Abstract
Background
The introduction of a second trimester quadruple test for fetal Down's syndrome adds the measurement of serum inhibin A (InhA) and unconjugated oestriol (UE3) to the existing repertoire of alphafetoprotein and intact human chorionic gonadotrophin. The aim of this study was to assess the stability of InhA and UE3 in whole blood and serum.
Methods
To determine whole blood stability, five extra blood specimens were obtained from each of 10 women attending an antenatal clinic. Samples were stored at room temperature for either two hours, one, three, five or seven days and centrifuged prior to analysis. Serum stability was studied by the analysis of surplus serum from 14 routine second trimester screening samples: seven stored at room temperature and seven stored at 4°C. An aliquot from each specimen was analysed two hours, one, three, five or seven days post centrifugation. Specimens were analysed for InhA and UE3 using the Beckman Access 2® Immunoassay analyser.
Results
No significant difference (P > 0.05) was shown in InhA or UE3 concentrations between the initial time point on the day of venepuncture and each of the subsequent analyses at one, three, five and seven days following collection for either whole blood or serum.
Conclusions
InhA and UE3 are stable in whole blood and serum for seven days.
Introduction
To improve screening efficiency for fetal Down's syndrome, the UK National Screening Committee has published recommended targets for detection rate (90%) and false-positive rate (2%) with a benchmark time frame of April 2010. The quadruple test will not achieve this, but it is recommended for women who present too late for first trimester combined screening. 1,2 The second trimester quadruple test incorporates the measurement of two additional serum markers, inhibin A (InhA) and unconjugated oestriol (UE3) to the existing screen which uses alphafetoprotein (AFP) and intact human chorionic gonadotrophin (HcG).
Secreted by ovarian granulosa cells, the corpus luteum and the placenta during pregnancy, InhA is a dimeric glycoprotein which acts to inhibit pituitary secretion of follicle-stimulating hormone. UE3 is a steroid hormone produced from the fetal precursor 16α-hydroxy dehydroepiandrosterone sulphate and a marker of placental integrity. An increased concentration of InhA and a decreased concentration of UE3 in maternal serum are associated with an increased risk of fetal Down's syndrome. 3
To avoid any storage-dependent artefactual changes in marker concentration affecting the calculated risk results, it is essential to know the stability of InhA and UE3 in both whole blood and serum prior to their introduction to the screening panel. Because the existing literature has focused solely on serum stability, 4 this study will enable an evidence-based maximum sample transit time to be determined for whole blood samples. Furthermore, it will also confirm maximum storage time and conditions for serum samples that are held in the laboratory prior to analysis by immunoassay methods used routinely in the screening programme. The aim of this study was therefore to determine the stability of both InhA and UE3 in whole blood and serum.
Subjects and methods
Whole blood study
Following ethical approval from the Fife and Forth Valley Research Ethics Committee, 10 women attending the antenatal clinic at Forth Park Hospital, Fife, and consenting to second trimester antenatal screening, were recruited to participate in the study. In addition to their routine antenatal screening sample, a series of five extra specimens of venous blood were taken by conventional venepuncture from each of the 10 subjects. The specimens were sampled into gel serum separator tubes (SSTs) (BD Diagnostics, Plymouth, UK). These whole blood samples were stored at room temperature (22°C) for either two hours, one, three, five or seven days and then centrifuged at 3500
Serum study
Following analysis of HCG and AFP for routine second trimester screening, surplus serum was stored in SSTs from 14 subjects: seven were stored at room temperature and seven stored at 4°C. All specimens had been separated by centrifugation within two hours of venepuncture. An aliquot from each specimen was analysed two hours, one, three, five and seven days post centrifugation.
Sample analysis
Each aliquot was analysed in triplicate for InhA and UE3 concurrently with commercial quality control material on the Beckman Access 2® Immunoassay analyser. The InhA assay is a two-step sandwich immunoassay employing a chemiluminescent signal system and paramagnetic particles to achieve phase separation. The UE3 assay is a competitive immunoassay employing a similar chemiluminescent signal system with solid-phase separation.
Statistics
The stability of both InhA and UE3 in whole blood and serum was investigated statistically using the mean result of each triplicate analysis. A one-way analysis of variance followed by Dunnett's post hoc analysis was used to compare whole blood and serum InhA and UE3 concentrations at the initial time point with subsequent time points to determine the stability of the analytes in each matrix. All statistical analyses were carried out using the GraphPad Software Inc, version 5, (San Diego, CA, USA).
Results
The concentration of InhA and UE3 in each specimen measured at each time point is shown in Figures 1a and b, respectively, for the whole blood stability study.
