Abstract
Background
Inadequate temperature affects the stability of intact parathyroid hormone (i-PTH) kits. Room temperature during transport modifies i-PTH results.
Method
Percent bound (%B/Bmax) and concentrations (pmol/L) of standards, controls (C1, C2) and pool from eight standard curves were divided into: group I (three curves from kits kept at room temperature for more than 48 h) and group II (five curves from kits kept at 2–8°C) during transport. i-PTH was measured using Scantibodies total i-PTH assay with RIAMAT-280.
Results
%B/Bmax for standards, C1 and C2 were significantly higher in group I versus II (P = 0.04). %B/Bmax for the pool were significantly lower in group I (P = 0.001). i-PTH pool concentration in group I was 51% lower (95% confidence interval, 47–53%, P = 0.001); differences were not significant for C1 (P = 0.25) and C2 (P = 0.57) in both groups.
Conclusion
Room temperature on i-PTH kit during transport alters the standard curve, resulting in a decrease in i-PTH. Using a pool as internal quality control allows the detection of these changes not detected by kit controls.
Introduction
Total intact parathyroid hormone (i-PTH) is an immunoradiometric assay (IRMA) manufactured by Scantibodies Laboratory (Santee, CA, USA).
The kit contains two PTH controls and seven standards. 1 It is stable at 2–8°C until the expiration date specified by the manufacturer. In the literature, there is only one reference dealing with the stability of PTH standards under different temperature conditions. 2
Our laboratory includes an aliquot of a human plasma pool 3 as an internal control. This pool is prepared from samples from different patients. Once the value of the i-PTH is known, plasma is separated into aliquots that are frozen at −20°C. 4
We noticed that pool results showed an important decrease within standard curves obtained from kits of the same lot number. However, the results of the two kit controls were within the range established by the manufacturer. That decrease could be due to degradation of PTH in the pool or degradation of kit components. We decided to investigate the transport conditions of the kits. One of the shipments had been made at room temperature, not respecting the conditions of temperature recommended by the manufacturer (2–8°C).
In the current study, we show the effect of room temperature on the i-PTH kit during transport and its consequences on i-PTH results.
Material and methods
Eight standard curves were prepared using the same lot number kits. Each curve included two controls with concentrations ranging from 3.8 to 5.3 pmol/L (control C1) and from 33.9 to 51 pmol/L (control C2), and one pool sample with concentrations ranging from 12.7 to 14.3 pmol/L.
Values of percent bound (%B/Bmax) and concentrations of standards, controls (C1, C2) and pool were divided into two groups (I and II). Group I included the values of three curves whose kits were kept at room temperature for more than 48 h and group II included values of five curves whose kits were kept at 2–8°C during transport.
The determination of i-PTH was performed by a second-generation IRMA (total intact Scantibodies assay) according to the manufacturer's protocol. 1 All samples were measured in duplicate on EDTA-plasma with an automated analyser (Byk-Sangtec RIAMAT-280, 75217 Birkenfeld, Germany) that performs automatic dispensing, washing and counting of the samples using a gamma counter calibrated to quantify iodine-125.
The Mann-Whitney test for non-parametric data was used to determine the significant differences in the concentrations of i-PTH and non-parametric Kruskal–Wallis test in the values of %B/Bmax for standard curves of groups I and II. P ≤ 0.05 was taken as the level of significance.
Results
