Abstract
We present the case of an eight-year-old boy with advanced isosexual precocity associated with an elevated serum total-beta human chorionic gonadotrophin (HCG) and markedly elevated serum total testosterone. Radiological investigation discovered a lesion in the left thalamus and no peripheral tumour. Serum:cerebrospinal fluid (CSF) HCG ratio was approximately 1:1, consistent with a central nervous system source of HCG, with thalamic germinoma strongly suspected. Consent was not obtained for biopsy of the lesion. The patient underwent multiagent chemotherapy with return of serum HCG to normal. We discuss mechanisms of HCG-mediated sexual precocity in both boys and girls and the importance of CSF HCG.
Introduction
Laboratory investigation of precocious sexual development is guided by clinical evaluation. It has been suggested that evaluation of the mechanism of sexual precocity be undertaken in all boys and only in girls exhibiting certain clinical features. 1 A practical approach to classifying sexual precocity is into gonadotrophin-dependent (or ‘central’) and gonadotrophin-independent (‘peripheral’) causes. This classification depends on accurate estimation of serum gonadotrophin concentrations, testosterone and oestradiol, requiring assays able to measure reliably low concentrations and distinguish prepubertal from pubertal values. 2
Human chorionic gonadotrophin (HCG)-secreting neoplasms are a recognized cause of precocious sexual development, almost exclusively in boys. These tumours may occur in the gonads, liver, retroperitoneum and mediastinum or within the central nervous system (CNS). 3 HCG-producing germ cell tumours of the CNS are rare and sexual precocity as the presenting problem is even rarer. The most common sites for these tumours are in the suprasellar or pineal regions with other sites, such as the basal ganglia, less common. 4 Choriocarcinoma is the archetypical germ cell tumour producing HCG; however, any germ cell tumour with syncytiotrophoblastic giant cells may produce HCG, including germinomas. 5 In addition, some mononuclear germinoma cells can produce HCG themselves. 6
Case report
An eight-year eight-month-old boy of Asian descent presented with a six-month history of rapid pubertal development characterized by deepening voice, development of facial and pubic hair, penile growth and the appearance of acne. His past medical history was unremarkable, his prior development was normal and he was on no regular medications. His mother's menarche was at age 13 and the pubertal development of his father was not known. Physical examination revealed a child with adult physique and height and weight approximately 90th percentile for age. Neurological examination was normal. Tanner staging was IV for genital development and III–IV for pubic hair. His testicular volumes were 5 mL (left) and 6 mL (right) with no palpable mass. Stretched penile length was 9 cm.
Salient biochemistry is shown in Table 1. Testing was remarkable for elevated total beta-HCG with normal alpha-fetoprotein (AFP), prepubertal concentrations of gonadotrophins and a markedly elevated total testosterone. All assays were performed on Beckman Coulter DXI-800 Access® analysers (Beckman Coulter Inc, Fullerton, CA, USA).
Selected biochemistry
HCG, human chorionic gonadotrophin (total-beta); LH, luteinizing hormone (limit of detection 0.2); FSH, follicle-stimulating hormone (limit of detection 0.7); total testo, total testosterone; AFP, alpha-fetoprotein; PRL, prolactin; TSH, thyroid-stimulating hormone; chemo, chemotherapy
*Free beta-HCG not detected, qualitative HCG test positive on serum
†Concurrent cerebrospinal fluid HCG 98 IU/L, AFP <2.0 μg/L
Radiological investigation was undertaken to search for a tumour. Magnetic resonance imaging (MRI) of the brain revealed an approximately 1 × 1 cm2 cystic lesion in the left thalamus with appearances most in keeping with a cavernoma (see Figure 1). MRI of the spine was normal. Computed tomography scans of the abdomen, pelvis and chest were normal, as was an ultrasound of the testes. Bone age of the left wrist was 12.5 y (Greulich and Pyle method). A positron emission tomography scan revealed no fludeoxyglucose-avid disease.