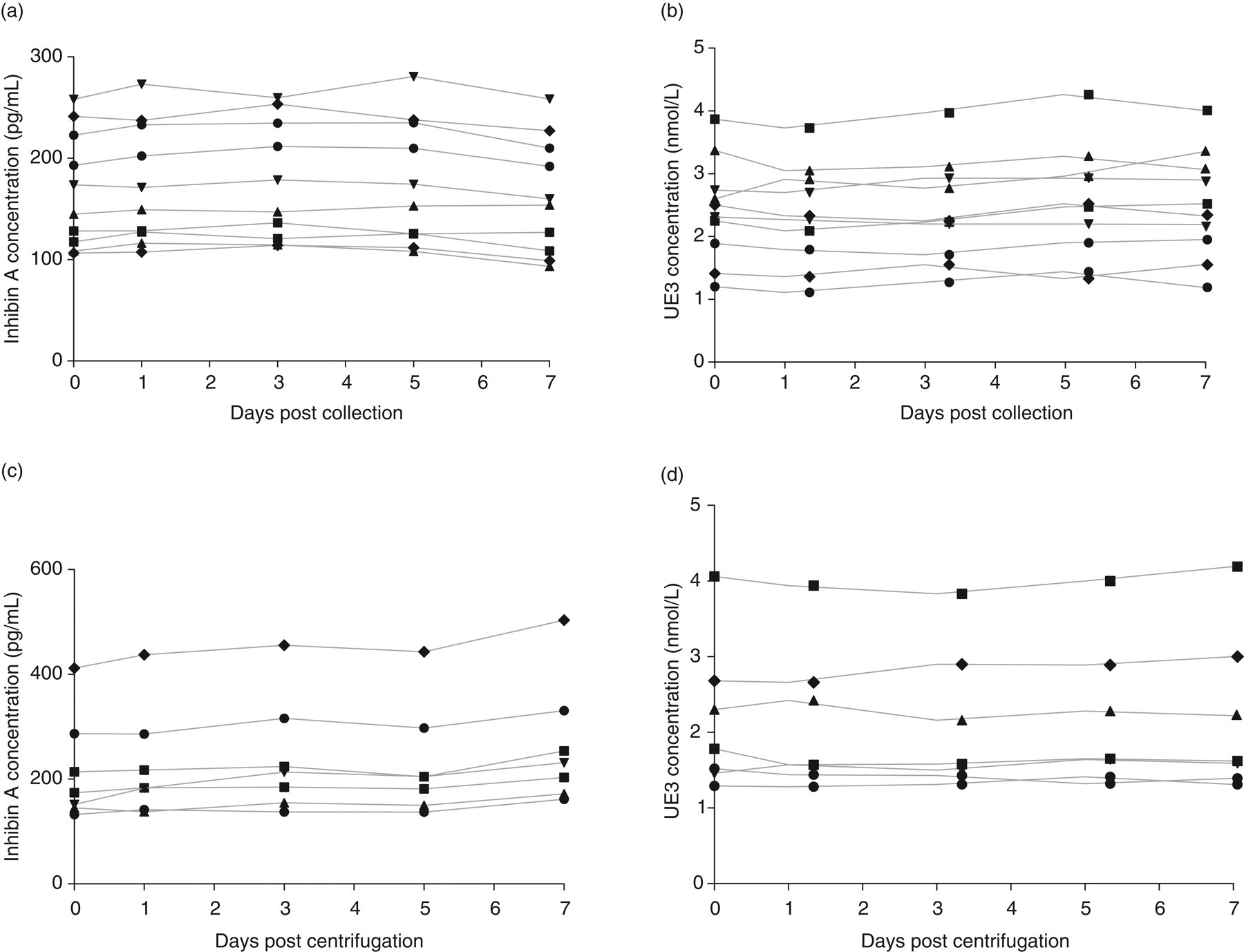
Concentration of (a) inhibin A and (b) unconjugated oestriol (UE3) in whole blood from 10 subjects analysed on the day of collection, and one, three, five and seven days post venepuncture stored at room temperature. Concentration of (c) inhibin A and (d) UE3 in seven serum specimens stored at room temperature analysed on the day of collection, and one, three, five and seven days post centrifugation
No significant difference (P > 0.05) was found in the concentration of these analytes between the initial time point on the day of collection and each of the other time points tested, demonstrating stability in whole blood for a period of seven days.
The results of the serum stability study at room temperature for InhA and UE3 are shown in Figures 1c and d, respectively.
Similarly, the serum specimens stored at room temperature demonstrated no significant difference in concentration of InhA or UE3 between the initial time point on the day of collection and any other time point tested post centrifugation. These results demonstrate the stability of both analytes in serum for a period of seven days. Although not statistically significant, a trend towards higher mean InhA concentration following storage of serum for seven days at room temperature was noted. This trend was not apparent in the serum specimens stored at 4°C, although the results are not presented here.
Discussion
The demonstrated stability of InhA and UE3 in whole blood for at least seven days at room temperature allows an evidence-based cut-off to be set for the acceptance of second trimester screening samples for analysis. However, it is clearly preferable that specimens reach the laboratory for analysis promptly following collection; this is particularly important in the context of screening, given the possibility that diagnostic testing may be required following a high-risk screening result. Furthermore, the demonstrated stability of both markers in serum excludes the need for immediate analysis of InhA and UE3 following centrifugation of blood samples; we would, however, advocate sample storage at 4°C following centrifugation to avoid any artefactual rise in InhA concentration at room temperature as demonstrated in our study. The results also allow storage of serum samples with confidence for up to seven days at 4°C following separation if analysis is delayed for exceptional reasons, such as analyser downtime. These results are in agreement with previous serum stability studies that showed InhA to be stable for nine days; however, UE3 was found to increase from seven days onwards. 4 This study used a different immunoassay platform and studied only refrigerated serum samples. Our study has shown both analytes to be stable for seven days in whole blood at room temperature and serum at both room temperature and 4°C.
With the recent trend across the UK towards rationalization of laboratory services for fetal Down's syndrome screening, samples are more frequently required to be transported across greater distances. Assuming transit temperatures of up to 22°C this study provides data on maximum transit times and storage conditions. In our laboratory, by setting the maximum transit time at five days post-venepuncture, we ensure there is no deterioration in sample integrity that would impact on the calculated risk result. A five-day limit would not, however, be appropriate for laboratories using free beta-HCG as a second trimester marker where significantly shorter transit times have been recommended for whole blood samples. 5
DECLARATIONS
