Abstract
Background
Renal stones in children, although rare, may be associated with morbidity and renal damage. Scottish children have a different ethnic composition and diet compared with paediatric populations previously studied. Urinary stone promoters include calcium, oxalate and urate. Postulated inhibitors include citrate and glycosaminoglycans (GAGs). We tested the hypothesis that Scottish paediatric stone-formers have higher excretion of urinary stone promoters (calcium/oxalate/urate) and/or lower excretion of stone inhibitors (citrate/GAGs) than children with isolated haematuria and controls.
Methods
In this case-controlled study, we measured creatinine, calcium, oxalate, urate, citrate and GAGs in random urine samples from 24 stone-formers (excluding inherited metabolic disorders), median age 10.2 (range 1.0–17.2) y; 25 patients with isolated haematuria, 6.3 (0.6–13.7) y; and 32 controls, 7.5 (0.8–14.7) y.
Results
Excretion of urinary promoters and inhibitors differed among stone-formers, haematuria and control groups for (median (range)): calcium (0.82 (0.02–2.19), 0.43 (0.08–2.65), 0.31 (0.04–2.12) mmol/mmol creatinine, respectively, P = 0.005), citrate (0.42 (0.13–0.72), 0.33 (0.05–0.84), 0.61 (0.11–1.75) mmol/mmol creatinine, P = 0.001), calcium:citrate ratio (1.68 (0.19–4.81), 1.30 (0.19–9.57), 0.54 (0.10–2.27) mmol/mmol, P < 0.0001) and the promoter:inhibitor ratio (calcium × oxalate)/(citrate × GAGs) (8.3 (1.0–82.5), 4.3 (1.2–69.5), 2.8 (0.3–13.2) mmol/g, P < 0.0001).
Conclusions
Scottish paediatric stone-formers had lower urinary citrate excretion and higher urinary calcium excretion, calcium:citrate ratio and promoter:inhibitor ratio compared with controls. Urinary calcium excretion and promoter:inhibitor ratio was also higher than children with isolated haematuria. Nevertheless, marked overlap between the stone-former and haematuria groups for promoter:inhibitor and calcium:citrate ratios suggests that some patients with isolated haematuria may be at future risk of urolithiasis.
Introduction
Renal stones in children are rare compared with adults, 1 but may nevertheless be associated with considerable morbidity and renal damage. It is important to identify risk factors (which may be different from those in adults and may be population specific) because this may allow specific interventions to reduce recurrence or prevent stone formation in those at risk.
The causes of urolithiasis have been extensively debated over the years with many conflicting theories suggested, but the reason why renal stones bind to and are retained in the kidneys of stone-formers remains unclear. Crystallization and growth of the stone is promoted by certain factors/constituents present in the urine. Reduced urine volume may allow saturation limits to be exceeded and hence solid particles to remain in the kidney. This process may be halted or impeded by the presence of certain ‘protective’ factors.
Accepted urinary promoters of urolithiasis include calcium, oxalate, urate and cystine. These stone promoters all have low solubility allowing crystal formation in concentrated urine. Postulated urinary inhibitors include citrate and glycosaminoglycans (GAGs). Growing children have high concentrations of GAGs in their urine, reflecting high turnover of cartilage and other connective tissues during growth. This may help to protect them against stone formation.
Previous studies in different paediatric populations have suggested that an increased urine calcium or oxalate, 2,3 lower levels of protective factors, 3–9 and/or imbalance between urinary promoters and inhibitors 5,7,9–12 increase the likelihood of stone-forming.
There have been no previous studies on urinary risk and protective factors for paediatric urolithiasis in the UK. Scottish children have a different ethnic composition and diet compared with paediatric populations previously studied. This retrospective, case-controlled study aimed to test the hypothesis that, after exclusion of patients with inborn errors of metabolism that cause a predisposition to urolithiasis, Scottish paediatric stone-formers have higher excretion of urinary stone promoters (calcium/oxalate/urate) and/or lower excretion of stone inhibitors (citrate/GAGs) than children with isolated haematuria and age-matched controls.
