Abstract
Background
Ischaemia-modified albumin (IMA) is a relatively new marker of ischaemia. IMA has not been studied previously in pleural effusions due to congestive heart failure (CHF). The aim of our study was to assess the diagnostic value of IMA in the pleural fluid (PF) and serum for the identification of pleural effusion due to CHF.
Methods
The concentrations of pleural fluid and serum IMA were measured in a total of 40 patients: 10 with CHF and 30 with non-cardiac failure (10 with acute pulmonary embolism, 10 with parapneumonic effusion and 10 with malignancy). The area under the curve (AUC) quantified the overall diagnostic accuracy of the tests.
Results
The study demonstrated that IMA concentration was higher in both pleural fluid and serum of CHF patients compared with non-cardiac patients. PF and serum IMA demonstrated AUCs of 0.927 (95% CI: 0.844–1.00, P < 0.001), and 0.792 (95% CI: 0.653–0.930, P = 0.006), respectively, for diagnosing effusions due to CHF. The sensitivity and specificity of PF-IMA for CHF at the cut-off concentration of ≥1.0 absorbance units was 90% and 80%, respectively. Its negative predictive value (NPV) was quite high (96%). Positive correlation was found between PF-IMA concentrations and serum-IMA (r = 0.540, P < 0.001).
Conclusions
Measuring IMA concentrations in serum and pleural fluid may be helpful in distinguishing pleural effusion due to a cardiac or non-cardiac aetiology.
Introduction
It is usual to characterize the pleural fluid as an exudate or transudate as a first step in evaluation patients with pleural effusion. Applying Light's criteria is one way of distinguishing between pleural exudates or transudates. However, up to 20–25% of transudates are misidentified as exudates, especially after receiving diuretic therapy and 5% of malignant effusions may be misclassified as transudates. 1,2 Moreover, in 5–25% of patients, the aetiology of pleural effusions is often not determined. 2
Ischaemia-modified albumin (IMA), a Food and Drug Administration-approved serum biomarker of cardiac ischaemia, is a risk stratification tool in patients who are suspected to have acute coronary syndrome. 3 IMA concentrations are measured by a albumin–cobalt binding method in which the property of human albumin is used to bind transition metals. In the presence of ischaemia, the amino or N-terminal of albumin is modified and subsequently affects transition metal binding. 4 Blood levels of IMA rise quickly after the onset of ischaemia (within 5–10 min), and continue to rise while the condition persists and return to normal 12–24 h after ischaemic condition. 5
Serum IMA has been reported to be a biomarker of ischaemia related to myocardial and skeletal muscle ischaemia, acute coronary syndrome, pulmonary embolism (PE), deep venous thrombosis and stroke. 4,6–9 In a previous study, serum IMA concentrations were related with left-ventricular ejection fraction in patients who had had a myocardial infarction treated with percutaneous coronary intervention and developed heart failure. 10 In another study, serum IMA concentrations were found to be correlated with the severity of dilated cardiomyopathy. 11 These previous studies led us to form the hypothesis that serum, and pleural fluid IMA concentrations may increase in pleural effusions developing because of congestive heart failure (CHF).
Methods
A total of 40 serum samples and pleural fluid samples obtained via thoracentesis were used from a collection of 106 samples stored at −80°C, all of which were obtained between December 2008 and August 2009. There were 10 patients with pleural effusions because of CHF, 10 with pneumonia, 10 with acute PE and 10 with malignancy. The study institutional ethics committee (Karadeniz Technical University, Faculty of Medicine, Trabzon, Turkey) approved the study protocol, and all participants gave written informed consent at the time of sampling.
A diagnosis of parapneumonic effusions (PPE) was based upon the presence of an effusion in patients with clinical and radiological evidence of acute pneumonia. In cases with PPE, the decision of chest tube insertion was given according to clinical, radiological and pleural fluid biochemical characteristics of the patients. A PPE was classified as simple (no organisms found in a culture or on a Gram stain), complicated (LDH > 1000 units/L; glucose <2.2 mmol/L; or pH < 7.2) or empyema according to the British Thoracic Society guidelines. 12 In PPE cases, microbiological analysis was also determined for the diagnosis of tuberculosis pleural effusions. It was excluded from the study.
