Abstract
Background
Measurement of metadrenalines has been recommended in the investigation of phaeochromocytoma. Urinary assays remain the most common; however, drug interference is still one of the main challenges for analytical systems. We have developed a liquid chromatography–tandem mass spectrometry (LC–MS/MS) method for the quantification of total urinary normetadrenaline and total urinary metadrenaline, which does not require extraction procedures prior to analysis.
Method
Total urinary normetadrenaline and total urinary metadrenaline were individually quantified by electrospray ionization in the multiple-reaction monitoring mode. Deuterated internal standards were used and an acid hydrolysis step was used to convert conjugated metabolites into free metadrenalines. Chromatographic separation was achieved using a Phenomenex Luna 3μ PFP column followed by analysis on an API 3200 LC–MS/MS.
Results
Linearity was exhibited across the calibration range for both normetadrenaline (r = 1, P < 0.0001) and metadrenaline (r = 1, P < 0.0001) with the limit of quantification of 0.05 and 0.02 μmol/L, respectively. Intra-assay imprecision for both normetadrenaline and metadrenaline was less than 5.5% with % coefficient of variations of less than 4%. Inter-assay imprecision was less than 13%. Neither noradrenaline or adrenaline interfere with the assay as determined by the spiking of samples with high concentrations of noradrenaline or adrenaline (P > 0.05). Acceptable analytical performance was seen with comparison to a high-performance liquid chromatography method and on External Quality Assessment returns.
Conclusions
An analytically simple and sensitive method has been developed and evaluated for the analysis of total urinary normetadrenaline and total urinary metadrenaline which is now in routine use.
Introduction
Phaeochromocytomas are neuroendocrine tumours that typically arise from catecholamine-producing chromaffin cells in the adrenal glands. Approximately 80–85% of phaeochromocytomas arise from the adrenal medulla. 1 Clinical symptoms are largely due to the action of excess catecholamine secretion, which has provided the basis for biochemical assays for the diagnosis of phaeochromocytoma. Traditional methods use the measurement of the catecholamines – noradrenaline (norepinephrine), adrenaline (epinephrine) and dopamine, all of which are derived from tyrosine. The catecholamines are metabolized to normetadrenaline and metadrenaline by two mechanisms – catechol-o-methylation, using catechol-O-methyl transferase (COMT) and deamination by mitochondrial monoamine oxidase. COMT methylates the C-3 hydroxyl position of the benzene ring forming normetadrenaline, metadrenaline and 3-methoxytyramine from noradrenaline, adrenaline and dopamine, respectively. These are subsequently converted to vanillymandelic acid. Metadrenalines (normetadrenline and metadrenaline) exist in both free and conjugated forms. Total normetadrenaline is used here to mean the sum of free and conjugated normetadrenaline and total metadrenaline is used here to mean the sum of free and conjugated metadrenaline.
The diagnosis of phaeochromocytoma requires biochemical evidence of inappropriate catecholamine production, which should be undertaken before imaging studies. There has been much debate over the years for the preferred biochemical method for the diagnosis of phaeochromocytoma. The measurement of metadrenalines has been shown to have a higher diagnostic sensitivity than catecholamines but a lower diagnostic specificity. It was recommended from the First International Symposium on Pheochromocytoma held in October 2005 at Bethesda, MD, USA, that ‘… initial testing for phaeochromocytoma should include measurements of fractionated metadrenalines in urine or plasma, or both, as available …’. No consensus was reached whether plasma or urine measurements should be the preferred test. 2 However, analytical and sample collection issues for plasma samples preclude this as a viable routine assay at the current time. Metadrenalines have a better diagnostic sensitivity due to their continuous production within phaeochromocytomas, which is a process that occurs independently of catecholamine secretion by the tumour. Also, the COMT enzyme, which metabolizes catecholamines to free metadrenalines, is present in high concentrations in phaeochromocytomas. 3 However, there are still limitations on the use of measurements of metadrenalines, these include patients with very small tumours and those with tumours that do not produce noradrenaline and adrenaline, i.e. dopamine secreting. 4
Metadrenalines are excreted in urine as both free amines and conjugated forms. The conjugated forms are mainly sulphate and glucoronide conjugates. Less than 3% of the total urinary normetadrenaline and metadrenaline excretion is normally due to the free unconjugated forms; 5 however, in patients with phaeochromocytoma the urinary output of metadrenalines is increased, with a disproportionate increase in normetadrenaline to metadrenaline free unconjugated forms. 6 The method reported here uses acid hydrolysis to allow total urinary normetadrenaline and total urinary metadrenaline to be assayed.
The routine measurement of total urinary normetadrenaline and total urinary metadrenaline is currently being undertaken by a number of laboratories using mainly high-performance liquid chromatography (HPLC) or immunoassays. The detection system on HPLC systems is usually an electrochemical detector, and the total normetadrenaline and total metadrenaline assay can sometimes be coupled with an assay for catecholamines as the analytes elute at different retention times. 3 The main limitation of HPLC assays are that they are prone to interference from the in vivo and in vitro affects of drugs and their metabolites, 7–9 with a high variability between analytical methods. In vitro interference is seen by the co-elution of other substances (for example drugs or their metabolites) with the analyte of interest, which causes difficulties in integration of the required peak on the chromatographic trace. Though sample preparation is usually minimal, the analysis time (which can be in the order of approximately 45 min) can limit the sample throughput. Good chromatography is required for the efficient separation of all peaks, which can require technical expertise to trouble shoot analytical problems.