Subjects and methods
Subjects
Three subject groups recruited from patients attending a Scottish tertiary paediatric referral centre were studied. Diet was unrestricted in all groups.
The stone-former group consisted of patients with a history of confirmed urolithiasis attending outpatient clinics or admitted to hospital for investigation or lithotripsy. All stone-formers were screened for cystinuria by quantitative ion-exchange amino acid chromatography. Patients with cystinuria (increased excretion of cystine, lysine, ornithine and arginine) were excluded from the study. Additionally, stone-formers with >90% calcium oxalate stones or markedly increased oxalate excretion were screened for primary hyperoxaluria by organic acid analysis (gas chromatography-mass spectrometry).
The haematuria group consisted of patients with isolated haematuria attending outpatient clinics. Patients with a history of nephrocalcinosis or urolithiasis were excluded from this group.
Outpatient controls were recruited from a relatively healthy paediatric outpatient population. Exclusions from this group were: haematological tumours, inborn errors of the distal nephron, X-linked hypophosphataemic rickets, renal dysfunction, severe illness, history of urolithiasis or nephrocalcinosis, or haematuria.
The study was approved by the Lothian Local Research Ethics Committee. Written informed consent was obtained from all parents or guardians (and where appropriate from the children) in the control group. The stone-former and haematuria groups were anonymized to protect patient confidentiality.
Power calculations from previous studies indicated that between five and 21 subjects were required in each group to achieve 80% power and two-tailed significance of 0.05 for calcium, 5 citrate 8 and GAGs. 5,6 No suitable studies were found for oxalate or urate.
Samples
Random (un-timed) urine samples were split into two aliquots. One aliquot was acidified to pH <2 (for the analysis of calcium and oxalate), and both were stored at −70°C until further analysis.
A few samples were insufficient for analysis of oxalate (n = 2, 2, 1 in the stone-former, haematuria and control groups, respectively) and/or GAGs (n = 1, 3 in the stone-former and haematuria groups). In addition, GAGs were not analysed in four samples in which creatinine was <1.0 mmol/L because positive interference may occur in this assay in very dilute urine samples (unpublished observations).
Analytical methods
Calculus analysis was by Fourier transform infra-red spectroscopy. 13
Urine calcium, creatinine and urate were analysed on the Roche Modular P analyser (Roche, Lewes, UK) using established laboratory methods (cresolphthalein complexone, Jaffe and uricase/peroxidase, respectively) according to the manufacturer's instructions. Between-batch coefficients of variation (CV) were 3.5%, 3.6% and 1.8% at 1.5, 7.1 and 0.5 mmol/L for each assay, respectively. Oxalate was analysed on the Fusion analyser (Ortho Clinical Diagnostics, Buckinghamshire, UK) by an established oxidase/peroxidase laboratory method linked to formation of an indamine dye. 14 Between-batch CV was 2.6% at 0.29 mmol/L. GAGs were measured by an established laboratory method involving binding of dimethylmethylene blue at alkaline pH. 15 Between-batch CV was 3.2% at 63 mg/L.
A commercial kit for manual analysis of citrate in foodstuffs was obtained from R-Biopharm Rhone (Glasgow, Scotland). The method involves conversion of citrate to oxaloacetate and acetate by citrate lyase, followed by a series of linked enzymatic reactions resulting in NAD production, measured at 340 nm. 16 The method was developed and validated for automated analysis of urinary citrate on the Roche Modular P analyser. The method was linear to 2.5 mmol/L. Samples with citrate concentration >2.5 mmol/L were appropriately diluted with deionized water and re-analysed. Mean recovery was 99% (range 96–100%). Within run CV was 0.5% at 1.00 mmol/L. Between run CV was 2.1% and 0.4% at 0.94 and 1.88 mmol/L, respectively.