The diagnosis of CHF was made according to clinical symptoms, including history, chest radiography, response to diuretic therapy and a left ventricular ejection fraction of ≤40% measured by echocardiography. All patients in the CHF group were classified as stage III or IV in accordance with the New York Heart Association functional classification system.
A malignant PF was defined by pleural biopsy or the presence of malignant cells on PF cytology. Diagnosis of acute PE was confirmed by spiral chest computed tomography pulmonary angiography in all patients. The diagnosis of PE was confirmed in the presence of at least one filling defect in the pulmonary arterial tree including the subsegmental level. PE cases with pleural effusion having accompanying co-morbidity such as CHF, malignancy and postoperative that may cause effusion were excluded. All patients underwent echocardiography where right and left ventricle end diastolic diameters were measured. Right ventricular dysfunction (RVD) was defined as right ventricle (RV)/left ventricle (LV) > 0.6 and these cases were classified as submassive PE. Patients with massive PE were excluded from the study. 13
Pleural fluid/serum protein ratio and pleural fluid/serum LDH ratio were calculated. Light's criteria were used to differentiate transudates from exudates. In subjects using diuretic therapy, albumin gradient (serum albumin − fluid albumin) was used. Albumin within abnormal serum concentrations –(<3.5, >5.5 mg/dL) was also excluded as it might affect biochemical analyses. The pleural fluid and serum IMA analysis was performed by an experienced technician who was blinded to the diagnosis associated with each fluid.
Biochemical analysis
IMA assay
Reduced cobalt to albumin binding capacity (IMA concentration) was analysed using the rapid and colorimetric method described by Bar-Or et al. 14 Two hundred microlitre of was filled into glass tubes and 50 μL of 0.1% cobalt chloride (Sigma, CoCl2 · 6H2O) in H2O was added. After gentle shaking, the solution was left for 10 min in order to ensure sufficient cobalt albumin binding. Fifty microlitres of dithiothreitol (DTT) (Sigma, 1.5 mg/mL H2O) was added as a colorizing agent and the reaction was quenched two minutes later by adding 1.0 mL of 0.9% NaCl. A colorimetric control was prepared for preoperative and postoperative serum samples. For the colorimetric control samples, 50 μL of distilled water was substituted for 50 μL of 1.5 mg/mL DTT. Specimen absorbencies were analysed at 470 nm using a spectrophotometer (Shimadzu UV1601, Auburn, Australia). The colour of the DTT-containing specimens was compared with that of the colorimetric control tubes. The results were reported as absorbance units (ABSUs).
Statistical analysis
Results are given as mean (median). Means were compared using the Student's t-test or Mann-Whitney's U test. Discrete variables are compared using Fisher exact test (chi-square test). The compatibility of the data with normal distribution was investigated using the Kolmogorov–Smirnov test. One-way analysis of variance (ANOVA) was used for parametric variables and Kruskal–Wallis analysis was used for non-parametric variables. We assessed the correlation between pleural IMA and serum IMA with Pearson rank correlation. Receiver operating characteristic (ROC) analysis was performed to assess the optimal cut-off values of serum and pleural fluid IMA for CHF. All confidence intervals were constructed to have a coverage of 95%. Data were analysed using the SPSS statistical software (version 13.01, serial number 9069728, SPSS Inc, Chicago, IL, USA).
Results
The patient characteristics are summarized in Table 1. The mean age was 64 ± 15 y, and 22 (55%) of the cases were men. The study included 10 patients with pleural effusions due to CHF, 10 cases with pneumonia, 10 with acute PE and 10 cases with malignancy. In-hospital mortality was 10% (4 patients, two patients with PPE, one patient with CHF and one patient with lung cancer).
Characteristics of the patients with pleural effusion
CHF, congestive heart failure; PPE, parapneumonic effusion
Values are given as the mean (standard deviation) and median (minimum-maximum)
*Statistical significant within groups
†Statistical significant within groups
‡‡Statistical significant (CHF group versus all other groups)
Serum creatinine concentrations (>1.2 mg/dL) were higher in five (13%) patients. However, in patients with elevated creatinine, both serum and PF-IMA concentrations were not significantly different than in patients without it (P = 0.57 and P = 0.44, respectively).