Immunoassays can be automated and allow an increased number of samples to be analysed, but usually significant sample pretreatment is required, which though can be automated adds to the analysis time. Incubation and washing steps also need to be taken into consideration. Immunoassays also lack the use of an internal standard to monitor the recovery through the extraction process, and they can also be prone to non-specific binding that may cause false-positive results.
Mass spectrometry is increasingly being utilized within the clinical biochemistry laboratory as this technique when coupled with liquid chromatography (LC) increases the analytical specificity for analytes. Tandem mass spectrometry (MS/MS) can be used to further enhance this specificity and multiple reaction monitoring (MRM) allows the quantification of multiple molecules at the same time. This allows both normetadrenaline and metadrenaline to be measured and quantified simultaneously.
LC–MS/MS methods are available for the measurement of both urine and plasma metadrenalines, 10,11 but usually require sample processing prior to analysis, for example by solid phase extraction. The method reported here is novel in that it uses an acid hydrolysis step to convert conjugated metabolites into free metadrenalines and does not require the use of solid phase extraction prior to sample analysis. 11 The main disadvantage with the use of LC–MS/MS is that a large capital expenditure may be required for the procurement of the instrumentation; however, as more laboratories obtain access to LC–MS/MS equipment for use with other assays, this will no longer be a problem and once in place the assay has minimal costs associated with routine use. LC–MS/MS methods allow an increased sample throughput due to the ease of automation both in the sample preparation and in the analysis (use of an automated pipetting work station to load onto a 96-well microtitre plate). Analysis time is usually significantly decreased as the quality of chromatography is not so important. Analytes are identified by the mass to charge ratio of the parent molecular ion and the daughter ion; therefore, separation of specific analytes from other components is not an issue as long as the molecular structures are different (these will not be detected). In the case of metadrenalines this is an issue for adrenaline and normetadrenaline as they are isobaric and produce the same fragmentation patterns when undergoing MS/MS. This is overcome by ensuring adequate separation of the two compounds by liquid chromatography.
Materials and methods
Chemicals and reagents
A single calibrator standard (Chromsystems, München, Germany) was used, which contained normetadrenaline and metadrenaline in concentrations of 4.020 and 1.459 μmol/L, respectively, when reconstituted with the required volume of 0.1 mol/L hydrochloric acid (HCl).
Instrumentation
Analysis was performed using a Shimadzu HPLC system with an auto sampler that allows both 96-well microtitre plates as well as individual glass vials, with the detection system being an API 3200 QTrap® tandem mass spectrometer (Applied Biosystems, Warrington, UK) using an electrospray-ionization ion source. The software used was Analyst version 1.4.1.
LC–MS/MS conditions
The chromatographic separation of total normetadrenaline and total metadrenaline was achieved using a Luna 3μ PFP (pentafluorophenyl propyl ligand bonded to luna silica) column (150 × 2.00 mm) with PFP 4 × 2.0 mm security guard cartridges (Phenomenex, Macclesfield, UK). An isocratic system was used with the mobile phase consisting of 97.5% 10 mmol/L ammonium formate pH 3.5 and 2.5% methanol, switching to 100% methanol for one minute between sample injections to remove waste products from the column. Two minutes was allowed for the column to re-equilibrate in mobile phase after the methanol wash, giving a total of 12 min between sample injections. The flow rate was 0.2 mL/min and 20 μL of sample was injected onto the column. These conditions allowed complete separation of total normetadrenaline and total metadrenaline from the catecholamines noradrenaline, adrenaline and dopamine even though they were not being quantified using this assay.
Ionization of the metadrenalines was achieved using electrospray ionization in positive ion mode. The temperature of the source was 550°C. Multiple-reaction monitoring was used with the transitions of precursor ions to product ions as detailed in Table 1.
Parameters for mass spectrometry for metanephrine analysis
DP, declustering potential; EP, entrance potential; CEP, collision cell entrance potential; CE, collision energy; CXP, collision cell exit potential
Sample preparation
Twenty-four-hour urine collections into HCl acidified collection bottles were used for this analysis. An aliquot was taken and centrifuged at 800
Assay performance
Intra- and inter-assay imprecision was determined using 20 and 40 aliquots of urine controls, respectively, at two different concentrations. Linearity studies were performed using dilutions of a patient sample with 0.1 mol/L HCl prior to acid hydrolysis. Recovery, relative to the internal standard, was determined by spiking urine samples with solutions of normetadrenaline (511 μmol/L) and metadrenaline (528 μmol/L) (Sigma-Aldrich), concentration determined by mass spectrometry. Each sample was prepared in duplicate, with duplicate paired samples where the spike was replaced by 0.1 mol/L HCl for determination of the base concentration. Assay interference by noradrenaline and adrenaline was investigated by the spiking of a patient sample with a high concentration of noradrenaline and adrenaline. Noradrenaline and adrenaline were both purchased from Sigma-Aldrich; stock solutions were prepared in 0.1 mol/L HCl and were spiked into urine samples at 5000 and 2500 μmol/L, respectively. A baseline urine sample was prepared diluted with 0.1 mol/L HCl to take into account the dilutions by addition of noradrenaline and adrenaline. The baseline urine, noradrenaline spiked urine and adrenaline spiked urine were each prepared 10 times.
Ion suppression was investigated by a postcolumn infusion of a solution containing approximately 5000 μmol/L normetadrenaline and approximately 5000 μmol/L metadrenaline at 50 μL/min into the mass spectrometer, and injecting a hydrolysed urine sample without internal standard. Limit of detection was determined by serial dilution of a patient sample and signal:noise (S:N) ratio of the peak versus the baseline was determined. The minimum concentration of metadrenaline and normetadrenaline that gave S:N ratios consistently >5 was chosen as the limit of detection.