Data analysis
All urinary analytes were expressed as a ratio to urine creatinine to correct for variations in urine concentration. Two additional ratios were calculated: (1) the calcium:citrate ratio and (2) the promoter:inhibitor ratio (calcium × oxalate)/(citrate × GAGs). These calculated ratios are both independent of creatinine excretion, which is an age-dependent variable in children. Urate was not included in the ratios because, although it is a theoretical stone promoter, it has not been shown to be a significant risk factor in any previous paediatric study.
In the control group, calcium, urate, oxalate and GAGs showed significant departures from a Gaussian distribution (P < 0.02, Shapiro–Wilk W test) while citrate showed a tendency to a non-Gaussian distribution (P = 0.06). Non-parametric statistics were therefore used for statistical analysis. Data were expressed as median (range). Kruskall–Wallis analysis of variance (ANOVA) was used for comparison of the three subject groups. If ANOVA indicated a significant difference among subject groups, Mann-Whitney U tests were then used for post hoc paired comparisons between the three groups. Spearman rank correlation was used to explore relations between variables. Statistical significance was accepted at P < 0.05 (2-tailed). All statistical calculations were performed using the Analyse-it™ software package, Version 1.71.
Results
Twenty-four stone-formers, nine girls and 15 boys, median age 10.2 (1.0–17.2) y; 25 patients with haematuria, 15 girls and 10 boys, median age 6.3 (0.6–13.7) y; and 32 paediatric controls, 19 girls and 13 boys, median age 7.5 (0.8–14.7) y participated in the study.
Renal calculi were available for analysis in 14/24 (58%) stone-formers. Median calculus weight was 107 mg (range 6–2113 mg). Two patients had >90% calcium oxalate stones. Both had normal oxalate excretion for age (0.029 and 0.048 mmol/mmol) and both had normal organic acid profiles, ruling out primary hyperoxaluria. One patient had a 100% calcium phosphate stone. Five patients had mixed calcium phosphate (12–85%) and magnesium ammonium phosphate stones. Four patients had mixed calcium oxalate (3–87%) and calcium phosphate stones, of whom three had normal oxalate excretion (0.024, 0.037 and 0.050 mmol/mmol), and one (in whom calcium oxalate comprised only 3% of the calculus) had slightly increased oxalate excretion for age (0.097 mmol/mmol) but a normal organic acid profile, ruling out primary hyperoxaluria. One patient aged 1.0 y had a mixed magnesium ammonium phosphate (90%) and ammonium urate (10%) stone. One patient had a mixed urate (60%) and ammonium urate (40%) stone but normal urate excretion for age (0.47 mmol/mmol).
Urinary excretions of calcium, urate, oxalate, citrate and GAGs, together with the calcium:citrate ratio and the promoter:inhibitor ratio [(calcium × oxalate)/(citrate × GAGs)], are shown in Table 1. Excretions of urate, oxalate and GAGs were not significantly different among the three groups (P ≥ 0.18). Only two stone-formers had oxalate excretions greater than the range observed in controls. One with moderately increased oxalate excretion (0.113 mmol/mmol) had a stone composed of 85% calcium phosphate and 15% magnesium ammonium phosphate. One with markedly increased oxalate excretion (0.260 mmol/mmol) had a stone composed of 64% magnesium ammonium phosphate and 36% calcium phosphate and had a normal organic acid profile, ruling out primary hyperoxaluria. Calcium and citrate excretion were both significantly different among the three groups (P = 0.005 and 0.001, respectively), and both the calcium:citrate ratio and the promoter:inhibitor ratio showed highly significant differences among groups (P < 0.0001, Figures 1a and b).