In patients with heart failure, the mean ejection fraction was 32 ± 7. Malignant pleural effusion cases were seen in patients with lung adenocancer (n = 6), mesotelioma (n = 1), small cell lung cancer (n = 1), gastric carcinoma (n = 1) and breast carcinoma (n = 1).
The localizations of thrombus in pulmonary emboli were as follows: main pulmonary artery in six cases, lobar pulmonary artery in three cases and segmentary thrombus in one patient. In cases with PE diagnosis, the mean RV/LV ratio was 0.62 ± 0.07 in echocardiography. In three (30%) patients there was RVD on echocardiography. Both serum IMA concentrations and PF-IMA concentrations were not significant in patients with echocardiographic RVD compared with patients without it ([mean 0.64 ± 0.06 ABSU versus mean 0.87 ± 0.16 ABSU; P = 0.99] [mean 0.90 ± 0.33 ABSU versus mean 0.90 ± 0.10 ABSU; P = 0.054]).
PPE were classified as simple in four cases, complicated in four and empyema in two cases. Microorganisms were isolated in pleural effusion of three PPE patients (Escherichia coli, Pseudomonas aeruginosa and Acinetobacter baumanii). Six (60%) cases with PPE underwent tube thoracostomy. In none of the patients with PPE was decortication needed.
In a case with CHF history and pleural effusion classified as exudate, PPE was diagnosed with clinical, radiological and laboratory findings and the effusion of the patient was decreased after antibiotic therapy.
