Abstract
Background
Non-esterified fatty acid (NEFA) levels are an important diagnostic tool in the investigation of neonatal hypoglycaemia. NEFA reference intervals have not been reported for neonates previously.
Methods
The objective of this study was to determine an NEFA reference interval for neonates.
Results
Heparinized plasma obtained from 106 healthy neonates in the first week of life was analysed using the Roche “Free fatty acid, Half-micro test” kit. Results were then analysed statistically for normality (Shapiro–Wilk test) and reference interval determined non-parametrically (bootstrap method).
Conclusions
NEFA levels displayed a non-Gaussian distribution and the reference interval (2.5th and 97.5th percentiles) was 0.2–1.5 mmol/L (90% confidence intervals 0.1–0.3 and 1.4–2.0 mmol/L, respectively). The NEFA reference interval in South African neonates less than a week old is similar to that described in infants (1–12 months), indicating that this reference range can be used over the entire neonatal period.
Introduction
Free fatty acids, also called non-esterified fatty acids or NEFAs, are an important energy source in the fasted state. 1 In the neonate, plasma NEFA levels are an important diagnostic aid in patients presenting with metabolic acidosis and/or severe/persistent hypoglycaemia.
Although reference intervals are available for plasma and serum NEFAs in adults, limited information is available in the paediatric age group, with only a few studies describing reference intervals in this age population, one of which was for infants. 2–4 This reference interval, determined by an enzymatic method, for the age group 1–12 months was 0.5–1.6 mmol/L (n = 12; 10th and 90th percentiles). 3
No reference interval has been reported for neonates younger than seven days old. We therefore embarked on a study to determine such a reference interval in this age group.
Objective
The objective of this study was to establish a reference interval for NEFAs in neonates presenting at Red Cross Children's Hospital.
Methodology
Ethics approval was obtained from the University of Cape Town Health Sciences Research Ethics Committee. Heparinized plasma was obtained from 106 neonatal samples sent to our laboratory for routine bilirubin analysis from the maternal obstetric units. Samples were stored refrigerated prior to analysis, which was performed within six hours in keeping with guidelines. Only samples from presumed normal neonates with physiologically normal bilirubin levels were analysed. All subjects were healthy and asymptomatic, with no evidence of dysglycaemia. The exclusion criteria included hospital admission, abnormal bilirubin results, requests for any other biochemical analyses, including abnormal glucose values, and haemolysed samples. All neonates were younger than seven days of age. NEFA levels were analysed using the Roche ‘Free fatty acid, Half-micro test’ kit. This enzymatic assay uses acyl coenzyme A (CoA) synthetase and acyl CoA oxidase to convert NEFA into enoyl CoA and hydrogen peroxide. The hydrogen peroxide in turn oxidizes 4-amino-antipyrine forming a red dye that absorbs at 546 nm. Statistical analysis was performed using the Shapiro–Wilk 5 test for normality and the bootstrap (non-parametric) method for the reference interval determination. 6 There are three major issues in the determination of reference intervals: limited number of samples, elimination of potential outliers and estimations of confidence intervals for the reference range limits. Bootstrapping provides a robust alternative to methods to parametric methods and has become more popular because of the availability of personal computers. It uses computer-intensive re-sampling of the original data sample to estimate distributions. In this case, this entailed random selection (with replacement) of 500 samples of 106 values from the original sample of 106. The median 2.5th and 97.5th percentiles were then determined for the 500 samples and a reference interval established with 90% confidence intervals. Log transformation of the data failed to convert the data to a normal distribution, confirming the utility of this non-parametric approach. The use of ‘bootstrapping’ allows confidence intervals for the reference ranges to be determined.
Results
The NEFA levels for these patients were found to have a non-Gaussian distribution (Figure 1). The reference interval (2.5th and 97.5th percentiles) determined was 0.2–1.5 mmol/L (90% confidence intervals 0.1–0.3 and 1.4–2.0 mmol/L, respectively), while the reference interval for the 10–90 percentile was 0.4–1.3 mmol/L (90% confidence intervals 0.4–0.5 and 1.2–1.4 mmol/L, respectively).
