Abstract
Background
The concentration of N-terminal propeptide of type I procollagen (PINP) in the serum reflects the rate of type I collagen formation. Intact PINP assay measures the trimeric propeptide while total P1NP assay measures both trimeric and monomeric forms. In this study we compared these two assays emphasizing the possible differences.
Methods
Intact and total PINP were measured from serum in healthy Finnish blood donors (n = 34) and in the patients with chronic renal failure before and after haemodialysis (n = 39). In addition, the serum of a normal man, pooled hospital serum samples and the serum of a patient with haemodialysis treatment were fractioned by gel filtration and trimeric and monomeric forms were located. Fractions were lyophilized and intact and total PINP were measured in each fraction. Samples from bedridden geriatric patients (n = 173) were also measured using intact and total PINP assays and a degradation marker of type I collagen (ICTP).
Results
The correlation between intact and total PINP in controls was 0.89 and their PINP concentrations were similar. In haemodialysis or bedridden geriatric patients, the PINP methods gave significantly different results. In gel filtration studies, intact PINP hardly measured monomeric form even if its concentration was disproportionately increased in haemodialysis patients. In bedridden geriatric patients, the difference of total and intact PINP correlated significantly to degradation marker ICTP.
Conclusions
Difference between total and intact assays for PINP seem to reflect degradation of pN-collagen rather than denaturation of intact propeptide.
Introduction
Type I collagen is the most abundant collagen in our bodies and is predominantly found on mineralized bones. It is a product of two genes, COL1A1 on chromosome 17 and COL1A2 on chromosome 7, that encode two chains of the collagen molecule, called α1(I) and α2(I) chains. Type I collagen deposited in tissues is derived from type I procollagen which is synthesized by fibroblasts and osteoblasts. Type I procollagen contains at both ends aminoterminal and carboxyterminal propeptides that are cleaved off by specific proteases during collagen maturation. The mature collagen molecule is ready to assemble into the collagen fibril, while the propeptides are liberated into the intracellular space and most of it finally into the circulation. When one mole of collagen is produced one mole of the trimeric propeptides are produced. The specific by-products are synthesized stoichiometrically during the synthesis of type I collagen and thus they are true bone synthesis markers. 1
The N-terminal propeptide of type I procollagen is abbreviated as PINP. The molecular weight of trimeric propeptide is about 35,000 and its constituent chains 14,250 (proα1-chain) and 5500 (proα2-chain). Proα1-chain has two domains, globular domain that resembles von Willebrand factor C repeat and a triple helical domain. Proα2 is lacking globular domain and it has only triple helical domain. In human serum, PINP exists in two forms that can be separated with gel filtration, although their apparent elution positions are much larger than the actual molecular weights. 2 The larger antigen is the trimeric propeptide (intact PINP) and the smaller antigen represents monomeric form, which contains only single proα1-chain.
The two PINP forms have different catabolic routes since trimeric PINP is rapidly cleared from blood circulation by scavenger receptors on liver endothelial cells. 3 The clearance of the monomeric form of PINP has not been fully studied. However, it is probably cleared via kidneys, as was shown by the similar monomeric fragment of aminoterminal propeptide of type III collagen. 4 From human amniotic fluid, PINP can be isolated as fetal antigen 2 and is found mainly in monomeric form. 5 The proportion of monomeric PINP has been reported to be elevated in patients with chronic renal failure. 6,7 However, two publications indicate that intact PINP is not affected by the glomerular function with renal patients. 8,9
Currently, there are two commercially available assays, intact PINP from Orion Diagnostica and total P1NP from Roche Diagnostics. When developing the former assay, extra concern was aimed not to measure monomeric form and finally the cross-reactivity to the monomeric PINP was only 1.2%. 2 The latter assay was devised to measure both intact and monomeric forms since these authors suggested that the trimeric PINP degrades quickly to the monomeric form in vivo and therefore the measurement of both forms would be favourable. 10,11 The aim of this study was to compare the results of intact and total PINP concentrations in conditions where large differences are expected.