IMA concentrations were measurable in all pleural effusions. The mean pleural fluid IMA concentration was 0.91 ± 0.22 (range: 0.4–1.47). The mean concentration of PF-IMA in CHF was significantly higher than non-cardiac effusions (1.11 ± 0.17 ABSU versus 0.84 ± 0.18 ABSU, respectively, P = 0.002). Moreover, the mean pleural fluid concentration of IMA in patients with CHF was significantly higher than that in patients with PE (P = 0.017), malignant effusions (P = 0.003) and PPE (P < 0.001).
The mean serum IMA concentration was 0.8 ± 0.22 ABSU (range: 0.07–1.26). In CHF, the mean serum IMA concentration was significantly higher than the mean concentration of the other three groups combined (0.79 ± 0.40 ABSU versus 0.75 ± 0.22 ABSU, respectively, P = 0.014). The serum IMA concentration was significant only between pleural effusion due to CHF and PPE (P = 0.023). There was no significant difference between IMA when CHF was compared with malignancy and PE (P = 0.32, P = 0.40, respectively). The distribution of serum and PF-IMA concentrations in CHF and other diseases were seen in Figures 1 and 2.
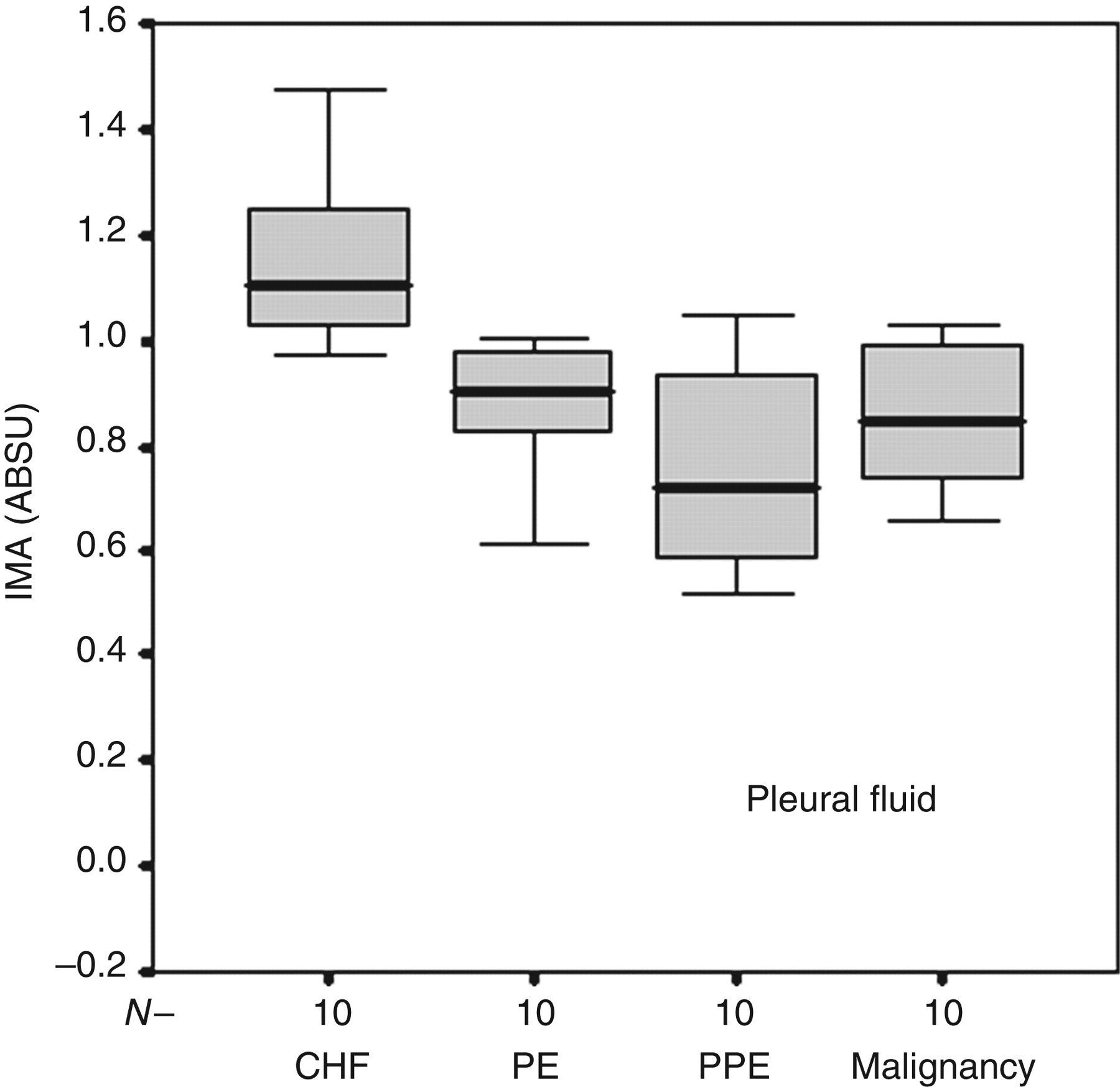
Box-plots showing IMA concentrations in pleural fluid among patients with CHF, PE, malignancy and PPE. IMA, ischaemia-modified albumin; CHF, congestive heart failure; PE, pulmonary embolism; PPE, parapneumonic effusion; ABSU, absorbance unit