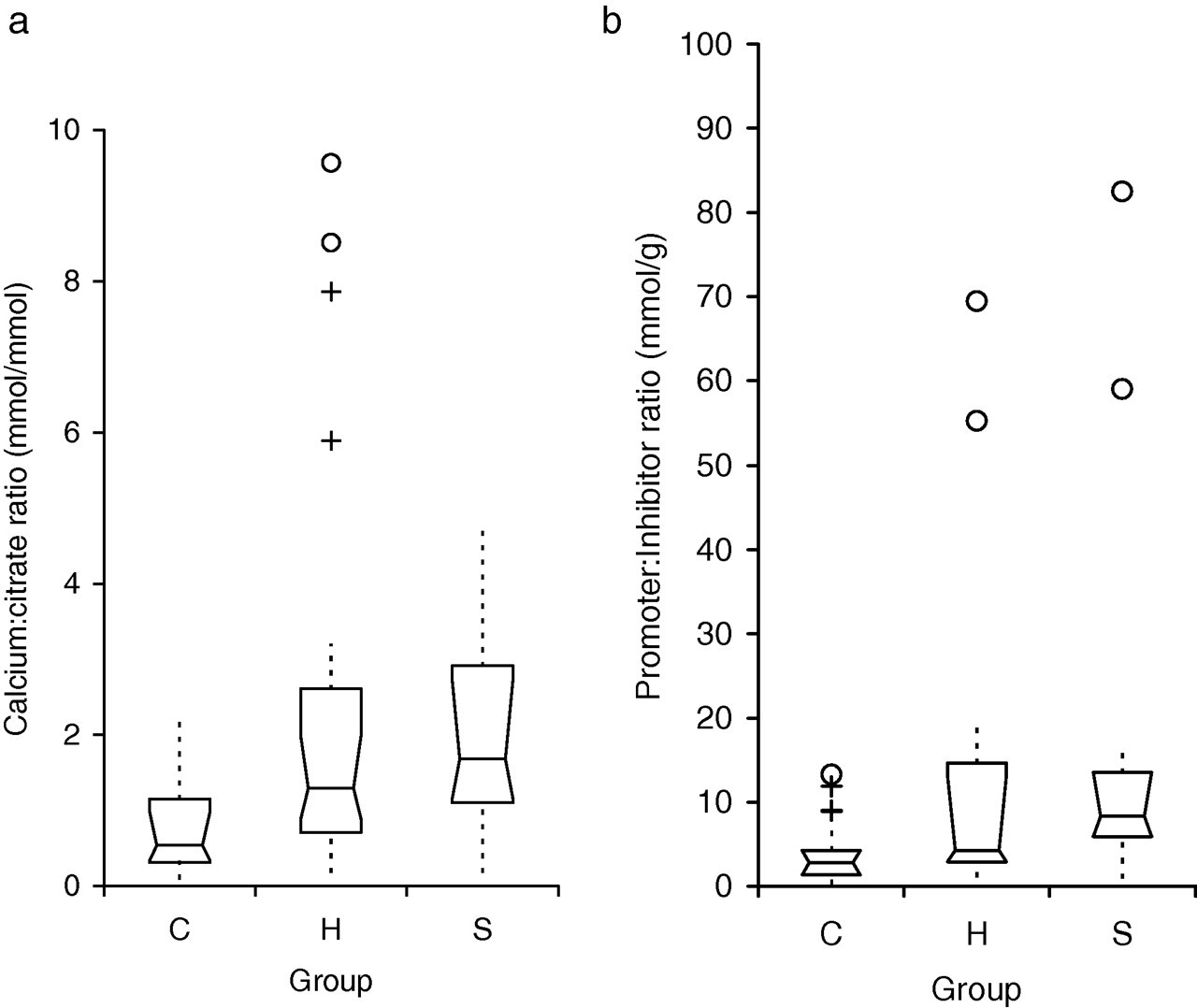
Box-Whisker plots comparing (a) calcium:citrate ratios (mmol/mmol) and (b) promoter:inhibitor ratios in the three study groups. Promoter:inhibitor ratio = [(calcium × oxalate)/(citrate × GAGs)] mmol/g Groups: C = control group, H = haematuria group, S = stone-former group [The notched box indicates the median, 25th centile and 75th centile. The dotted line connects the nearest observation within 1.5 interquartile ranges (IQR) of the lower and upper quartiles. Crosses (+) and circles (○) indicate observations more than 1.5 and 3.0 IQRs, respectively from the quartiles
Stone promoters (calcium, urate and oxalate), stone inhibitors (citrate and GAGs), calcium:citrate ratio and promoter:inhibitor ratio (calcium × oxalate)/(citrate × GAGs) in paediatric stone-formers compared with children with isolated haematuria and controls
Ca, calcium; Cr,creatinine; Ur, urate; Ox, oxalate; Cit, citrate; GAGs, glycosaminoglycans
Data are expressed as median (range)
Pairwise comparisons between groups (see Subjects and Methods): a P < 0.01, b P ≤ 0.001, c P < 0.05 versus controls. d P < 0.05 versus haematuria group
Note: A few samples were insufficient for analysis of oxalate (n = 2, 2, 1 in stone-former, haematuria and control groups, respectively) and/or GAGs (n = 1, 3 in stone-former and haematuria groups). GAGs were not analysed in four samples in which creatinine was <1.0 mmol/L because positive interference may occur in very dilute urine samples. See Methods
Post hoc pair-wise comparisons between groups indicated that, compared with controls, stone-formers had significantly lower citrate excretion (P = 0.005) and significantly higher calcium excretion (P = 0.002), calcium:citrate ratio (P ≤ 0.0001) and promoter:inhibitor ratio (P < 0.0001, Table 1). In the haematuria group, citrate excretion was also significantly lower (P = 0.001), and the calcium:citrate and promoter:inhibitor ratios were significantly higher (P = 0.013, 0.002 respectively) than in controls, although calcium excretion showed no significant difference between these two groups (Table 1).
