroid disorders. In July 2008, his thyroid function was tested routinely. A normal TSH of 2.78 mU/L, but a very high fT4 concentration of 70.8 pmol/L was found. The fT3 concentration was within the reference range. Because of these discrepancies between TSH, fT4 and fT3 levels, and the fact that the patient was clinically euthyroid, analytical interference in the fT4 assay was suspected. Analysis of that sample with another immunoassay system demonstrated a completely normal fT4 concentration of 17.7 pmol/L (Table 1, Alternative method).
Both the third and fourth patients were on levothyroxine treatment for previously diagnosed autoimmune hypothyroidism. Analytical interference in the TSH assay was considered by the endocrinologist of our hospital, because the TSH results constantly remained elevated (Table 1, Modular E). To investigate possible interference, TSH dilution tests showed non-linear results. The same was found after incubation of the patient sample in mouse serum. The measurement of TSH after pretreatment with specific heterophilic blocking tubes containing blocking reagents to eliminate a possible heterophilic antibody showed incongruent results as well. Therefore, material was sent to another laboratory to assay thyroid function with a different platform. Surprisingly, the alternative method measured significantly increased TSH concentrations in comparison to the Roche method, whereas it is known that this method measures at average 14% lower than the Roche method (Figure 1). In the alternative assay, linearity to dilution was found in both patients, which makes the possibility of TSH interference in that assay less likely. Therefore, the Roche TSH concentrations seemed to be falsely decreased in the patients. It seemed that both patients were under-treated with levothyroxine. This was partly unnoticed by the concomitant falsely increased measured fT4 concentrations (Table 1, Modular E).
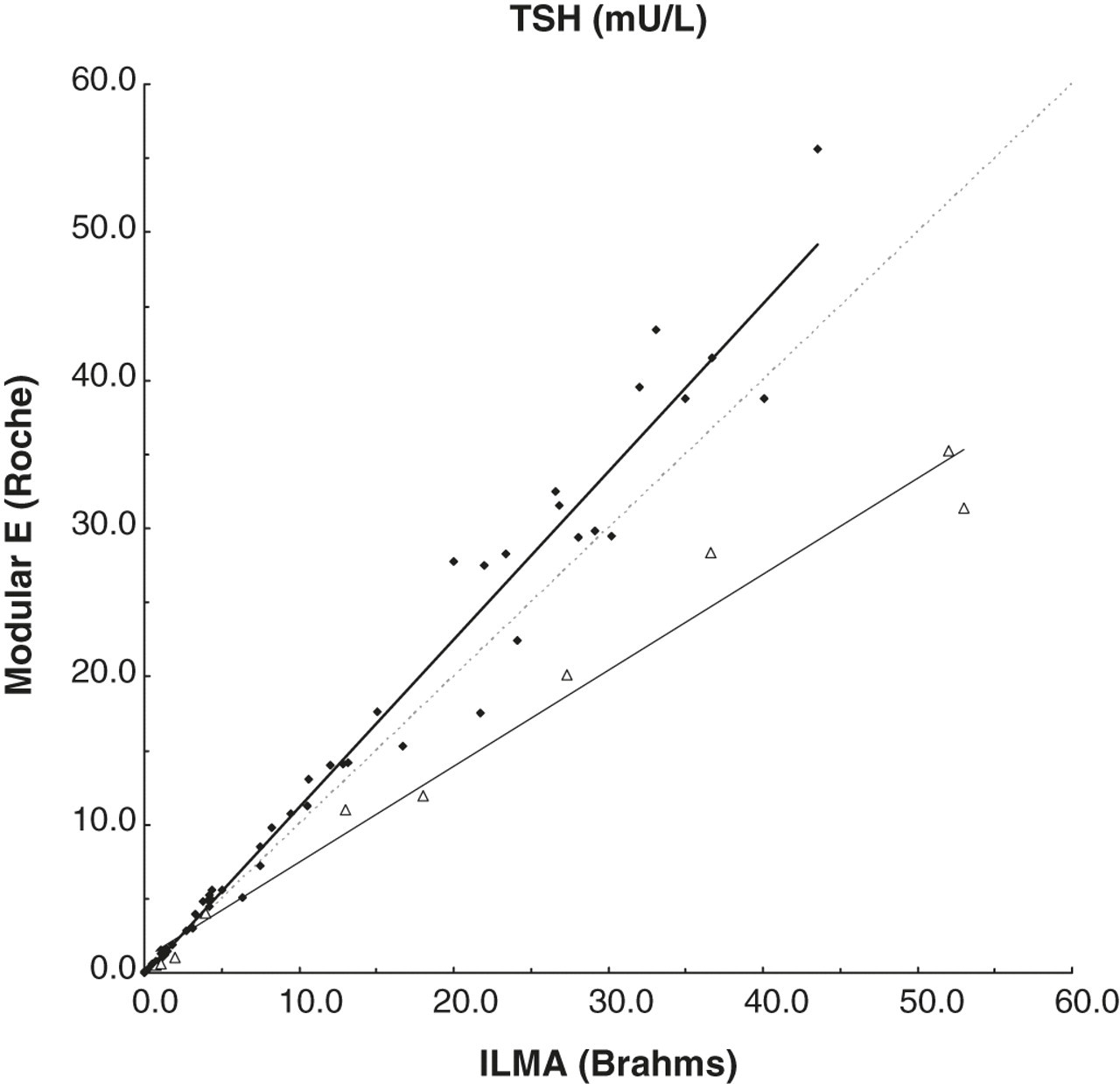
Thyroid-stimulating hormone (TSH) (mU/L) concentrations determined by luminescence immunometric assay (ILMA) and the ruthenium-dependent Roche method in 51 samples without known antibodies to ruthenium (♦; Passing-Bablok, Roche = 1.147 and ILMA–0.0328) and in 10 samples of five subjects with interference due to antiruthenium antibodies (Δ; in subject 2, TSH was not analysed by ILMA)
In the fifth patient, a 27-year-old girl with diabetes mellitus type I, thyroid function tests were done annually to check for autoimmune thyroid disease. Repeated tests showed normal TSH of 1.18 mU/L, increased fT4 of 36.9 pmol/L and increased fT3 of 11.5 pmol/L. In a later sample drawn in December 2008, after introduction of the new generation fT4 assay, the fT4 concentration was just within the reference value (21.5 pmol/L). Because of the previously obtained experience, analytical interference was suspected, and TSH, fT4 and total T3 concentrations were measured using another method which gave completely normal values (Table 1, Alternative method).