Method comparison
Comparative data were collected using 50 patient samples, which were analysed using a Chromsystems reagent kit (Chromsystems) on an HPLC with electrochemical detection (ECD). These samples were analysed according to the kit instructions, which allowed for quantification of total normetadrenaline and total metadrenaline after acid hydrolysis (the same as the LC–MS/MS method). IQC samples were analysed on each batch and were within acceptable limits and the laboratory that analysed the samples participates in an external quality assurance scheme with good performance. The same 50 patient samples were analysed over a similar time frame by the LC–MS/MS method described in this paper; therefore, the integrity of the samples was the same for both assays.
Results
LCMS method
Mechanism of MRM transition
Figure 1 shows the electrospray product ion scan in positive-ion mode by infusion of approximately 5000 μmol/L normetadrenaline and approximately 5000 μmol/L metadrenaline. The transitions monitored are detailed in Table 1. In source fragmentation causes water loss (–18) from the parent transition ion, this is then fragmented further to the daughter ion by loss of a protonated methoxy group (–HOCH3) (Figure 2).
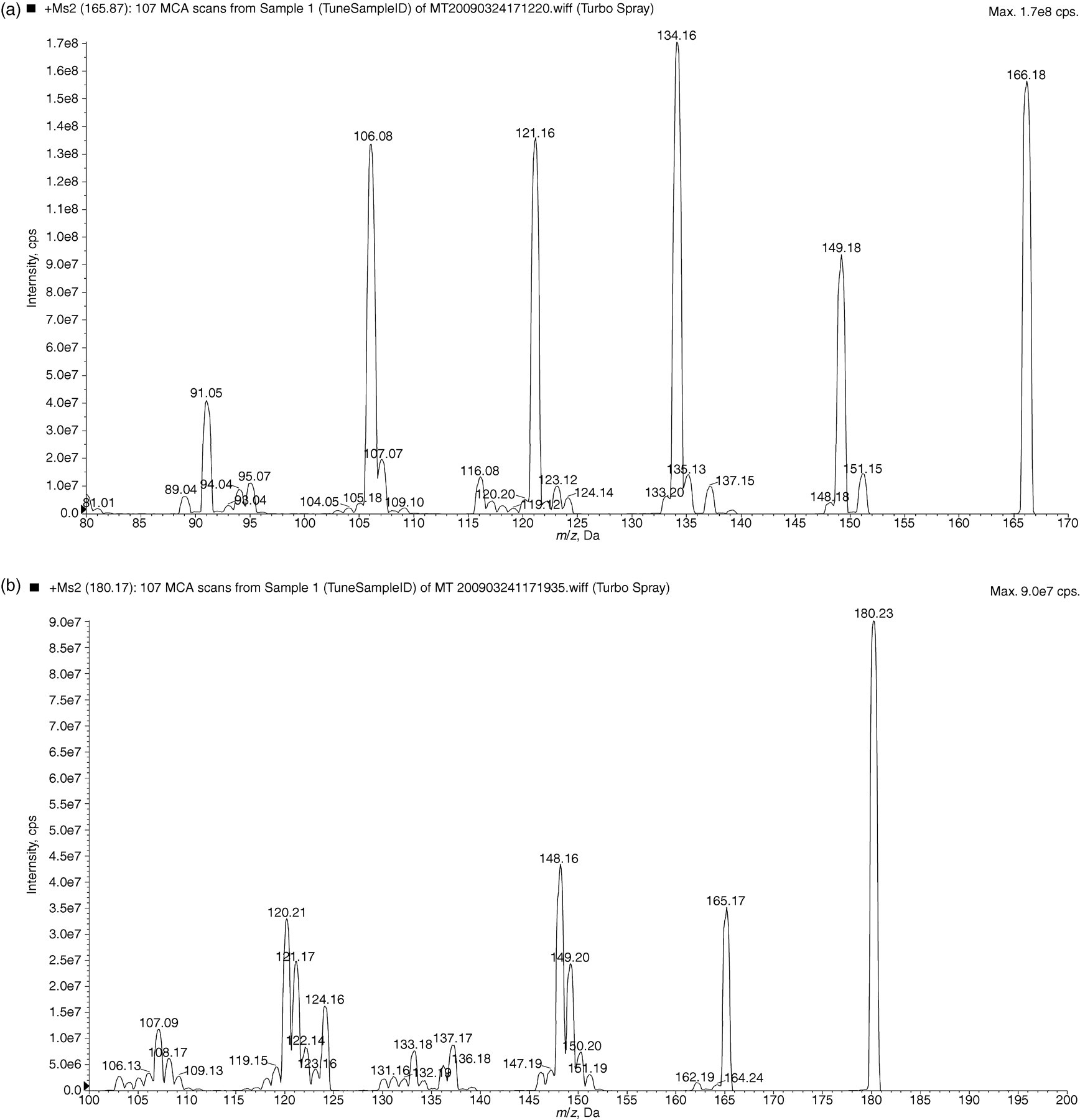
Product ion mass spectrum for (a) normetadrenaline and (b) metadrenaline
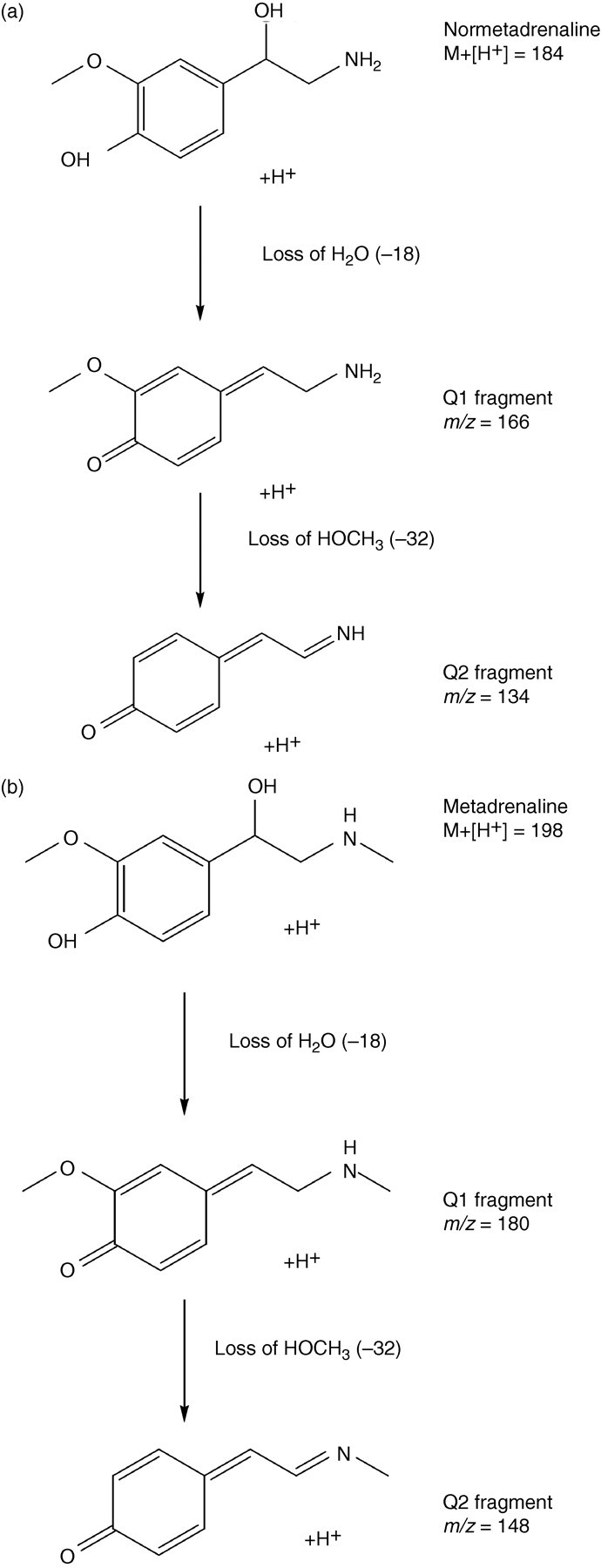
Mechanism of MRM transition of (a) normetadrenaline and (b) metadrenaline, MRM, multiple reaction monitoring
Ion suppression
