Abstract
Background
We have previously reported an ultrasensitive fluorometric assay for measuring cellular cholesterol. Although this technique is reliable, the use of the assay has limitations due to the requirement for special equipment. It is therefore difficult to apply this assay for the routine determination of cellular cholesterol.
Methods
A colorimetric assay to measure cellular cholesterol was established that utilizes reagents widely used for the measurement of cholesterol in blood samples in conjunction with a random access chemistry analyser ARCHITECT c8000 that is also common in clinical laboratories.
Results
This colorimetric assay showed excellent linearity and recovery. The within-run coefficients of variation were less than 2.5%. The sensitivity of this method, with its detection limit of 1.29 μmol/L, was found to be superior to that of the fluorometric assay we have developed previously. In platelets obtained from patients with diabetes, both the free cholesterol and cholesterol ester content were significantly increased.
Conclusions
Using this technique, measurement of cellular cholesterol could be performed routinely without the requirement for special reagents and equipment.
Introduction
As the incidence of atherosclerotic vascular disease has increased in recent years, as caused by excess concentrations of cholesterol, measurement of cellular cholesterol has become of major importance. 1,2 Recently, increased platelet cholesterol has been reported as a risk factor for coronary artery disease. 3 Hyperactive platelets with increased platelet cholesterol may be partly responsible for the accelerated atherogenesis. It has therefore been suggested that measuring platelet cholesterol could aid in predicting the possibility of accelerated atherogenesis.
The enzymatic determination of cholesterol by either colorimetric or fluorometric methods is simple and highly sensitive, allowing us to determine directly total cholesterol (TC) as well as free cholesterol (FC) using a single reagent and a base. 4 We have reported an ultrasensitive fluorometric assay for measuring cellular cholesterol. 5 Using this method, we found a significant increase in the cholesterol ester (CE) content in platelets from patients with atherosclerotic disease. 6 Although this technique is fully automated and reliable, the use of this assay has limitations due to the need for special equipment and its ability to perform only a confined menu of tests. It is therefore difficult to apply this assay for the routine determination of cellular cholesterol.
We describe herein an enzymatic determination of cellular cholesterol by a colorimetric method using reagents used for the measurement of TC and FC in blood samples in conjunction with a random access chemistry analyser that is common in clinical laboratories. The cholesterol content in platelets was assayed using this colorimetric technique, and its accuracy and usefulness were evaluated.
Materials and methods
Subjects studied
Fifteen healthy volunteers (8 men, 7 women; mean age, 33.0 ± 6.23) with normal plasma lipid concentrations and euglycaemia were selected for the study of cholesterol composition in circulating platelets. Peripheral blood platelets were also collected from 50 diabetic patients (23 men, 27 women; mean age, 48.0 ± 12.05). There were six patients (12%) with hypercholesterolaemia, 18 (36%) with hypertriglyceridaemia and 14 (28%) with hypertension. All samples were obtained with informed consent.
Sample preparation
Preparation of human platelets was performed as described previously. 5 Briefly, after counting the numbers of platelets using an XE-2100 multiparameter automatic haematology analyser (Sysmex, Kobe, Japan), isolated platelets were resuspended at a concentration of 4 × 107/mL of 0.2 mol/L Tricine-NaOH buffer (pH 7.5) containing 0.4% Triton X-100. The mixture was then vortexed and kept at −20°C until assay.
Cholesterol assay
TC and FC concentrations were measured using a Determiner L TC II, Determiner L FC assay reagent and Determiner Standard Serum For Lipid Assay (Kyowa Medex, Tokyo, Japan) on a random access chemistry analyser ARCHITECT c8000 (Abbott Laboratories, Abbott Park, IL, USA) according to the manufacturer's instructions with alternation of the sample/reagent ratio. For reagent preparation, a 35-μL sample was preincubated with 150 μL of reagent-1 for five minutes at 37°C, and 50 μL of reagent-2 was then added. After five minutes, the absorbance was measured at dual wavelengths of 604 and 804 nm. To measure CE, FC was subtracted from TC.
Statistical analysis
Grouped data are expressed as the mean ± standard deviation (SD). Variables between groups were evaluated using the Mann-Whitney U test.
Results
Linearity and recovery test
Linearity plots were obtained for TC, FC and CE assays using samples containing different numbers of platelets. All the assays evaluated showed a linear relationship between the platelet counts and the concentrations of cellular cholesterol (Figure 1a). Samples with additional cholesterol were made by adding a 1/10th volume of standard serums to five different platelet samples. The means of the percent recovery for TC, FC and CE assays compared with expected values were 99.2 ± 1.0%, 99.9 ± 1.1% and 98.0 ± 2.0%, respectively.
