Abstract
Interference in immunoassay caused by endogenous immunoglobulin is a cause of incorrect laboratory results that can drastically affect patient management. Two cases of immunoglobulin interference in serum follicle-stimulating hormone (FSH) assays are presented. These cases illustrate two common mechanisms for false-positive interference in two-site (sandwich) immunoassays. The first case describes a circulating autoimmune FSH immunoglobulin complex (‘macro’-FSH), which has not been previously described for FSH, and the second a cross-linking antibody directed against the assay reagents. Immunoglobulin interference was detected and characterized using a combination of method comparison, immunosubtraction and size exclusion chromatography.
Introduction
Interference in immunoassay, although rare, is a cause of incorrect laboratory results that can drastically affect patient management. 1 This is often caused by endogenous immunoglobulins (Ig) directed against either the assay reagents causing blocking or cross-linking of the sandwich assay components (high-affinity human anti-animal antibodies or weaker polyspecific heterophilic antibodies) or directed against the analyte itself (autoimmune) leading to the formation of Ig analyte or ‘macro-hormone’ complexes. We describe two cases of false-positive assay interference in serum follicle-stimulating hormone (FSH) measurement, illustrating both mechanisms, including the first described case of false-positive interference in an FSH assay due to an autoimmune Ig–FSH complex (‘macro-FSH’). In each case assay interference was suspected on the basis of discordance between the serum FSH and leutinizing hormone (LH) results and the clinical context. Once suspected, Ig interference was detected and characterized using a combination of method comparison, immunosubtraction and size exclusion chromatography.
Cases
Case 1
A 24-year-old female with a previous successful pregnancy presented with hot flushes, nausea, bloating and left iliac fossa pain five months after an elective termination of pregnancy. A low serum oestradiol concentration of 87 pmol/L (reference interval 100–750) together with previous secondary amenorrhoea raised concerns of ovarian failure, although the patient was not amenorrhoeic at presentation. At presentation serum FSH concentration (Perkin-Elmer DELFIA assay) was 78.9 IU/L (reference interval, follicular phase 2.9–8.4 IU/L) and serum LH concentration (DELFIA assay) was 11.1 IU/L (reference interval, follicular phase 1.3–8.4 IU/L). However, within four months the patient conceived again. Serum FSH became undetectable during pregnancy, but notably this occurred more slowly than would normally be expected. Serum FSH was 8.7 IU/L when the serum β-human chorionic gonadotropin (βHCG) concentration was 2422 IU/L, a βHCG concentration, which is usually associated with undetectable FSH using the DELFIA assay. Following a miscarriage her serum FSH concentration again rose to elevated levels (32 IU/L). Subsequently, the patient had a second successful pregnancy, during which serum FSH concentrations again became undetectable with the same time course, returning to high values (20.6 IU/L) after delivery.
Case 2
A 16-year-old male was investigated following a past history of acute lymphoblastic leukaemia when he was age 2, treated with chemotherapy (daunorubicin and epirubicin) and testicular irradiation for a gonadal relapse when he was age 9. He has since remained in remission and progressed through puberty un-aided, but due to a small left testicle, serum testosterone and gonadotrophins were measured (Siemens Immulite 2000, Siemens Healthcare Diagnostics Ltd, Camberley, UK) with results showing FSH concentration >170 IU/L (reference interval 1–8), LH 37 IU/L (reference interval 0.5–9) and testosterone 4.7 nmol/L (reference interval 11–36).
Neither patient had experienced significant contact with animals nor had either been treated with recombinant Ig therapy.
Investigations
Table 1 describes the method comparison and immunosubtraction data. Serum FSH concentrations were measured from a single serum sample (volume permitting) using five different methods in routine use in clinical chemistry laboratories. Values varied by a factor of seven and four for Cases 1 and 2, respectively. External Quality Assurance data for these methods at the time of analysis showed that these differences could not be accounted for by method bias as the cumulative bias from the All Laboratory Trimmed Mean was as follows: DELFIA −13%, Abbott −2%, Elecsys 0% and Immulite 8% (data used with permission, UK NEQAS for Peptide Hormones, Edinburgh, UK). Polyethylene glycol (PEG) precipitation is a quick and simple method to detect Ig interference in peptide hormone assays;
2
this technique has been best described for ‘macroprolactin’ interference in serum prolactin assays.