Ion suppression was seen between 1 and 10 min for both total normetadrenaline and total metadrenaline (Figure 3). Normetadrenaline elutes between 4.0 and 4.5 min and metadrenaline elutes between 7.0 and 7.5 min. An internal standard is used for both normetadrenaline and metadrenaline so this would take into account any ion suppression effects, and the sensitivity is not affected at the concentrations that are required. The method was modified to include a switch to waste between 0 and 2.5 min, which reduced the ion suppression for both normetadrenaline and metadrenaline (data not shown).
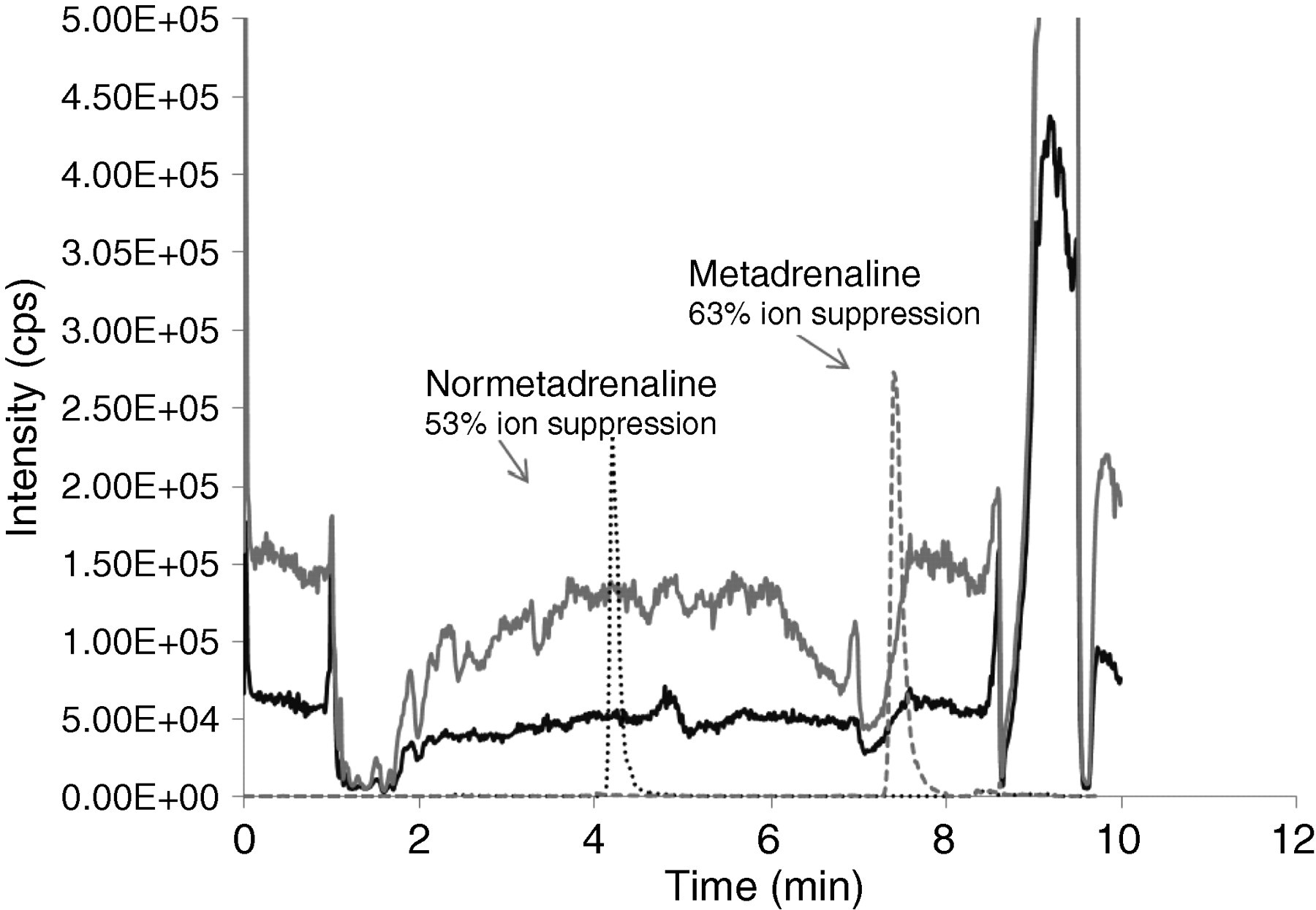
Ion suppression data. Solid black line – normetadrenaline infusion and patient sample injection; solid grey line – metadrenaline infusion and patient sample injection; dashed black line – normetadrenaline; and dashed grey line – metadrenaline
Imprecision, linearity and recovery
Calibration curves and IQC were run with every batch of specimens. A one-point calibration was carried out in duplicate at the start and end of a batch, and both normal and abnormal IQC were analysed at the start and end of a batch. All calibrations were included in result analysis. The IQC material was used to determine both intra- and inter-assay imprecision levels. The intra-assay imprecision for both total normetadrenaline and total metadrenaline was less than 5.5% in all cases, with % coefficient of variation (%CVs) of less than 4% in the abnormal concentration range. The %CVs were slightly increased on the inter-assay imprecision but were less than 10% (Table 2). The assay showed linearity upon dilution of a patient sample for both total normetadrenaline and total metadrenaline (Figure 4). Pearson correlation was carried out to show a statistical assessment of linearity: total normetadrenaline (r = 1.00, P < 0.0001, 95% CI 0.99–1.00) and total metadrenaline (r = 1.00, P < 0.0001, 95% CI 0.98–1.00). The lower limit of quantification for total normetadrenaline was 0.05 μmol/L and total metadrenaline 