In the last patient, an 86-year-old woman, also on levothyroxine treatment for previously diagnosed autoimmune hypothyroidism, thyroid function tests showed quite varying results. Combinations appropriate to subclinical hypothyroidism were seen with elevated TSH of 11.00 mU/L, fT4 of 20.3 pmol/L and fT3 of 6.1 pmol/L. Later results were interpreted as euthyroid hyperthyroxinemia (TSH 1.92 mU/L, fT4 30.1 pmol/L and fT3 6.4 pmol/L). It was decided to verify whether analytical interference could play a role, and so residual plasma was sent to another laboratory. Again, falsely decreased TSH levels and falsely elevated fT4 and fT3 levels were confirmed (Table 1, Alternative method).
Methods and results
All the original thyroid function tests in the above presented patients were measured by the Roche Modular E170 analyser (Roche, Mannheim, Germany) in the laboratory of Medial (Hoofddorp, the Netherlands). The TSH limit of detection was 0.01 mU/L. The inter-assay coefficient of variation (CV) was less than 3.0%, ranging from 0.60 to 30.0 mU/L. The fT4 detection limit is 0.3 pmol/L, while the inter-assay CV was 2.8% and 3.6% at fT4 concentration of 14.6 and 81.8 pmol/L, respectively. The detection limit for the fT3 assay is 0.4 pmol/L; inter-assay CV was 4.5% and 7.6% at levels of 22.9 and 4.0 pmol/L, respectively. To evaluate the possibility of interference in the fT4 and fT3 and later in the TSH assay as well, remaining samples were assayed for TSH, fT4 and/or total T3 in another laboratory (AMC, Amsterdam, the Netherlands). At that site, TSH concentrations were measured by solid phase two-site luminescence immunometric assay (ILMA; Brahms, Berlin, Germany). The limit of detection was 0.01 mU/L, and the inter-assay CV was 3.6 (15.7 mU/L) − 7.7% (0.22 mU/L). FT4 concentrations were assayed by two-step fluoroimmunoassay (FIA; Delfia, PerkinElmer, MA, USA) with an inter-assay CV of 5.4% and 6.8% at fT4 concentrations of 14.5 and 43.7 pmol/L, respectively. The assay detection limit was 2.0 pmol/L. Total T3 levels were measured by an ‘in-house’ radioimmunoassay (RIA). The detection limit was 0.3 nmol/L and the inter-assay CV was less than 5% at the reference range. Due to the kind of interference found (i.e. the combination of falsely decreased values in the immunometric assay and falsely increased values in competitive assays), interference due to antiruthenium antibodies was suspected. To examine this possibility, residual samples of the six patients were analysed in the Roche Diagnostics Laboratory. There, it was demonstrated that the found discrepancies were indeed explained by interfering antiruthenium antibodies. The use of a modified research conjugate to prevent the interference by ruthenium antibodies significantly increased TSH and significantly decreased fT4 and fT3 concentrations in subjects 3, 4, 5 and 6, subjects 1, 2, 4 and 5, and subjects 1, 4 and 5, respectively (see Table 1, Modular E versus Roche Diagnostics Laboratory).