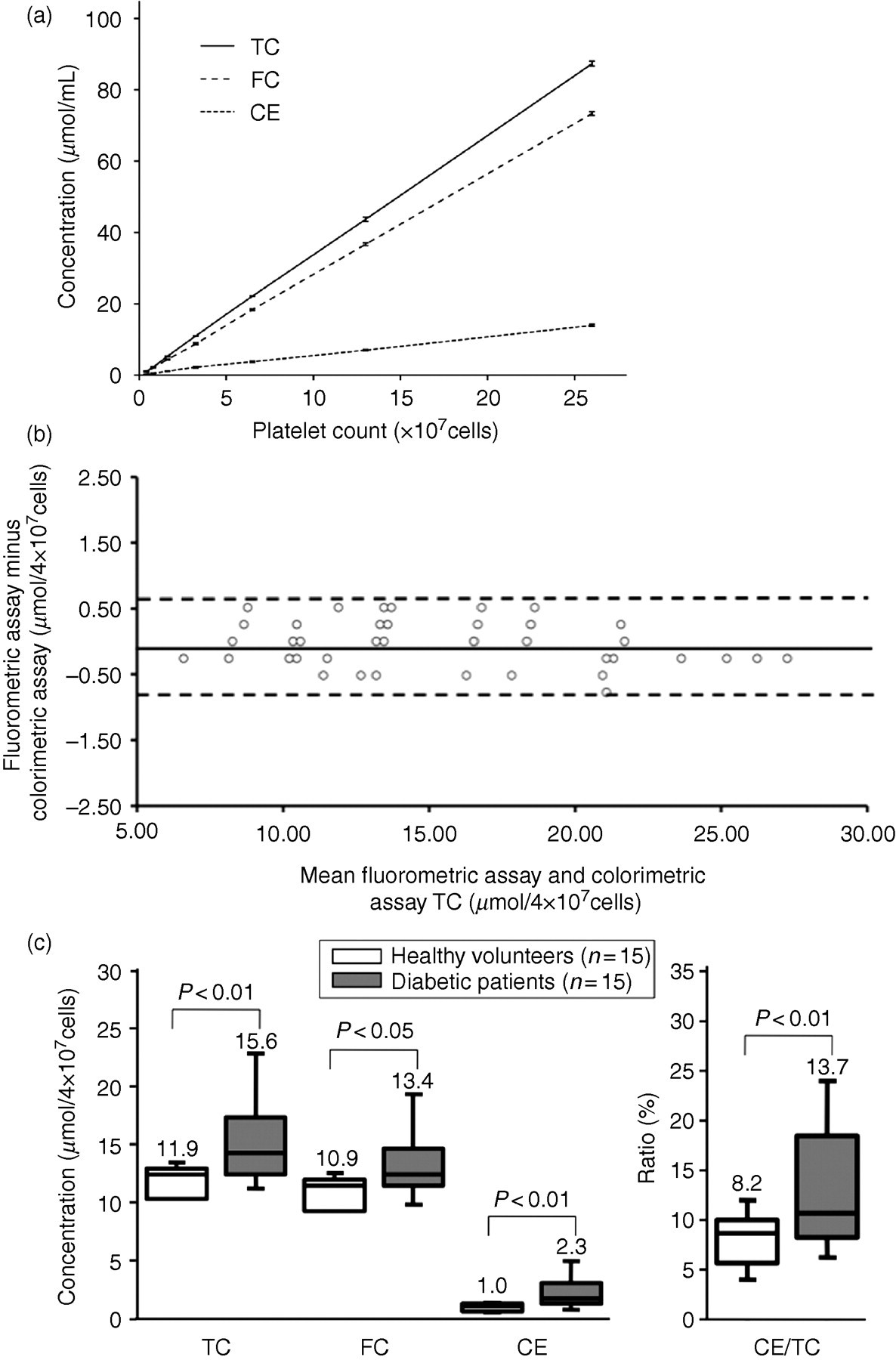
(a) Linearity of the TC, FC and CE assays. Aliquots of the same platelet samples diluted with buffer were used for each assay. Each data point represents the average ± SD of data collected in three experiments. (b) Bland–Altman plots show the measurement of TC in platelets using the colorimetric and fluorometric assays. Relative differences between the colorimetric and fluorometric assays for TC concentrations in platelets are plotted against the mean concentrations determined by both methods. The mean differences are shown by solid lines, and the 95% limits of agreement are displayed by dashed lines. (c) The mean and range of cellular cholesterol in the platelets. Box–whisker plots of TC, FC, CE and CE/TC in the platelets from diabetic patients and normal individuals are displayed. TC, total cholesterol; FC, free cholesterol; CE, cholesterol ester
Within-run precision
Two control samples of platelets from healthy volunteers were used to examine the within-run coefficients of variation (CVs). Each sample was measured in 20 replicates. The mean SDs were 10.3 ± 0.2 μmol/4 × 107 cells and 20.1 ± 0.3 μmol/4 × 107 cells. The CVs were 2.23% and 1.48%, respectively.