0.02 μmol/L. A recovery experiment was carried out over four concentrations for both total normetadrenaline and total metadrenaline (Table 3), which showed the average recovery for total normetadrenaline to be 100% and for total metadrenaline an average of 92% recovery.
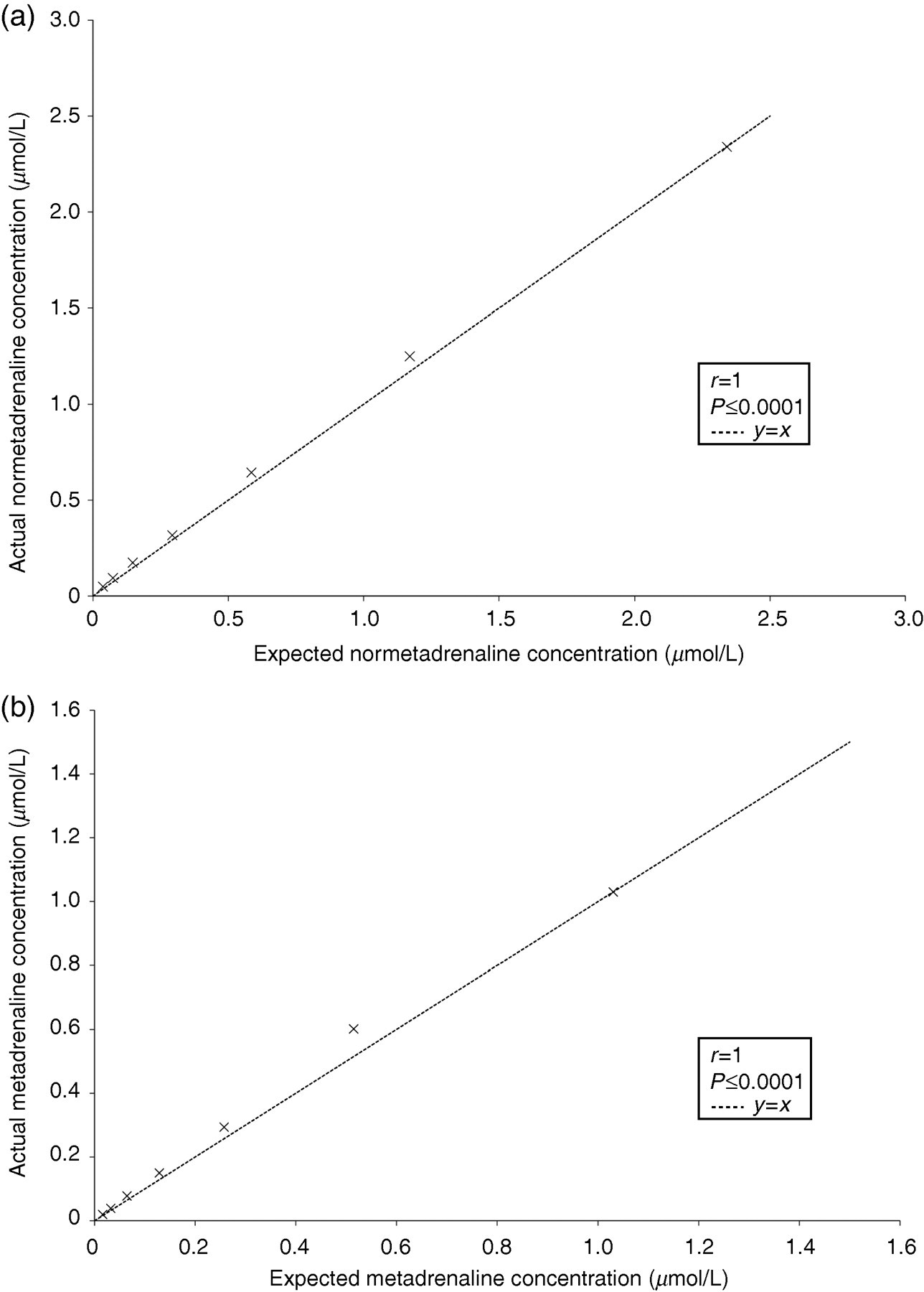
Linearity of a patient sample for (a) normetadrenaline and (b) metadrenaline for the liquid chromatography–tandem mass spectrometry urinary metadrenalines method
Imprecision data for the analysis of total urinary normetadrenaline and total urinary metadrenaline by LC–MS/MS
LC–MS/MS, liquid chromatography–tandem mass spectrometry; %CV, % coefficient of variation
Recovery characteristics of total urinary normetadrenaline and total urinary metadrenaline by LC–MS/MS
LC–MS/MS, liquid chromatography–tandem mass spectrometry
Samples were prepared in duplicate and the mean was taken
Interferences
This method allows complete separation of normetadrenaline and metadrenaline from the catecholamines. Interference from noradrenaline and adrenaline was investigated using high concentration of both of the catecholamines. Urine samples were spiked with noradrenaline (5000 μmol/L) and adrenaline (2500 μmol/L). The normetadrenaline and metadrenaline concentrations in these samples were compared with unspiked values (baseline urine was diluted with 0.1 mol/L HCl to take into account the dilution factor of diluting with noradrenaline and adrenaline). Ten repeat preparations of all samples were made. A t-test was applied, and the results are shown in Table 4. Interference by noradrenaline showed P = 0.9946 (normetadrenaline) and P = 0.1868 (metadrenaline) and for adrenaline P = 0.2825 (normetadrenaline) and P = 0.9176 (metadrenaline). This shows that neither