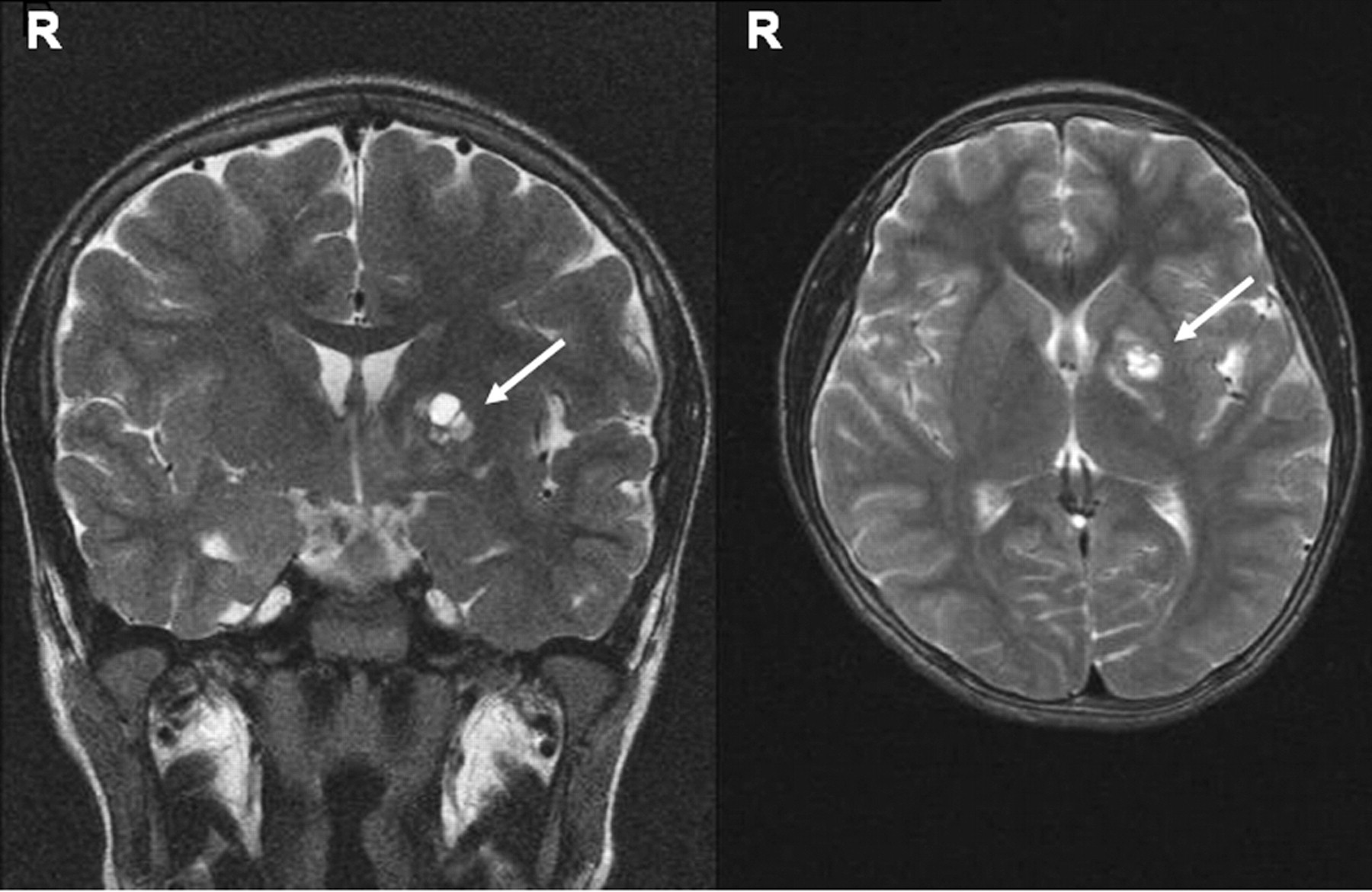
T2-weighted coronal (left) and axial (right) magnetic resonance imaging images of the brain. The patient's right side is indicated with an ‘R’. Arrow to lesion
During this period, the chemical pathology laboratory was approached for an opinion. With the limited sample available to the laboratory at the time, serum free beta-subunit of HCG was not detected (Immulite® 2000, Siemens Healthcare Diagnostics, Deerfield, IL, USA) and a qualitative HCG test on serum was positive (detection limit 25 IU/L, TestPack Plus hCG Combo®, Inverness Medical, Bedford, UK). Lumbar cerebrospinal fluid (CSF) was taken and tested for HCG, which revealed a concentration of 98 IU/L (concurrent serum 104 IU/L). The patient's serum alkaline phosphatase was elevated during this period, up to 724 U/L (reference interval 54–369 U/L, Beckman Coulter); however, the opportunity was missed to test for malignancy-associated isoenzymes.