Comparing the stone-formers with the haematuria group, both groups had similar citrate excretion but calcium excretion was higher in the stone-formers (P = 0.02, Table 1). Calcium:citrate ratio was similar in the two groups, with some individuals in the haematuria group having ratios even higher than in the stone-formers (Figure 1a). Although the promoter:inhibitor ratio was significantly higher in the stone-former group than in the haematuria group (P = 0.04, Table 1), there was also a large overlap in values between the two groups (Figure 1b).
In the control group, there was no significant difference between males and females for any individual urinary constituent, calcium:citrate ratio or promoter:inhibitor ratio (P > 0.19), indicating that excretion was not related to gender.
Age was not significantly different among subject groups (P = 0.18). Within the control group there were significant inverse correlations between age and the individual urinary constituents calcium, urate, oxalate, citrate and GAGs, all expressed as ratios to creatinine (r s = −0.45, −0.76, −0.38, −0.70 and −0.82, P = 0.01, <0.0001, 0.03, <0.0001 and <0.0001, respectively), indicating the strong relationship of these ratios with age.
There was no relationship between the calcium:citrate ratio or the promoter:inhibitor ratio and age in the control group (P = 0.82 and 0.35, respectively). In contrast to the control group, older patients in the haematuria group tended to have a higher promoter:inhibitor ratio (r s = +0.51, P = 0.02) and two patients aged 10.9 and 11.6 y had particularly high ratios of 69.5 and 55.2, respectively (Figure 1b). Calcium:citrate ratio was not age-related in the haematuria group (P = 0.82). Stone-formers themselves showed no correlation between age and calcium:citrate ratio (P = 0.11) or promoter:inhibitor ratio (P = 0.24).
Discussion
We have established that in a Scottish paediatric population, urine calcium:creatinine ratio was higher, citrate:creatinine ratio was lower, calcium:citrate ratio was higher and promoter:inhibitor ratio was higher in the urine of stone-formers compared with controls.