In particular, the observation of interference due to antiruthenium antibodies in the TSH assay raised the question as to whether other assays on this platform would also give incorrect results in patients with antiruthenium antibodies. To answer this issue, all six patients were asked to donate some blood for further evaluation. Three patients, subject 4, 5 and 6, agreed to participate in the study. Their samples were collected by venipuncture and plasma and serum was used to assay 18 different analytes on both Roche Modular E and a ruthenium-independent platform in duplicate. TSH and new generation fT4 assay concentrations were measured to check whether the ruthenium-induced interference was still present. These ruthenium-independent methods were similar to the methods mentioned before. Cortisol concentration with the alternative platform was determined by Immulite 2000 (Siemens, IL, USA). Estradiol and progesterone were determined by RIA (Siemens and Orion Diagnostica [Espoo, Finland], respectively). Concentrations of CA 15.3, CA 19.9, CA 125 and CEA were determined in another laboratory (VUMC, Amsterdam, The Netherlands) by Centaur (Siemens). In this laboratory, AFP, HCG, folate and vitamin B12 levels were measured as well using the Architect (Abbott, IL, USA). LH, FSH and prolactin concentrations were assayed by immunofluorometric assay (IFMA; Delfia). Finally, ferritin and troponin-I levels were measured by Beckman Dxi (Beckman Coulter, CA, USA) immunoassays (UMCU, Utrecht, The Netherlands). The results of similar tests were compared, taking into account the known differences between the distinct methods. This was approached as follows. Data derived from the Dutch external quality program 2009 were used to calculate the Modular E/Alternative method mean ratio and range (defined by mean method ratio ± 2SD) for each assay. Subsequently, subject Modular E/Alternative method ratios were calculated for all different parameters. Interference by antibodies to ruthenium was suspected when the subject ratio was below the method range for immunometric assays or above the method range for competitive assays in more than one subject. Measured concentrations and calculated ratios are presented in Table 2.
Assay results and calculated method and patient ratios of different immunoassays in three subjects with antiruthenium antibodies
cd, cycle dependent; nd, not determined; TSH, thyroid-stimulating hormone; fT4, Roche free thyroxine; CA, cancer antigen; CEA, carcino embryonic antigen; AFP, alpha-fetoprotein; HCG, human chorionic gonadotropin; LH, luteinizing hormone; FSH, follicle-stimulating hormone
Method ratios and ranges were derived from data of the Dutch external quality program 2009 (n = 12). The range is defined by mean ratio ± 2 SD. Discrepant subject ratios as defined in the text are presented in bold.
*Determined by immunofluorometric assay (IFMA), Brahms
†Determined by Roche new generation fT4 assay
‡Determined by fluoroimmunoassay (FIA)/IFMA, Delfia
§Determined by Centaur, Siemens
#Determined by Architect, Abbott
**Determined by Beckman Dxi, Beckman Coulter
††Determined by Immulite 2000, Siemens
‡‡Determined by radioimmunoassay (RIA), Siemens
§§Determined by RIA, Orion Diagnostica
As expected, significant interference was found in the Roche TSH assay again. Also, in the new generation fT4 assay, ruthenium-induced interference appeared to be still present. However, all the other assays, with the exception of AFP, progesterone and troponin, as well as competitive and immunometric, did not show striking interference, that could be attributed to antiruthenium antibodies. In all three subjects, AFP Modular E/Alternative method ratios were below the method AFP range, which might suggest interference. In the progesterone assay, potential interference by antiruthenium antibodies could not be investigated because in all three subjects progesterone values were below the detection limit. For Troponin, it was not possible to calculate the Modular E/Alternative method ratio as the alternative method did not participate in the mentioned external quality program.
Discussion
In general, interference in immunoassays can be caused by many different mechanisms including heterophilic antibodies, interfering drugs and rheumatoid factor. 1,2 Moreover, assay-specific interference has been described, such as antithyroid hormone antibodies resulting in falsely elevated or falsely lowered analyte concentrations depending on the assay type. 6 Likewise, a method-specific interference due to antiruthenium antibodies in the Roche fT4 and fT3 assays has been described. 3–5 The presence of antiruthenium antibodies falsely elevates the measured concentrations of fT4 and fT3. In these competitive electrochemiluminescence immunoassays, a ruthenium complex is used as the label. During the assay, free hormone in the sample reacts with anti-T4- or anti-T3 ruthenium-labelled antibody in the presence of biotinylated T4 or T3 and streptavidin-coated microparticles. The higher the free concentration of hormone in the sample, the lower the amount of ruthenium-labelled antibody binding to the biotinylated T4 or T3 microparticle-complex. In the final step, the amount of light emission during the electrochemiluminescence reaction depends on the quantity of ruthenium present in the measuring cell. The antiruthenium antibodies diminish the amount of signal, resulting in falsely elevated fT4 and fT3 concentrations.