Expert review of the original MRI brain suggested a diagnosis of thalamic germinoma. Parental consent was not obtained for biopsy. The biochemistry was consistent with this diagnosis and the patient underwent multiagent chemotherapy with subsequent reduction of serum HCG and total testosterone to <5 IU/L and 0.6 nmol/L, respectively, over four weeks.
After two cycles of chemotherapy, the thalamic lesion remained the same size on MRI and appearances were still most in keeping with cavernoma. Given the risk of secondary central precocious puberty due to prolonged exposure to sex steroids, treatment with a gonadotrophin-releasing hormone agonist was commenced in an attempt to preserve final adult height.
Discussion
Mechanism of HCG-mediated sexual precocity
There are two potential mechanisms of sexual precocity in patients with CNS HCG-producing tumours. 7 The first, most common and the mechanism seen in boys, is a gonadotrophin-independent sexual precocity due to HCG stimulation of the luteinizing hormone (LH) receptor in the testes, thereby producing excessive testosterone and sexual precocity. This is due to the strong structural similarity between LH and HCG. The CNS produced HCG would obviously require access to the systemic circulation for this to occur. This is probably the mechanism in our patient, due to the prepubertal concentrations of gonadotrophins: an anatomically ‘central’ gonadotrophin-independent cause. The second mechanism is gonadotrophin-dependent sexual precocity due to the proximity of the lesion to the hypothalamus, although any mass lesion close to the hypothalamus may produce gonadotrophin-dependent precocious puberty. 7 The mechanisms are not well understood but may directly relate to the location and type of lesion. 8
Sexual precocity in prepubertal girls with HCG-secreting CNS tumours is much rarer than in boys, with only a handful of cases reported in the literature. 9–13 Using gonadotrophin assays with satisfactory lower limits of quantitation, this sexual precocity in girls has been demonstrated to be gonadotrophin-independent. Both LH and follicle-stimulating hormone (FSH) are required physiologically to produce oestrogen from the ovary. Several theories have been proposed to account for gonadotrophin-independent precocity in these cases, including HCG having both LH- and FSH-like activity, secretion of another substance by the tumour with FSH-like activity or the tumour having aromatase activity in addition to its HCG secretion, thus converting HCG-stimulated ovarian androgens to oestrogen and thereby causing isosexual precocity. The rarity of sexual precocity caused by CNS HCG-producing tumours in girls may be due to the lower occurrence of these tumours in girls and rare simultaneous presence of high tumour aromatase activity and HCG secretion. 13
HCG and the blood–brain barrier
One study of pregnant women in the third trimester suggested the normal ratio of serum:CSF HCG is in the order of 290:1, 14 indicating HCG crosses the blood–brain barrier (BBB) poorly. The mechanism of HCG crossing the BBB is not known, but may be diffusion across the capillary endothelium. 14 The form of HCG most prevalent in normal third trimester serum is intact HCG. 15 In those with peripheral HCG-producing neoplasms without cerebral metastases, the ratio is similar with a mean of 286:1 (63:1–1546:1), 16 implying malignancy associated HCG crosses the BBB similarly to intact HCG.
If the same principles of HCG crossing the BBB held for primary CNS sources of HCG, then one might expect a reverse ratio of similar magnitude, i.e. serum:CSF ratio of about 1:280 (with some consideration of method detection limits). Rogers et al. 17 described a series of patients with HCG-producing CNS tumours, four of whom had pineal lesions with a mean serum:CSF ratio of 1:10 (1:1.7–1:18.4). The magnitude of this reverse ratio is considerably smaller than in systemic HCG-producing neoplasms, suggesting that the pineal region has more ‘privileged’ access to the blood than would be predicted from passage across the BBB alone. This also implies that CSF HCG is more sensitive than serum for detection of these tumours, which has been the experience of other authors in some cases. 18,19 One must also consider the integrity of the BBB, as a leaky or abnormal barrier, as one might expect around a malignant CNS tumour and/or effects of raised intracranial pressure, will also facilitate greater passage of proteins, including HCG.