The median of %B/Bmax of standards show a significant increase (P = 0.04) in group I compared with group II (Figure 1).
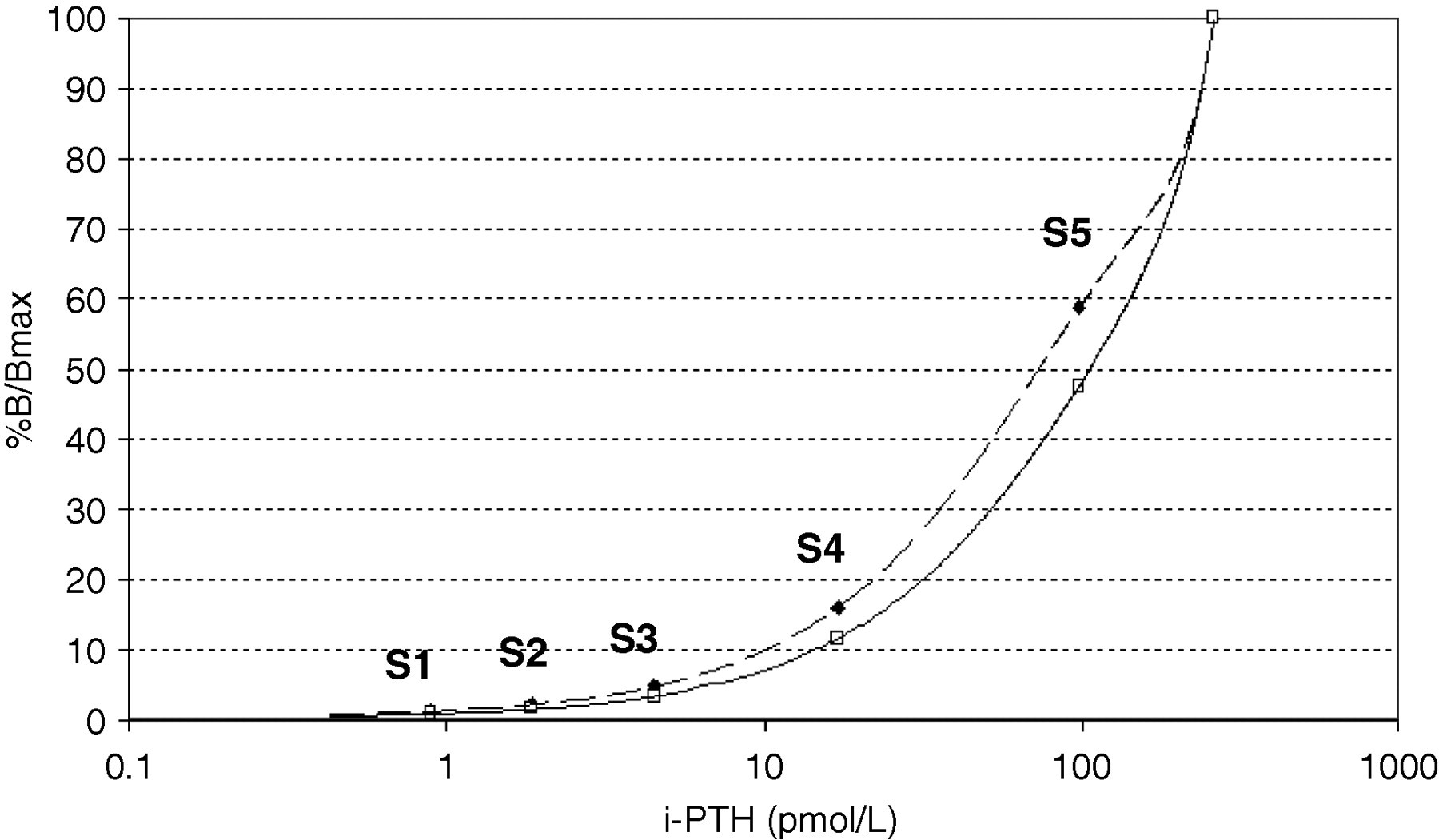
Median values of %B/Bmax of standard curves showing a significant increase (P = 0.04) in kits kept at room temperature for more than 48 h. Dashed line represents standard curve from kits kept at room temperature. Solid line represents standard curve from kits kept at 2–8°C. S1: i-PTH standard 0.9 pmol/L; S2: i-PTH standard 1.9 pmol/L; S3: i-PTH standard 4.5 pmol/L; S4: i-PTH standard 17 pmol/L; S5: i-PTH standard 98 pmol/L. i-PTH standard 0 and 259 pmol/L were employed to normalize the standard curve. i-PTH, intact parathyroid hormone
The median of % B/Bmax for C1 and C2 were significantly higher in group I than in group II (P = 0.04). By contrast, the median of %B/Bmax for the pool was significantly lower in group I (P = 0.001).
The median of i-PTH concentration for the pool was significantly higher in group II (13.5 pmol/L) than in group I (6.6 pmol/L), this decrease being 51% (95% confidence interval, 47–53%, P = 0.001). Differences were not significant for C1 (P = 0.25) and C2 (P = 0.57) in both groups (Table 1).
Values of percent bound (%B/Bmax) and i-PTH concentration (pmol/L) for control 1 (C1), control 2 (C2) and pool in group I (room temperature) and group II (2–8°C)
i-PTH, intact parathyroid hormone; IQR, interquartile range; CI, confidence interval
P = 0.04 for median values of %B/Bmax of C1, C2 and P = 0.001 of pool; P = 0.001 for median values of pool; P = 0.25 for median values of C1; P = 0.57 for median values of C2
Discussion
The kit for the quantitative determination of i-PTH is stable at 2–8°C until the expiration date. 1
Any change in the stipulations provided by the manufacturer, such as overcoming the temperature conditions, can affect the kit stability and promote the degradation of polyclonal antibodies, standards and controls. In the literature, there is only one study dealing with the deterioration of PTH in the standards stored at −70°C. No study has looked at the degradation at room temperature.
Our results show that kits kept at room temperature provided values of %B/Bmax of standards and controls higher than those obtained from kits kept at 2–8°C. This could be due to degradation of PTH standards and controls resulting in fragments of PTH recognized by two polyclonal antibodies. As this degradation affects standards and controls equally, the standard curve is apparently correct and controls are within the range set by the manufacturer.
The concentration of the i-PTH pool significantly decreased in group I (6.6 pmol/L) compared with group II (13.5 pmol/L). Degradation of this pool was ruled out when it was proved that i-PTH values obtained from kits kept at room temperature were significantly lower than those obtained from kits kept at 2–8°C, and values returned to the initial ones when transport conditions were correct.
This decrease in i-PTH pool concentration was in part due to a change in the standard curve caused by incorrect transport conditions (Figure 1).
From a clinical point of view, this difference in i-PTH values could potentially decrease by more than 50%, resulting in a shift from clearly abnormal to normal results.
In order to detect changes in i-PTH concentration when curves are apparently correct and results of the controls are in the range, the use of external controls or, as in our case, the use of a pool as an internal control is highly recommended.
Conclusions
Room temperature on the i-PTH kit during transport alters the standard curve, causing a decrease of up to 51% in the i-PTH results.
Using a pool as internal quality control allows the detection of these changes not detected by kit controls.
DECLARATIONS