noradrenaline nor adrenaline interfere with the measurement of normetadrenaline or metadrenaline. In vitro interferents such as drugs have not been investigated; however, this method has been running routinely for over a year and no chromatographic interference has been observed. In vivo drug interference will still be affected.
Assay interference of noradrenaline and adrenaline on total urinary normetadrenaline and total urinary metadrenaline measurements
Baseline sample was diluted with 0.1 mol/L HCl to take into account the dilution factor from the spike. Ten repeat samples for each specimen were prepared. Statistical significance was determined using the t-test
Method comparison
Fifty patient samples were analysed by both the LC–MS/MS method and an HPLC-ECD method. Both assays quantified total normetadrenaline and total metadrenaline. Only data from 48 of the 50 samples analysed by HPLC-ECD could be included in the method comparisons due to assay interference in two of the samples which affected the peak height of the internal standard therefore prohibiting the reporting of any results on these samples. These samples were re-analysed and the same characteristic was seen and is likely to be caused by a drug interference co-eluting at the same retention time as the internal standard. (This potential drug interference was not seen on the LC–MS/MS method.)
The two methods were assessed for statistical significance using the Bland-Altman method (Figure 5): normetadrenaline (P = 0.0030, 95% CI −0.3466 to −0.0752) and metadrenaline (P = 0.0006, 95% CI −0.2227 to −0.0663). This showed a slight positive bias for the LC–MS/MS method compared with the HPLC-ECD method; however, this is clinically insignificant and is likely to be a calibration issue.