Our finding of significantly higher calcium excretion in stone-formers agrees with two previous studies performed in the USA and Hungary. 2,17 Although other investigators did not report a significant difference, 4,6,7,10 three of these studies were carried out in Turkey, an area endemic for bladder (infection) stones whose composition differs from upper urinary tract calculi. Endemic bladder stones are common in developing countries and are associated with a low protein, high cereal diet. Improvement in nutrition tips the balance in favour of upper urinary tract stones. 18,19 Our observation of higher calcium excretion in stone-formers than in controls, in contrast to the lack of significant difference in the Turkish studies, may reflect higher milk intake beyond infancy in a northern European paediatric population than in Turkish children. A Westernized trend for excess dietary salt and animal protein may also lead to an increase in urinary calcium and increased risk of stone formation. 20 Kaneko has suggested that Japan's low prevalence of paediatric urolithiasis could be partially attributed to low dietary intake of calcium and sodium. 21 While the recording of dietary factors was beyond the scope of this study, with the drinking of cow's milk freely available and encouraged in Scotland, including provision in Scottish schools, and the tendency for a high salt intake in the Scottish diet, the risk of hypercalciuria and hence calcium-based calculi is likely to be higher than in populations with lower intake of calcium and salt. Unfortunately it was not possible in our study to assess sodium intake indirectly because: (1) 24 h urine collections for sodium measurement are not practical in children and incomplete collections may give misleading results, (2) although fractional excretion of sodium in ‘spot’ urine samples may give a surrogate estimate of recent sodium intake, it was deemed unethical to collect the paired blood samples required for its calculation from healthy control children.
Citrate prevents crystallization of calcium salts by coating the surface and binding to calcium ion, forming a soluble complex, thereby reducing urinary saturation of the calcium salt. This in turn inhibits nucleation and crystal growth. 4 The binding allows the stone salts to stay in solution at much higher urine concentrations and thus be removed from the kidney more effectively. 22 Low levels of urinary citrate therefore may be a significant cause of renal stones. 23 Our observation of hypocitraturia in paediatric stone-formers has previously been described by several authors, 3,4,7,8 although others, 5,10,24 have failed to show a difference in citrate excretion compared with controls.
There are conflicting reports of the role of hyperoxaluria as a risk factor for urolithiasis in children. Few of the stone-formers in our study in whom stone analysis was available had predominantly calcium oxalate stones. In children who did have calcium oxalate stones or moderately/markedly increased oxalate excretion, primary hyperoxaluria was ruled out by organic acid analysis. Our observation of similar oxalate excretion in stone-formers and controls is in agreement with some, 4,6,10 but not all, 3,7 previous reports. It may be that relatively low fruit and vegetable intake in the Scottish diet may be a factor, but this would require confirmation in a prospective study relating dietary intake to oxalate excretion.
In our study, urate excretion was also similar in stone-formers and controls. To our knowledge, no previous study has shown urinary urate measurement to be significantly different in paediatric stone-formers. Urate stones represent a small percentage of the total paediatric stones formed (4–8% in Europe). 23 Only one of the stone-formers in our study had a predominantly urate stone. Urine urate excretion was normal for age in this patient but she was on a ketogenic diet for severe epilepsy and this may have resulted in chronically low urine pH, causing a predisposition to urate stone formation. Urinary pH must be measured on very fresh urine as a point-of-care test and was beyond the scope of our study.
GAGs (specifically heparan sulphate) are thought to inhibit calcium oxalate crystal growth and aggregation by binding to the crystal. 25 Two previous studies reported lower excretion of GAGs (expressed as a ratio to creatinine) in stone-formers compared with controls, 5,6 but a third found no difference between the two groups. 17 GAGs are very age-dependent in children, being higher in infancy and decreasing with age, 26 and therefore controls must be very closely age-matched to avoid systematic biases. In our study, we found no significant difference in GAG:creatinine ratio between stone-formers and controls, in agreement with Harangi et al. 17 It is possible that, as suggested by Harangi et al., the degree of sulphation of GAGs, and not their total concentration, is more important, with heparan sulphate being the crucial protective factor. 27 Because heparan sulphate is only a minor component of the GAGs present in human urine (in which chondroitin sulphate predominates), measurement of total GAGs would not be expected to discriminate between stone-formers and controls.