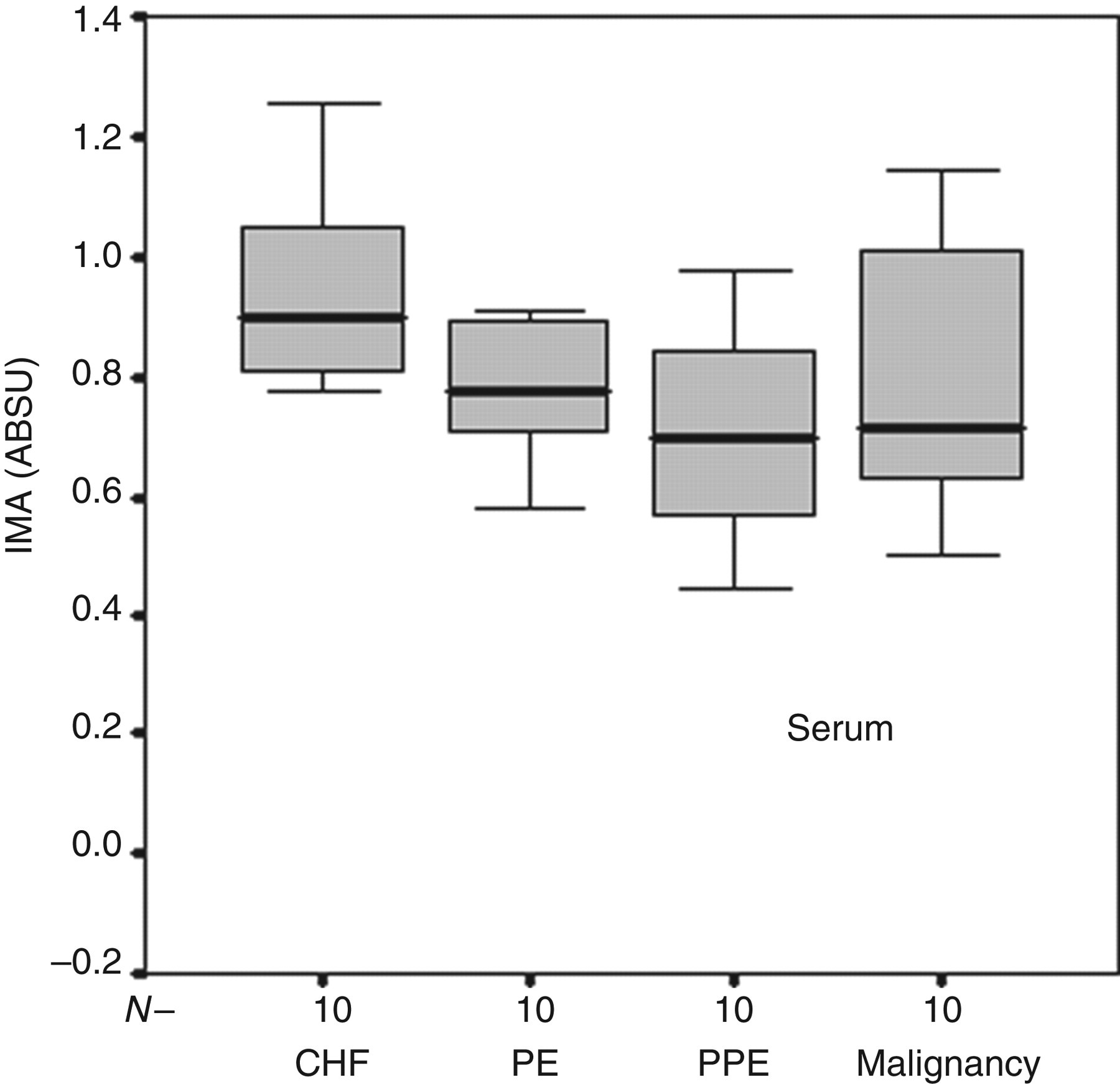
Box-plots showing IMA concentrations in serum among patients with CHF, PE, malignancy and PPE. IMA, ischaemia-modified albumin; CHF, congestive heart failure; PE, pulmonary embolism; PPE, parapneumonic effusion; ABSU, absorbance unit
The mean pleural fluid concentration of IMA in patients with CHF was significantly higher than the serum concentration of IMA in patients with CHF (1.15 ABSU versus 0.94 ABSU, P = 0.009).
While pleural fluid neutrophils percentage was 67% in PPE, it was 39% in malignant effusions (P = 0.031). The pleural fluid protein concentration was significantly low in CHF patients compared with PPE cases (P = 0.003). Also, PF-WBC count and LDH concentrations were detected to be significantly low in cases with CHF than other patient groups (P = 0.004, P < 0.001, respectively) (Table 1).
ROC analysis was used to identify the optimal serum and PF-IMA cut-off value for CHF. The area under the ROC curve was 0.927 (95% CI: 0.844–1.00, P < 0.001) for pleural fluid IMA. The sensitivity and specificity of PF-IMA for CHF at the cut-off level of ≥1.0 ABSU was 90% and 80%, respectively. The area under the ROC curve was 0.792 (95% CI: 0.653–0.930, P = 0.006) serum IMA concentrations for CHF (Figure 3). Serum IMA ≥0.8 ABSU showed high sensitivity and specificity for CHF 80% and 63%, respectively (Table 2).