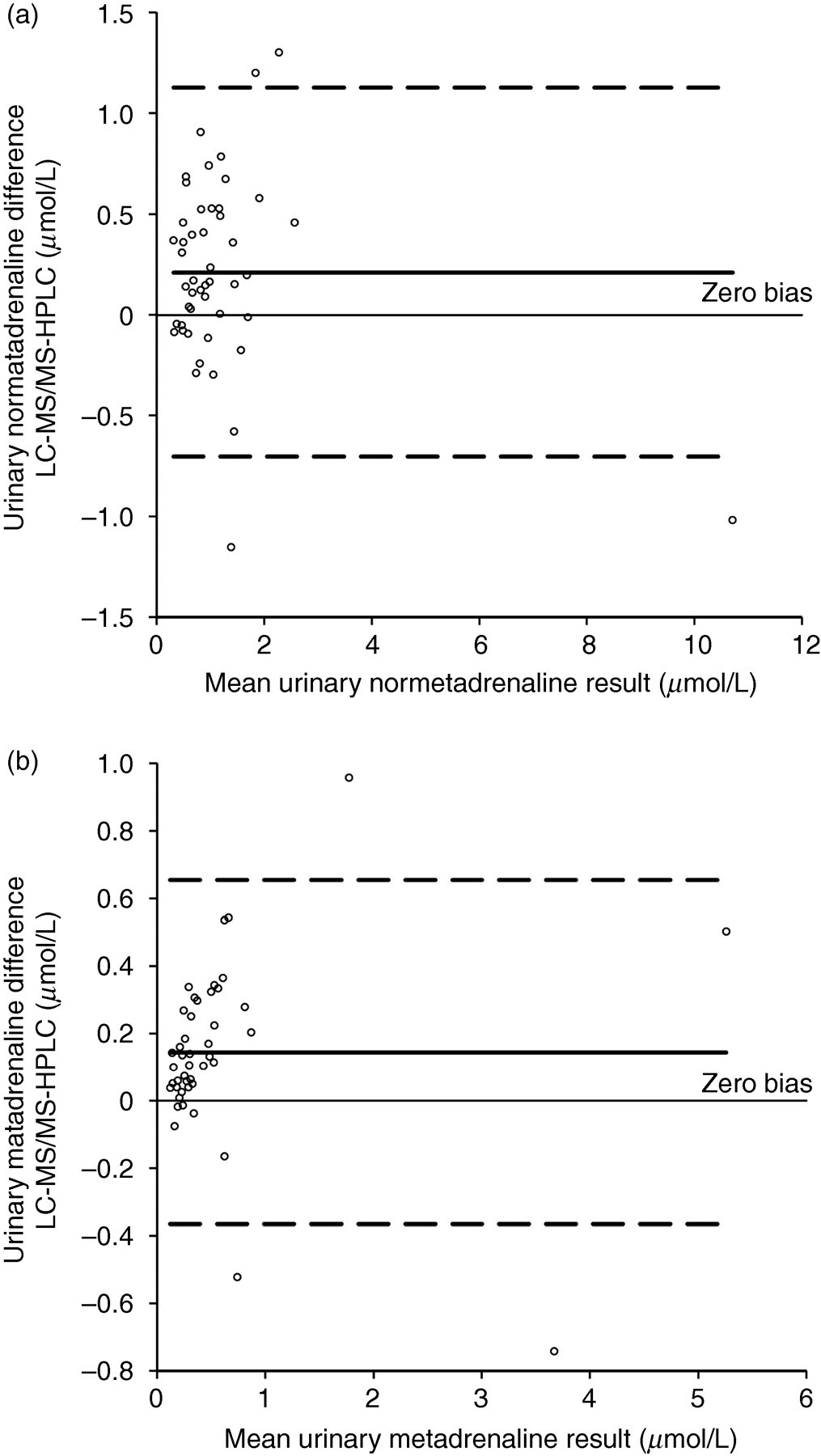
Bland-Altman difference plot comparing liquid chromatography–tandem mass spectrometry and high-performance liquid chromatography methods for (a) urinary normetadrenaline and (b) urinary metadrenaline. The dashed lines represent the 95% confidence interval
Discussion
Measurement of urinary catecholamines is routinely offered by many laboratories as their first-line test for the biochemical investigation of phaeochromocytoma. Over the past few years it has been shown that urinary metadrenalines have improved sensitivity and specificity for phaeochromocytoma and as such HPLC and immunoassay methods have become available. HPLC methods have historically been used for the detection of urinary catecholamines and more recently metadrenalines; however, these methods have been reported to be subject to interference from several drugs. 9 Patients with phaeochromocytoma are likely to have hypertension and therefore are likely to be taking several medications. LC–MS/MS has the advantage that in vitro drug interference should be minimized due to the increased analytical specificity. However, in vivo drug interference could still cause falsely elevated results.
Manual immunoassay methods are also commonly used, but the main advantages of LC–MS/MS methods are that they are less labour intensive and have lower running costs.
The LC–MS/MS method developed here for the quantification of total urinary normetadrenaline and total urinary metadrenaline does not require the use of a solid phase extraction prior to analysis. This has important benefits such as reducing the cost of analysis and the time required to process samples. The method described uses an acid hydrolysis step to convert conjugated metabolites into free metadrenalines and the hydrolysates are injected directly into the system. The system is robust and has been in routine use for over a year and has minimal downtime due to column blockages or degradation. Column costs have been minimal with a columns lasting for over 1500 injections before column degradation takes place.
Adrenaline and normetadrenaline are isobaric and produce the same fragmentation patterns when undergoing MS/MS. The use of in-source fragmentation (loss of water) has been utilized to minimize the effect of ‘cross talk’ from the isobaric compounds in the mass reactions used for the determination of adrenaline and normetadrenaline. However, cross talk in the normetadrenaline transition from adrenaline was still evident. It is therefore important that chromatographic separation of the compounds is achieved. Due to catecholamines and the metadrenalines being small polar compounds, there is often poor retention on traditional reverse phase chromatographic solid phases. Here a PFP-doped reverse phase column is used to enhance retention and selectivity increase chromatographic efficiency. This allowed complete baseline separation of the catecholamines – noradrenaline, adrenaline and dopamine – from normetadrenaline and metadrenaline (Figure 6); therefore, interference is potentially eliminated. Patient samples were spiked with high concentrations of noradrenaline and adrenaline and no interference was observed. We have run this method routinely for over a year and have chromatographically separated all peaks of interest without any problems of co-elution of peaks due to in vitro drug interference. There is still the potential for in vivo interference which would be the same for any analytical technique.