In addition to the influence of antiruthenium antibodies on the Roche fT4 and fT3 assays, we observed in four of our patients (subjects 3–6), interference due to these antibodies on the Roche TSH assay. Interference due to antiruthenium antibodies might also explain the relatively low Roche TSH concentration in subject 1. To our best of knowledge, this report describes the first cases of patients with erratic decreased TSH concentrations due to the interference of antiruthenium antibodies in the literature. Two earlier papers of Ando et al. 3 and Sapin et al. 4 primarily reported interference of antibodies to ruthenium affecting the Roche fT3 assay. In a recent paper of Heijboer et al., 5 antiruthenium interference was demonstrated for the first time in the Roche fT4 assay. In those patients, no interference was observed in the TSH assay, although ruthenium is present in the TSH assay as well. Indeed, interference due to antiruthenium antibodies is less expected in the robust TSH immunometric assay in comparison with the more difficult competitive fT4 and fT3 measurements. The extremely low plasma fT4 and fT3 concentrations make it necessary to use an oligomerized ruthenium label to generate sufficient signal. Such a label is more susceptible to interference by antiruthenium antibodies than the monomerized ruthenium label used in the Roche TSH assay. Nevertheless, in our subject the TSH assay is influenced significantly by antiruthenium antibodies. The effects of these antibodies were clearer with increasing TSH levels. As TSH concentrations in the patients described by Heijboer et al. were just within or below the reference values, possible interference on the TSH assay was probably not noted.
The observation of interference due to antiruthenium antibodies in the TSH assay prompted us to investigate whether other assays using a monomerized ruthenium label on this platform would also give incorrect results in patients with antiruthenium antibodies. We compared several other assays of the Roche module with ruthenium-independent platforms using samples containing antibodies to ruthenium. The Modular E/Alternative method ratios did not differ from those obtained in the patient samples with exception of the AFP ratios. However, the AFP concentrations of the samples used in the Dutch external quality control program were much higher than present in the subject samples of this study. Therefore, the calculated method ratio may not be usable and as a result, antiruthenium antibody interference is hard to demonstrate in the Roche AFP assay in this study. The same applies to the Roche progesterone assay. For the other assays, we conclude that the results are not consistent with clinically significant interference in the Roche assays. We do not have an explanation as to why the interference is not seen in these assays. An abnormal ratio for a given compound is used as an indication that interference is likely. However, a normal ratio does not totally exclude the presence of interference. In subject 6, the patient ratio in Table 2 does not indicate interference in the TSH assay, whereas marked interference was present in the fT4 assay in that sample, and in the TSH assay in a previous sample (Table 1). The samples of the data presented in Table 2 were not sent to Roche Diagnostics Laboratory for further analysis. Therefore, it is possible that a slight degree of interference is missed as the samples were not treated with the specific antiruthenium blocking agent.
In our patients, the falsely elevated fT4 and fT3 concentrations led to confusion in the interpretation of the thyroid function tests. Elevated fT4 and fT3 concentrations in combination with TSH levels within the reference range are termed euthyroid hyperthyroxinemia. The differential diagnosis of this condition includes some rare disorders like pituitary thyroid hormone resistance, TSH-secreting pituitary adenoma or familial dysalbuminemic hyperthyroxinemia. However, this condition can also be caused by analytical problems due to interfering antibodies, thyroid hormone binding protein abnormalities or medication influencing the thyroid hormone binding to binding proteins. Consequently, additional investigations are necessary to confirm whether true hyperthyroxinemia may exist or not. As a consequence, the relatively common occurrence of antiruthenium antibodies is an important problem. 4 Roche Diagnostics introduced a new generation fT4 assay at the end of 2008 with reduced susceptibility to antiruthenium interference. We agree with McKillop et al. 7 that the introduction of this new fT4 assay has reduced the interference from antiruthenium antibodies, but unfortunately the problem has not been solved completely. Consecutive measurement using both the old and the modified assay do demonstrate an improvement in response to antiruthenium antibodies, but still falsely increased fT4 concentrations are apparent (i.e. subject 5).
It is thought that the development of antibodies to ruthenium could be by intake of ruthenium through the food chain or exposure to residues in the clothes, as ruthenium is used as an oxidant in the dying process of clothes. More likely is that these antibodies are heterophile, and the reaction against ruthenium is co-incidental. 8,9
In conclusion, ruthenium antibodies significantly interfere in the Roche TSH assay, and continue to disturb the new generation fT4 assay. Although these antibodies can interfere theoretically in all assays on the Modular E170 platform, clinically important interference was found in the thyroid function assays, without marked interference in the other assays.
DECLARATIONS