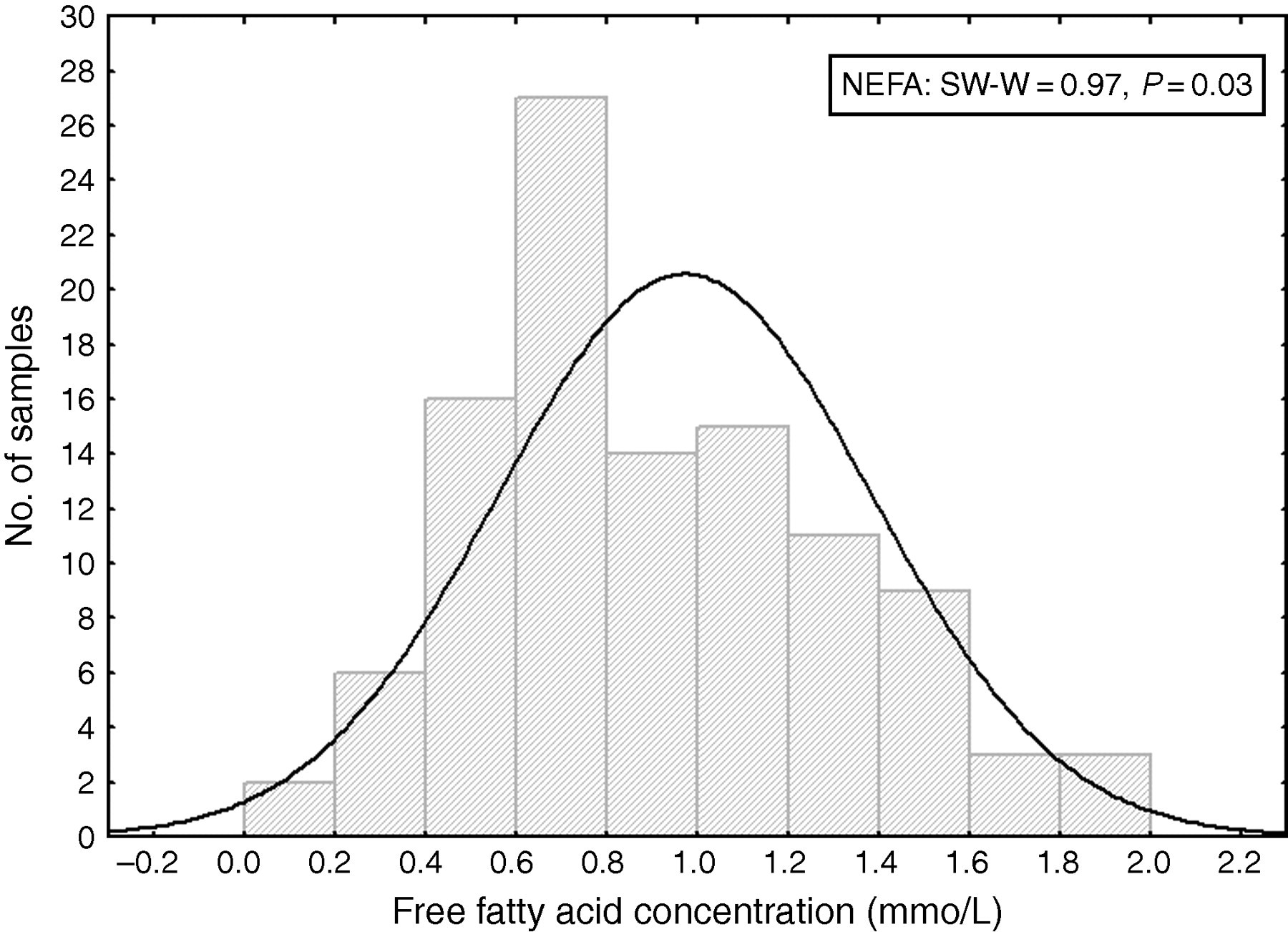
Frequency (non-Guassian) distribution of non-esterified/free fatty acid levels in South African neonates less than one week old. The solid line shows a simulated Gaussian distribution. SW, Shapiro–Wilk test for normality
Discussion and conclusion
This is the first description of a free fatty acid reference interval in neonates younger than a week old. We have established this interval for plasma free fatty acids in healthy South African neonates younger than seven days of age, using the Roche ‘Free fatty acid Half-micro test’ kit. We were unable to obtain samples from older healthy neonates and could therefore not extend this reference interval to include the entire neonatal period. However, this NEFA reference interval (0.4–1.3 mmol/L; 10th and 90th percentile) is similar to that described by Bonnefont et al. 3 for infants aged 1–12 months (0.5–1.6 mmol/L; 10th and 90th percentile, respectively), also established using an enzymatic method. We are therefore confident that our reference interval for NEFA may be used across the entire neonatal period.
DECLARATIONS