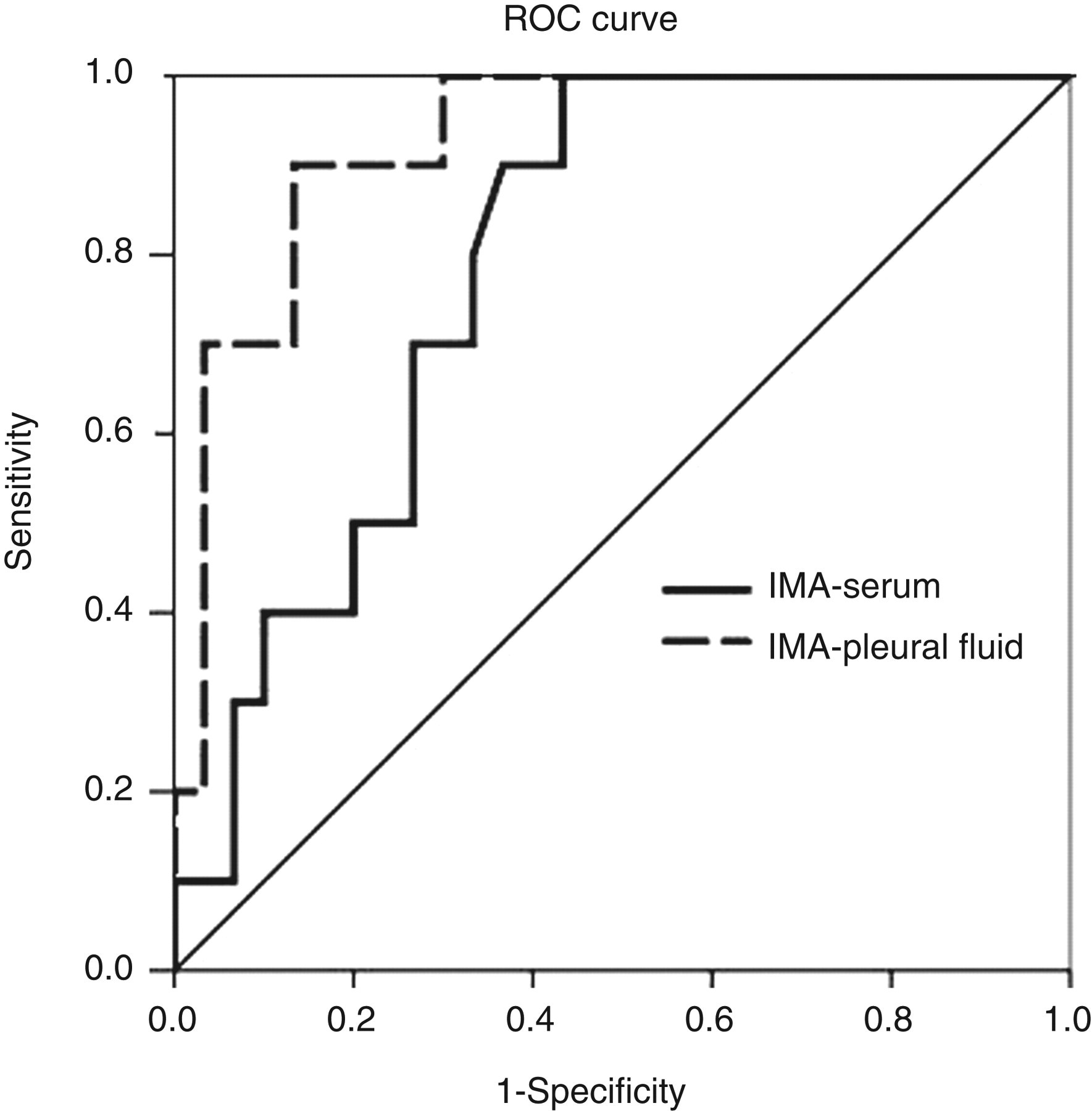
ROC curves of PF-IMA and S-IMA for differentiating CHF from non-CHF. The area under the ROC curve was 0.927 (95% CI: 0.844–1.00, P < 0.001) and 0.792 (95% CI: 0.653–0.930, P = 0.006), respectively. ROC, receiver operating characteristic; PF-IMA, pleural fluid ischaemia-modified albumin; S-IMA, serum ischaemia-modified albumin; CHF, congestive heart failure; CI, confidence interval
Predictive value of ischaemia modified albumin in the cardiac pleural effusion
NPV, negative predictive value; PPV, positive predictive value; IMA, ischaemia-modified albumin; ABSU, absorbance unit
Data are presented with 95% confidence interval in parentheses
Two of the 10 (20%) patients with pleural effusion due to heart failure were falsely classified as exudative by Light's criteria. All of them exhibited PF-IMA concentrations of ≥1.0 ABSU (1.09 and 1.35 ABSU, respectively) and serum IMA concentrations of ≥0.8 ABSU (0.87 and 1.26 ABSU, respectively).