Magnesium measurement was not performed in our study. Its role as a possible protective factor against urolithiasis in children is controversial. Although two studies in Turkey and Argentina have reported low urinary magnesium excretion in some children with urolithiasis, their definition of hypomagnesuria was based on published literature values for different populations and neither study had a control group. 28,29 Most studies that have included a control group have reported no difference in magnesium excretion between stone-formers and controls. 3,6,7,17
Several previous studies, while finding little or no difference between paediatric stone-formers and controls for individual urinary constituents, have reported statistical differences in the ratios of promoters to inhibitors in various combinations. 5,9–12 Our finding of a higher calcium:citrate ratio in stone-formers than in controls is in agreement with two earlier studies. 5,12 Other promoter:inhibitor ratios that have been calculated by previous investigators are oxalate/(citrate × GAGs) 10,11 and oxalate/citrate. 9 The promoter:inhibitor ratio calculated in our study combined the most important risk and protective factors for urolithiasis identified by previous studies in children, namely (calcium × oxalate)/(citrate × GAGs). One advantage of both the calcium:citrate ratio and the (calcium × oxalate)/(citrate × GAGs) ratio is that both are independent of creatinine excretion, which is highly dependent on age and muscle mass. We found that the promoter:inhibitor ratio incorporating these four parameters was much higher in stone-formers than in controls, suggesting that this combined ratio may be a good indicator of the risk of urolithiasis in a Scottish paediatric population.
Haematuria is a sign of renal damage, including that caused by hypercalciuria and/or nephrolithiasis, although only a small proportion of children with haematuria have stones. It was therefore considered to be important to establish if risk factors that differentiate stone-formers from controls also differentiate stone-formers from patients with isolated haematuria not associated with urolithiasis. Only one previous study, to our knowledge, has included children with isolated haematuria without stones as a comparison group. 12
In our haematuria group, calcium excretion was significantly lower than in stone-formers and did not differ from controls, although there were large overlaps in the range of calcium excretion in all three groups. However, citrate excretion in the haematuria group was lower than in controls and was similar to the citrate excretion observed in the stone-formers, consistent with the possibility that at least some patients with haematuria may be at higher risk of urolithiasis should promoter levels increase. Indeed, the calcium:citrate ratio of patients with haematuria was significantly higher than in controls, in agreement with Batinic et al. 12 Several individuals in the haematuria group had high calcium:citrate ratios exceeding those in the stone-former group. Like the calcium:citrate ratio, the ‘four constituent’ promoter:inhibitor ratio in the haematuria group was also higher than in controls (although lower than in stone-formers), suggesting that at least some individuals with isolated haematuria may be at increased risk of future urolithiasis. A prospective longitudinal cohort study would be required to ascertain whether individual patients with haematuria and persistently high calcium:citrate ratios or ‘four constituent’ promoter:inhibitor ratios eventually develop urolithiasis, and to determine which ratio gives a better prediction of risk.
In summary, our study showed that stone-formers from this Scottish paediatric population have a higher excretion of urinary calcium and a lower excretion of urinary citrate compared with a control group, and a higher excretion of urinary calcium compared with children with isolated haematuria. The study also showed that the calcium:citrate and the ‘four constituent’ promoter:inhibitor ratios in stone-formers were significantly higher than those found in controls, and the promoter:inhibitor ratio was also higher than in patients with isolated haematuria. Both the calcium:citrate and the promoter:inhibitor ratios showed marked overlap between stone-formers and patients with haematuria, suggesting that some patients with isolated haematuria may be at increased risk of future urolithiasis. Urinary GAG measurement appeared to have no value as a predictor of paediatric urolithiasis.
DECLARATIONS