The site of sampling of CSF also appears important. In patients with HCG-producing CNS tumours and ventriculoperitoneal shunts, sampling of lateral ventricular CSF HCG can yield very low or undetectable concentrations when concurrent lumbar CSF HCG is high. 17,20
An increased CSF HCG implies a primary CNS source of the HCG or CNS involvement by a peripheral HCG-producing tumour (with or without an abnormal BBB). The magnitude of the serum:CSF ratio has been suggested previously to detect the presence of cerebral metastases in patients with peripheral HCG-producing tumours; 21 however, one study did not find a serum:CSF ratio of 60:1 useful in this regard. 22
Our patient had a serum:CSF HCG ratio of approximately 1:1 and this abnormal ratio helped confirm the presence of a CNS tumour.
Prognostic value of raised HCG
In addition to its value in diagnosis and monitoring, the prognostic value of serum and/or CSF HCG has been studied in patients with primary CNS germinoma. The literature is divided on whether germinomas with high HCG concentrations have poorer prognosis and/or high recurrence rates than those with normal HCG concentrations. 23–27
A more recent retrospective review of 103 patients with primary CNS germinoma found serum HCG does not appear to influence prognosis. 28
The various histological types of germ cell tumours produce different amounts of HCG, with choriocarcinoma typically having higher CSF and serum HCG than other subtypes 4 and a poorer prognosis than germinoma or teratoma. 29 A recent retrospective analysis of unselected primary CNS germ cell tumours found patients with elevated tumour markers (serum and CSF HCG and/or AFP) had a poorer five-year survival independent of tumour histology, although these differences did not reach statistical significance. 30 A subgroup analysis in biopsy-proven germinoma found no survival difference between high and normal HCG cases.
Relevance of HCG assays and molecular forms
HCG exists in many different forms and there is evidence that different assays recognize these various forms to different degrees. 15 Neoplasms may produce intact HCG, but more typically produce hyperglycosylated HCG and/or free beta-subunits. 15 One needs to bear these facts in mind when considering the literature as not all studies that discuss HCG-producing tumours have described their assay methods. As an example, ‘beta-HCG’ may refer to total beta-HCG (intact plus free beta-subunit) or the free beta-subunit alone.
Basic investigations on our patient suggested no assay interference and that the HCG was probably an intact (alpha plus beta-subunit) form due to high concentrations in a total beta HCG assay, negative free beta-subunit and positivity on a qualitative assay that measures intact HCG.
Shinoda et al. 4 have published a detailed review of intracranial germ cell tumours with high concentrations of HCG. Of those presenting with precocious pubertal development, our patient is at the low end of the spectrum in terms of serum HCG concentrations. Of interest, two boys with significantly higher serum HCG concentrations were not noted to have precocious puberty. Why some prepubertal boys with considerably higher serum HCG concentrations than our patient do not develop precocious puberty, while others with similar or lower serum concentrations develop precocious puberty, is not known but may relate to differences in molecular forms produced and possibly LH receptor-related factors as well. Conceivably, some cases of precocious puberty described in boys with these tumours close to the hypothalamus may actually be gonadotrophin dependent, as serum gonadotrophin concentrations are not reported in all cases.
There has been much research on HCG-mediated binding to the LH receptor, and it appears that both the alpha- and beta-subunits are of importance in binding and activating the receptor. 31 Indeed, there is experimental evidence that the individual subunits are devoid of activity at the LH receptor. 32 It is well known that the beta-subunit confers biological specificity to the glycoprotein hormones LH, FSH, HCG and thyroid-stimulating hormone (TSH), which share a common alpha-subunit. The beta-LH and beta-HCG peptides share close sequence homology. The beta-HCG peptide is 24 amino acids longer than the beta-LH peptide, with 80% of the first 115 amino acids being identical. Evolutionarily, it is thought that the beta-HCG genes (of which there are seven) evolved from an ancestral beta-LH gene and that a single base deletion in this ancestral beta-LH gene caused the 24-amino-acid extension resulting in the longer beta-HCG peptide. 33 HCG, when present in very high concentrations, can also stimulate the TSH receptor and cause thyrotoxicosis. 34
Lastly, interferences in HCG immunoassays are well described, as are methods of their detection. Serum HCG detectable (on routine assays) in men is always abnormal and it is clearly important to establish any raised HCG as being pathological and not an interference, lest the patient be exposed to unnecessary investigations, procedures or treatment.
DECLARATIONS