Detection limit
The detection limit of this colorimetric assay was defined as the cholesterol concentration at which the mean −3 SD for a diluted control sample was not exceeded by the mean +3 SD for a blank. Each sample, including the blank, was measured in five replicates. All the blank measurements were 0 μmol/L, and the measurement for the lowest diluted control serum was 1.29 μmol/L. The sensitivity of this new method is almost twice as high as that of our previous fluorometric assay with a detection limit of 2.59 μmol/L. 5
Correlation with our ultrasensitive fluorometric assay
Bland–Altman plots are shown in Figure 1b. The concentrations measured using the present method were found to have means 0.04 μmol/4 × 107 cells (range: −0.3 μmol/4 × 107 cells to 0.5 μmol/4 × 107 cells) higher than those determined by the previous fluorometric assay. Nearly all of the data points were located within the 1.96–SD intervals. These intervals were of acceptable range.
Measurement of TC, FC and CE content in human platelets
We have reported a significant increase in the CE content in platelets from patients with atherosclerotic disease using a previous fluorometric method. 6 Using this colorimetric assay, we measured the cellular TC, FC and CE content in human platelets with or without diabetes, as described in Materials and methods. In platelets obtained from diabetic patients, TC, FC, CE and CE/TC were significantly increased (Figure 1c). There were no significant relationships between platelet cholesterol and plasma cholesterol (data not shown).
Using this technique, measurement of cellular cholesterol could be performed not only in platelets, but also in cultured haematopoietic cell lines and in human peripheral blood mononuclear cells (data not shown). Thus, this method can be used for the measurement of cellular cholesterol in different cell types.
Discussion
Examination of cellular cholesterol provides important information regarding the risk of atherosclerotic vascular disease. Although various kits for cholesterol quantitation are commercially available, most are costly and have been designed for manual use of a microplate reader. Thus, there is considerable demand for a more generally available automated assay to analyse cellular cholesterol at low cost.
We have developed a technique for enzymatic determination of cellular cholesterol by a colorimetric method. This method utilizes reagents for the measurement of cholesterol in serum in conjunction with an analyser that is commonly used in clinical laboratories. This method requires the addition of only one cellular lipid extraction step to the routine measurement of cholesterol in blood samples. The sensitivity of this assay is superior to that of our previous fluorometric assay. The high sensitivity of this method was achieved by alternation of the sample/reagent ratio, as described in the Materials and methods. This alternation is made possible by employing miniaturized reaction chambers (minimal volume = 160 μL) for the ARCHITECT c8000. Using this assay, highly sensitive measurement of cellular cholesterol can be performed in daily clinical practice without the need for special reagents or complicated equipment.
In the present study, no significant relationships were observed between platelet cholesterol and plasma cholesterol. Ravindran et al. 3 also found no correlation between platelet cholesterol and plasma cholesterol. Although the translocation of cholesterol to the microparticle fraction has been speculated as a cause of this discrepancy, it is still a matter of debate why platelet cholesterol showed no correlation with plasma cholesterol in these studies. We have previously found a negative correlation between the accumulation of CE in platelets and an abundance of hSR-B1/CLA-1 on the surface of the platelets, based on platelets obtained from patients with atherosclerotic disease. 6 Although further study is needed, a similar mechanism may be involved in the increase in the CE content in platelets from diabetic patients.
It has been reported that increased lipid content in platelets enhances their reactivity. 3 The contribution of hyperactive platelets with increased platelet cholesterol to the progression of atherogenesis associated with coronary artery disease is speculated. Therefore, routine measurement of cellular cholesterol in platelets may allow us to take a more personalized therapeutic approach to prevent atherosclerotic cardiovascular disease in patients with high-risk factors.
DECLARATIONS