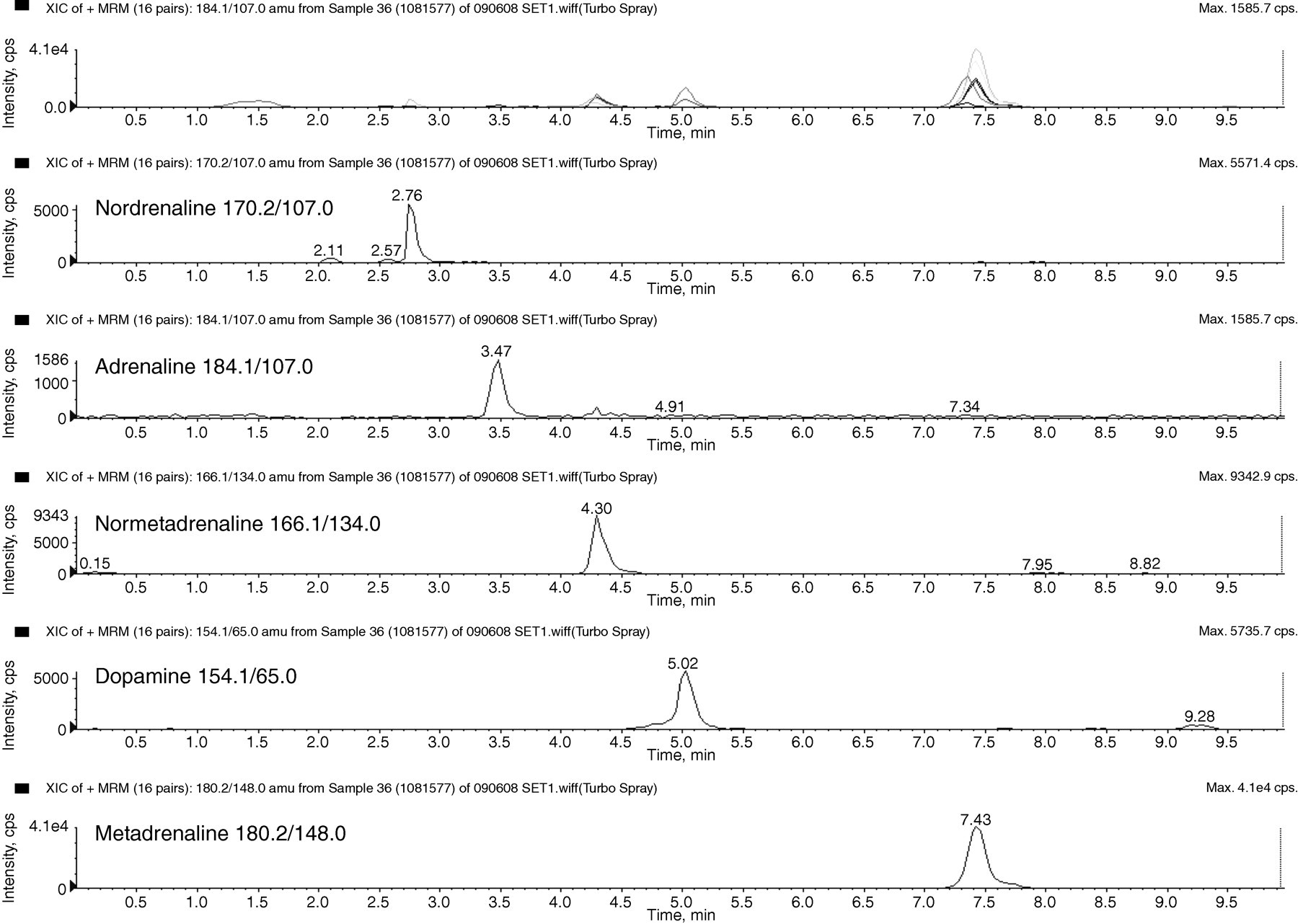
Chromatograms of the individual extracted ions for catecholamines and metadrenalines in a urine sample to show baseline separation. Concentrations of the analytes are as follows: normetadrenaline 1.29 μmol/L, metadrenaline 2.01 μmol/L, noradrenaline approximately 0.15 μmol/L, adrenaline approximately 0.12 μmol/L and dopamine approximately 0.27 μmol/L
This method has not been optimized for the detection and quantification of the catecholamines. There is the potential to develop this method further to allow the quantification of urinary free adrenaline, urinary free noradrenaline, urinary free dopamine, urinary total metadrenaline and urinary total normetadrenaline in one injection. However, a separate unhydrolysed urine sample would have to be analysed to maintain equivalence with current urinary free catecholamine methods.
This LC–MS/MS method has been validated and implemented into routine use. A recent external quality assurance return (UKNEQAS Urinary Catecholamines Distribution 122) gave B-scores of 1.5 and 7.1 for total metadrenaline and total normetadrenaline, respectively, and C-scores of 12.0 and 7.1 for total metadrenaline and total normetadrenaline, respectively. The all-laboratory-trimmed-mean is currently based on HPLC methods due to no other users analysing samples by LC–MS/MS. The within-assay precision for both analytes was less than 5.5% and between-assay precision less than 13%.
In conclusion, an analytically simple, specific and sensitive method has been developed and evaluated for the analysis of total urinary normetadrenaline and total urinary metadrenaline without the requirement for solid phase extraction which is now in routine use.
DECLARATIONS