Positive correlation was found between PF-IMA concentrations and serum-IMA (r = 0.540, P < 0.001) (Figure 4).
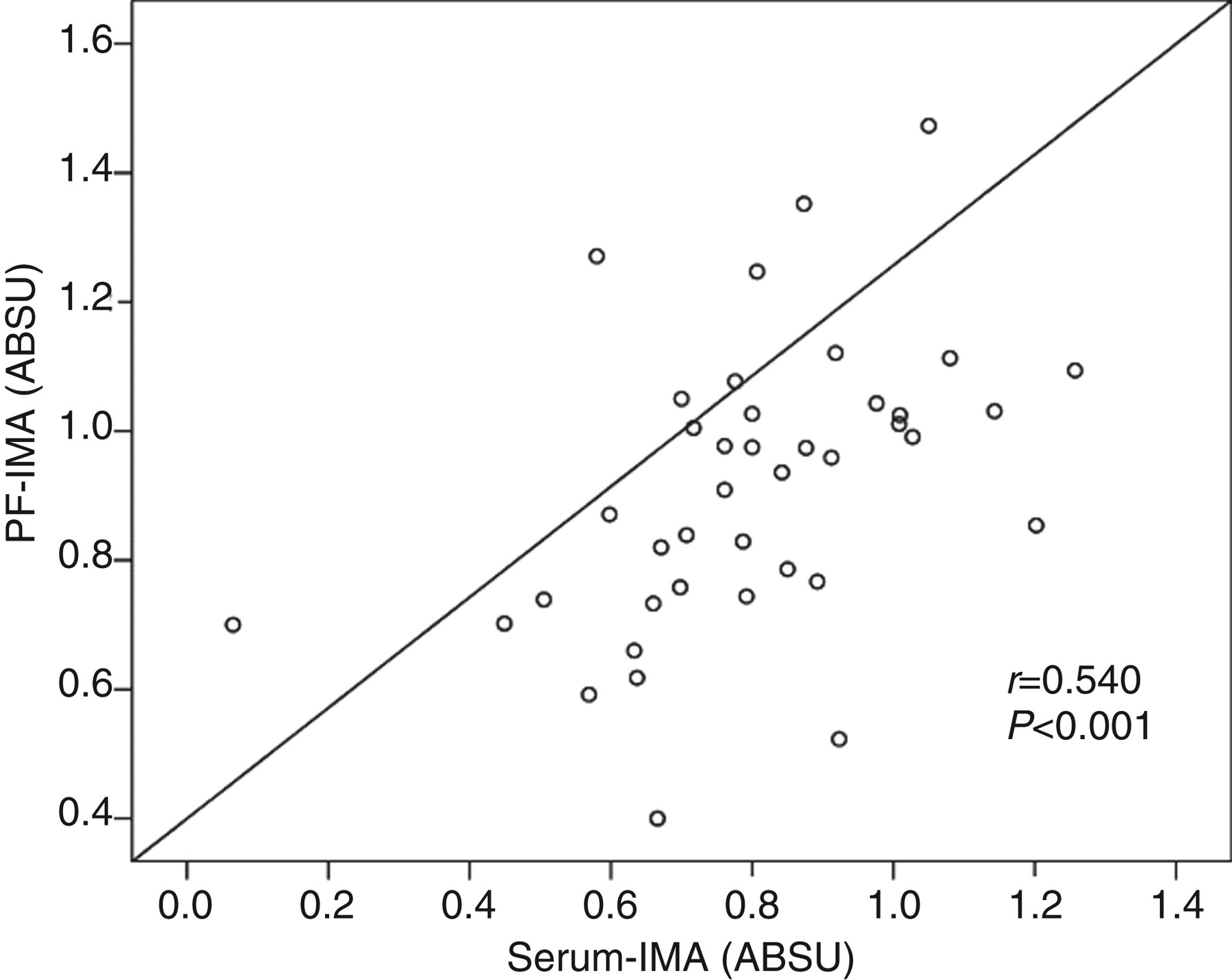
Multiple linear regressions between PF (pleural fluid) ischaemia-modified albumin (IMA) and serum IMA
Discussion
This is the first study to report IMA concentrations in both serum and pleural fluid. Although the number of patients was small, we found that IMA concentrations were higher in both pleural fluid and serum of CHF patients compared with non-cardiac patients. However, when the serum IMA concentrations of CHF cases were compared with the serum concentrations of other cases, there was only significant difference between CHF and PPE. In addition, positive correlation was present between PF-IMA and serum-IMA concentrations (r = 0.540, P < 0.001).