3
One-hundred and fifty microlitres of patient serum were mixed by vortexing with an equal volume of a 25% (w/v) solution of PEG 6000 (BDH, Dorset, UK) in deionized water. The mixture was centrifuged at 17,000
Method comparison and immunosubtraction studies
FSH, follicle-stimulating hormone; PEG, polyethylene glycol
*Range of data from 10 healthy controls
Gel filtration chromatography was used to examine the nature of the interference further. In both cases a high molecular mass form of FSH immunoreactivity was detected (Figure 1a). The high molecular mass peak was co-incident with the elution volume of IgG in Case 2, but eluted earlier, with a smaller elution volume than IgG, in Case 1. Using the M r of albumin, prolactin and IgG and their respective elution times to calibrate the gel filtration column, gave an apparent M r of 47 kDa for FSH. This is clearly bigger than the known M r of 30 kDa and may be due to the asymmetry of the FSH heterodimer. 6 Using the same calibration the high M r FSH immunoreactivity in Case 1 was approximately 40 kDa bigger than IgG consistent with an Ig:FSH complex.
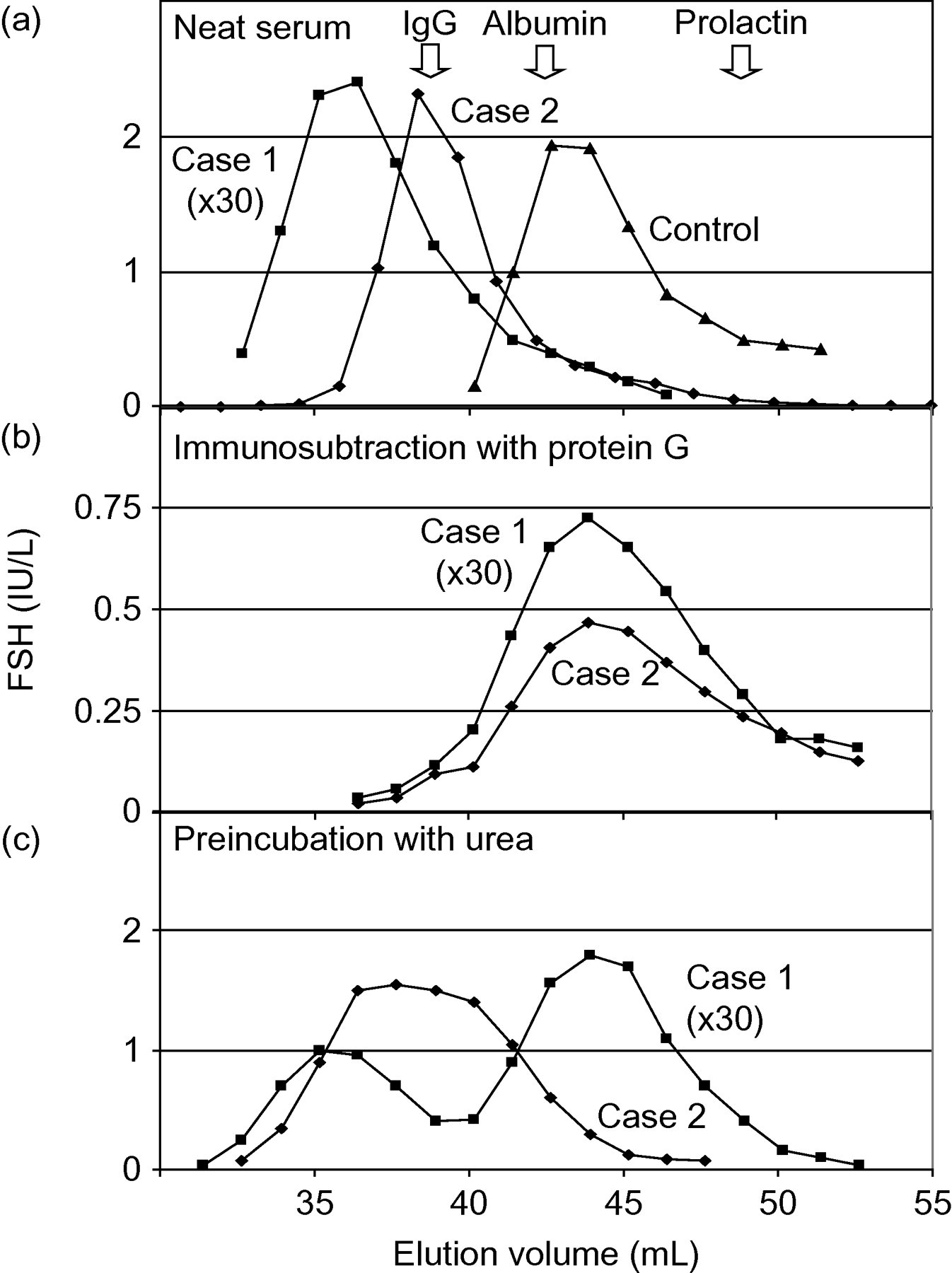
Gel filtration chromatography of patient serum. This was performed as described by Fahie Wilson (11) using Sephacryl S-300 (GE Healthcare Technologies, Berkshire, UK) and TRIS-buffered saline containing 1 g/L bovine serum albumin. FSH was detected using the DELFIA assay. (a) Elution profiles for both patients' serum samples and a control sample from a menopausal subject. (b) Chromatography repeated after incubation of serum samples with two volumes of protein G sepharose (GE Healthcare Technologies, Berkshire, UK) and centrifugation at 30 min at 1400
In both cases the high M r immunoreactivity was removed by incubation with protein G sepharose (Figure 1b), again confirming that an IgG species was responsible for the interference. We believe that protein G has removed a cross-linking IgG in Case 2, whereas in Case 1, it has removed a higher molecular mass Ig:FSH complex.
