Abstract
Aim
To survey laboratories enrolled in the Royal College of Pathologists of Australasia (RCPA) Chemical Pathology Quality Assurance Programme (QAP) for vitamin A and E testing to determine differences between methods of analysis.
Methods
A detailed questionnaire covering the major aspects of serum vitamin A and E analysis was sent to all participating laboratories in 2007.
Results
Thirteen out of the 22 laboratories completed the questionnaire. Methods between laboratories showed a great deal of commonality. All respondents performed a liquid extraction step, which included the addition of an internal standard, followed by high-performance liquid chromatography (C18 columns with predominantly methanol-based mobile phases) with spectrophotometric detection. Major inter-laboratory differences were whether the sample was protected from light, the extraction solvents and ratios used, the drying down temperature used post-liquid extraction and choice of calibrator.
Conclusions
The questionnaire highlighted discrete methodological differences between laboratories. These findings provide direction to enable the Vitamins Working Party of the Australasian Association of Clinical Biochemists to further investigate the dispersion in results between participants of the RCPA QAP vitamin programme.
Introduction
Since 1999, the Royal College of Pathologists of Australasia (RCPA) Quality Assurance Programme (QAP) has conducted an assessment of analytical performance for vitamin analysis. 1 Analytes in this programme include vitamin A (retinol), vitamin E (α tocopherol), β-carotene and total carotenoids. Each month, participating laboratories analyse two samples alongside their patient samples. Twelve samples are analysed within a six-month cycle. The Australasian Association of Clinical Biochemists (AACB) Vitamins Working Party (VWP) 2 was formed to support this vitamin programme.
In 2003 the VWP distributed a short questionnaire to establish the general methods used for analysis in the participating laboratories. This questionnaire established that all laboratories performed vitamin A, E and β-carotene by chromatographic separation and spectrophotometric detection with some differences in sample handling between laboratories.
In 2003–2004 the VWP conducted homogeneity testing, reconstitution/solubility studies and material stability studies on the RCPA QAP vitamin material. 3 There were no significant observations made; however, minor modifications to the reconstitution and storage procedures were recommended.
In 2007, the VWP carried out a more detailed questionnaire specifically for vitamins A and E. The aim of the 2007 questionnaire was to determine specific differences in discrete steps in the analytical methods. The results from the 2007 questionnaire are reported here.
Materials and methods
A detailed vitamin A and E method questionnaire was sent to the 22 participants in the RCPA QAP vitamin programme.
This questionnaire examined:
Section A: Sample preparation
The addition of a stabilizer before or during processing, the ratio of sample to solvents in the extraction process, use of an internal standard, type of extraction tube used, and the dry-down temperature and the reconstitution solvent used. The 2003 questionnaire had previously examined lighting conditions the samples were subjected to.
Section B: Chromatographic analysis
Stationary and mobile phases and the detection system used.
Section C: Measurement of related analytes
Additional analytes in relation to vitamins A and E were requested.
Section D: Reporting
Details and sources of reference intervals, including age-related ranges.
Section E: Standardization
Whether the participant would be prepared to change to a common calibrator if one was introduced or recommended. The source of calibrators used is routinely requested as part of the programme method information.
Results
Thirteen of the 22 laboratories (59%) completed the questionnaire.
Section A: Sample preparation
Tubes
The majority (11/13) used a combination of borosilicate glass and plastic during their extraction/reconstitution steps. One respondent used glass only and the other used plastic only.
Stabilizer
Most (11/13) laboratories did not add a stabilizer. One laboratory used 0.01% butylated hydroxytoluene and the other 25% ascorbate solution during the extraction procedure.
Internal standard
Eleven of the 13 laboratories used a combination of retinol acetate and/or tocopherol acetate as their internal standard/s. Two laboratories used δ tocopherol.
Extraction
All laboratories used a liquid extraction technique. The use of ethanol as a precipitating reagent with hexane as the extraction solvent was the most common (n = 7) combination. Six of these seven laboratories used an equal ratio of sample to precipitating reagent while one laboratory used 1:2 ratio. There was no consensus within this group for the ratio of ethanol to hexane used; responses ranged from 1:2.5 to 1:15. Another laboratory used a combination of methanol and hexane. Alternative precipitating reagents were as follows: none (n = 1), ammonium sulphate (n = 1), ethyl alcohol (n = 1), isopropanol (n = 1) and ethanol + petroleum ether (n = 1). These were used in conjunction with isopropanol, 90% propanol, heptane/petroleum ether, 50:50 hexane/isopropanol and petroleum ether, respectively.
Concentration of extract
The dry-down temperature ranged from room temperature (n = 3) to 70°C (n = 1), with a median of 37°C. No relationship was evident between the temperature selected and the solvent used. The 2003 questionnaire had previously determined that most laboratories used a stream of nitrogen gas for concentration. One laboratory reported not drying down their extract.
Reconstitution
There was reasonable consensus between laboratories regarding the solvent used: ethanol (n = 6), methanol (n = 3), mobile phase (n = 2) and chloroform (n = 1).
Section B: Chromatographic analysis
All used high-performance liquid chromatography (HPLC) with either ultraviolet/visible (n = 11) or diode array detection (n = 2). Twelve laboratories used isocratic separation and one used gradient. All used C18 columns. The majority (n = 9) of mobile phases were 97.5–100% methanol. Others included acetonitrile, ammonium acetate, chloroform, ethanol, ethyl acetate, isopropanol, tetra-hydrofuran or triethylamine.
Vitamin A (λ max325 nm) was detected between 324 and 340 nm (mode = 325 nm). Vitamin E (λ max292 nm) was measured between 282 and 295 nm (mode = 294 nm).