In this study, pleural fluid IMA concentrations was more valuable than serum IMA concentration in distinguishing cardiac effusions and non-cardiac effusions with ROC curves indicating a sensitivity of 90% and a specificity of 80% for the determination of pleural effusions due to CHF. Otherwise, specificity, sensitivity and negative predictive value (NPV) of IMA serum concentration was lower than pleural fluid IMA. The specificity, sensitivity and NPV of serum IMA ≥0.8 level for cardiac effusion were 63%, 80% and 90%, respectively. Although Light's criteria exhibited high value in the exclusion of exudates, the finding of transudate indicated a low sensitivity of 64% in the determination of cardiac effusions. 15
The origin of IMA in pleural fluid is unclear, although it has been suggested that it might diffuse easily into the pleural space because of its small molecular size, as with NT-proBNP. 16 In contrast, the concentrations of most cytokines are lower in pleural fluid. However, pleural IMA concentrations higher than serum IMA may be related to some cytokines produced by pleura due to pleural involvement. 17 In addition, serum IMA concentrations may be affected from conditions such as exercise, serum albumin concentrations and renal failure. 18 However, the IMA results were not affected by either the concentration of albumin or the concentration of creatinine. All these demonstrate that as in our study measurement of these markers in pleural effusion may directly show the underlying disease and more objective results could be derived.
Brain natriuretic peptide (BNP), N-terminal pro-brain natriuretic peptide (NT-proBNP) and ST2 (interleukin-1 receptor family member), which are myocyte stress biomarkers, have been investigated in the diagnosis of pleural effusions previously. 19–22 Both serum and pleural fluid concentrations of NT-proBNP were found to be significantly higher in pleural effusions developed because of CHF than the effusions that occur owing to other causes. 19,20 In previous studies, in distinguishing cardiac effusion from non-cardiac effusion, the pleural level of NT-proBNP was found as different threshold concentrations and its specificity and sensitivity were 87–100% and 92–100%, respectively. 19–22 In this study, while pleural fluid IMA level sensitivity was parallel to NT-proBNP, its specificity was lower. Thus, these two markers are released into the circulation similarly. On the other hand, NT-proBNP has a longer half-life of approximately 1–2 h, leading to higher circulating levels and also has much variable clinical range (0–3500 pg/mL). 23 However, IMA concentrations increase in blood in 5–10 min after injury and return to normal levels within a short time. 5 Additionally, IMA measurement is an inexpensive and simple technique. All these are important factors for a biomarker to be useful clinically. 24 Therefore, serum IMA concentrations may be better in the diagnosis of pleural effusions in cases with CHF.
CHF was also present in 10–15% of cases with PE and in this situation mortality increases 2–8-fold. 25,26 It is, however, difficult to diagnose PE in patients with CHF. Given its sensitivity and specificity, serum IMA may have diagnostic value in patients with minimal pleural effusion where thoracenthesis is not indicated. Also, in this study, according to Light's criteria, two (20%) patients with CHF were misclassified false-positively as exudative effusion. However, both serum and PF-IMA concentrations were found to be higher than the determined cut-off concentrations.
There are several limitations to this study. Firstly, this is a pilot study to examine the diagnostic value of IMA concentrations to distinguish cardiac and non-cardiac pleural effusions and more studies would be needed to establish firm cut-off points. We were also not able to include any transudative pleural effusions of non-cardiac origin, such as effusions associated with hepatic or nephrotic syndrome. Investigating the diagnostic utility of IMA concentration in this patient group would be of particular interest, because Light's criteria are not able to separate them. Another limitation of our study is that we have no other echocardiographic data (e.g. such as left ventricular thickness or end-systolic diameter) regarding patients with CHF. Finally, in CHF diuretic treatment response might be followed by the serum IMA concentration. However, this approach may be addressed by further studies.
In conclusion, in IMA concentrations in serum and PF may be helpful in distinguishing cardiac and non-cardiac pleural. However, this approach should be confirmed in further prospective trials.
DECLARATIONS