We also attempted to disrupt potential Ig:FSH complexes by preincubation of the patients' serum samples with 4 mol/L urea (Figure 1c); this had a dramatic effect with Case 1, extensively dissociating much of the IgG–FSH complex with the release of free FSH, but had no effect with Case 2.
Discussion
Our studies indicate that the high FSH immunoassay result in Case 1 was due to the presence of an autoimmune anti-FSH antibody leading to the formation of an immunoreactive IgG–FSH (macro-FSH) complex, as would be expected when FSH secretion is suppressed during pregnancy the complex disappears. The complex has an apparently higher molecular mass than IgG alone and is sensitive to urea-induced dissociation. In contrast, our findings in Case 2 suggest the presence of either a heterophile or human anti-animal antibody interfering with the FSH assay, despite the failure of the heterophile-blocking reagent to remove this effect. While heterophilic-blocking reagents are highly specific for the detecting of Ig-based assay interference they are not 100% sensitive, 7 a finding we have also observed when using them. Following protein G immunosubtraction, FSH immunoreactivity for Case 1 fell from 20.6 to 5.7 IU/L, which is within the reference interval (DELFIA assay) and consistent with normal gonadal function. However, for Case 2, the serum FSH concentration remained elevated after protein G treatment, falling from 740 to 222 IU/L. Despite the assay interference FSH is likely to be genuinely raised in this patient, consistent with his serum LH levels and as a consequence of poor gonadal function.
Autoantibody–hormone complexes (‘macro’-hormones) have been described for several peptide hormones (e.g. HCG, LH and thyroid-stimulating hormone) and most notably prolactin where their occurrence can be as high as 16%. 3,8 As Case 1 is likely to represent an anti-FSH autoantibody, this represents the first published description of ‘macro’-FSH. False-positive results due to autoimmune complexes with other serum proteins such as creatine kinase, amylase and, more recently, troponin 9 are well described. The variable immunoreactivity, of the autoimmune Ig:hormone complexes to different assays, is often explained on the basis of assay antibodies targeting different epitopes, which may or may not be blocked by the autoimmune Ig. 8 However, such results could also be explained by different relative affinities between assay capture antibodies and the autoimmune antibody in question. The difference in the assay results obtained due to the putative heterophilic interference in Case 2 is less well characterized. The effect of heterophilic antibody interference on any particular assay will be complex and will depend, in part, on the antibodies used, the assay design and the inclusion and concentration of any heterophilic-blocking reagents added by the manufacturer. While distinguishing between assay interference due to human anti-animal, heterophilic or autoimmune antibodies is largely academic, once suspected it does influence how the interference can be detected and blocked. Thus, reagents added to immunoassays to block heterophilic antibody interference will be ineffective against autoimmune hormone complexes.
As higher throughput immunoassay platforms are developed, the number of immunoassays performed in hospital laboratories has increased dramatically with results increasingly guiding clinical decision-making. As interference with immunoassays is generally rare (excluding some prolactin assays) it can be very difficult to detect. Possible individual risk factors include high levels of rheumatoid factor and occupational exposure to animals or recombinant monoclonal antibodies. Once antibody interference is suspected, laboratories can use several simple procedures for confirmation. No single test is comprehensive and a combination of these tests undertaken with analyte and assay-specific reference data may be more sensitive. 2 Many cases of clinical misdiagnosis have been described due to reliance on incorrect immunoassay results have been described. 10 Heightened vigilance when interpreting immunoassay results is critical and can be challenging if a clinical correlate for the analyte is not available.
DECLARATIONS