Section C: Measurement of related analytes
Most (n = 11) laboratories do not report additional results. One laboratory also reported a vitamin E:lipid ratio. Another reported retinyl esters and retinol-binding protein with vitamin A results.
Section D: Reporting
Twelve laboratories responded to the questions on reference intervals (Figure 1). Half of these have one reference interval for each of vitamins A and E. Others provided age-related reference intervals for vitamin A (n = 6) and vitamin E (n = 8). Those with a lower limit of 0.7–0.8 μmol/L for vitamin A did not provide a paediatric range. One laboratory provided a literature-based pregnancy reference interval for vitamins A and E. 4
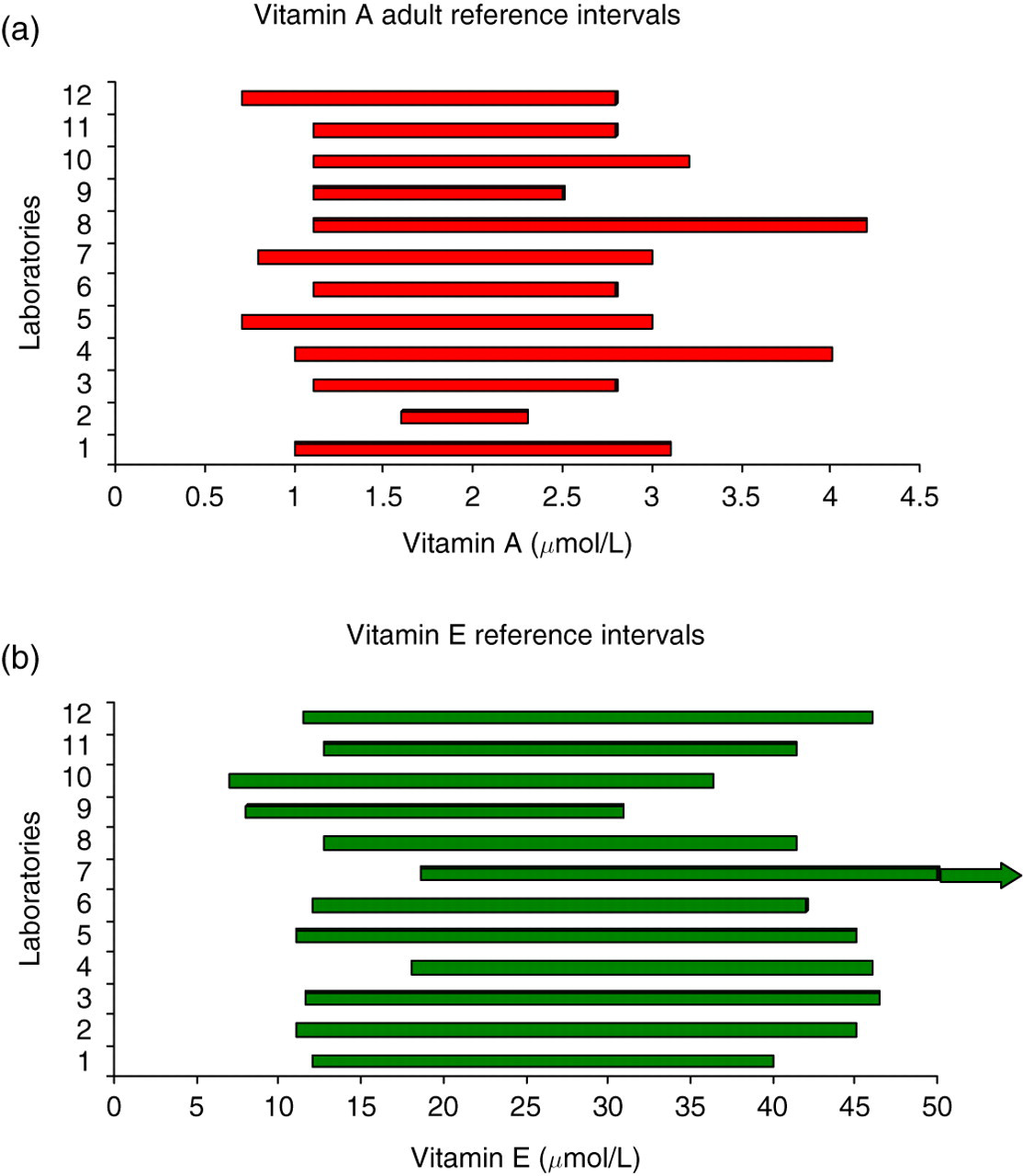
(a) Plasma/serum vitamin A adult reference intervals across laboratories. (b) Plasma/serum vitamin E adult reference intervals across laboratories. Laboratory 7 only reported a lower end range for vitamin E
Section E: Standardization
Ten of the 13 respondents supported the use of a common calibrator. Of the remaining three, one made no comment, another declined and the third responded yes, pending the inclusion of β-carotene in the material.
Discussion
There is little data in the literature comparing methods used in medical testing laboratories for vitamin A and E analysis. This survey has demonstrated considerable commonality between serum/plasma vitamin A and E methods, with all respondents performing liquid extraction sample preparation and analysis by HPLC (C18 columns with predominantly methanol-based mobile phases and spectrophotometric detection). Differences include whether the sample was protected from light, extraction solvents and ratios used, temperature for concentration of the extract and the calibrator used.
The questionnaire has served a useful purpose in highlighting discrete methodological differences between laboratories. The VWP plans to conduct further investigations to determine how these methodological differences contribute to variation in RCPA QAP results returned.
DECLARATIONS