Research design and methods
Subjects
Serum samples of controls were from 34 apparently healthy Finnish blood donors (Finnish Red Cross; 26 men, 8 women; ages between 19 and 62 years) collected in April–May 2004. Serum samples of patients with chronic renal failure (n = 39) were obtained in Oulu University Hospital during May–June 2005 before and after haemodialysis. Geriatric patients (n = 173) were bedridden older long-term inpatients from Municipal Hospital in Helsinki, Finland and the serum samples were collected in September–October 2005. The inclusion criteria for them were age over 65 years, chronically impaired mobility, stable general condition and no medication (vitamin D supplements, glucocorticoids, antiepileptics, etc.) affecting calcium or bone metabolism. 12 All the samples were aliquoted and they were kept frozen at −20°C. Both intact PINP and total P1NP were measured in all the samples. In addition, serum cross-linked carboxyterminal telopeptide (ICTP) was measured from geriatric bedridden patients. 13 The studies were approved by the Ethical Committee of Oulu University Hospital or Helsinki University Central Hospital and conformed to the Declaration of Helsinki. All the patients gave a written informed consent. If the patient was severely cognitively impaired, the informed consent was obtained from the participant's surrogate or health-care power of attorney.
Gel filtrations of serum samples
Serum samples of a normal adult men, a patient with haemodialysis-treatment and pooled serum samples from Oulu University Hospital were fractioned by gel filtration at room temperature, using a column of 1.5 × 110 cm of Sephacryl™ S-300 (Amersham Pharmacia, Biotech AB, Uppsala, Sweden). The chromatographies were performed with 0.2 mmol/L (NH4)HCO3, pH 7.4, at a flow rate of 7.5 mL/h and 20 min fractions were collected. Each fraction was measured by radioimmunoassay for synthetic peptide (SP15), which detects both intact and monomeric forms. The synthetic peptide contains 18 amino acids (pQEEGQVEGQDEDIPPIYC) from the aminoterminal end of proα1-chain of PINP and it is analogous to assay of rat/mouse PINP published earlier. 14 The fractions containing the proα1-chain of PINP immunoreactive antigens were lyophilized, dissolved in 200 μL phosphate-buffered saline (PBS) and measured with intact and total PINP assays.
Intact PINP assay
Procollagen, intact PINP [125I] radioimmunoassay kit (Orion Diagnostica UniQ PINPTM, Espoo, Finland) was performed in accordance with the manufacturer's recommendations. All serum samples, calibrators and controls were measured in duplicate. Intra- and inter-assay coefficients of variation (CV) were 9.8–10.2% and 6.0–9.8%.
Total P1NP
Total procollagen type I aminoterminal propeptide was measured in single analysis by the electrochemiluminescence immunoassay technique using total P1NP reagents and an Elecsys 2010 analyzer according to the manufacturer's instructions from Roche Diagnostics (Mannheim, Germany). The intra- and inter-assay CVs were calculated by the manufacturer and reported to be 2.3–3.7% and 1.8–2.9% respectively. These analyses were performed on two separate days and control samples (PreciControl bone level 1, 2 and 3) gave CV values between 2.4% and 6.2%.
Cross-linked carboxyterminal telopeptide of type I collagen (ICTP)
ICTP antigen was measured in duplicates in the serum samples using the ICTP [125I] radioimmunoassay kit from Orion Diagnostica (UniQ ICTPTM, Espoo, Finland) according to the manufacturer's instructions. Intra- and inter-assay coefficients of variation were 2.8–6.2% and 4.1–7.9%, respectively.
Statistics
Statistical analysis was performed with Analyse-it for Microsoft Excel (Analyse-It Software Ltd, UK). For statistical analysis of haemodialysis effect, paired samples t-test was used. Correlation was tested by the Pearson correlation coefficient. Differences were considered significant at P < 0.05. Linear correlations and Passing–Bablok regressions 15 were used to calculate slopes and intercepts. If two methods are compared and they give similar results, the 95% confidential interval (CI) of slope should include value 1 and the 95% CI of intercept value zero (Table 1).
Linear correlations (r) and Passing–Bablok regressions of the total and intact PINP in different groups
*Significant difference between methods (P < 0.05)
Results
The correlation between intact and total PINP was 0.89 in healthy blood donors and the slope and the intercept were similar in both cases (Table 1 and Figure 1). However, the correlation in haemodialysis patients was only 0.77 and the 95% CI of slope was higher than value 1 and the 95% CI of intercept was below value 0 (Table 1). The average concentration of total P1NP before haemodialysis was about five times higher than intact PINP: intact PINP (mean ± SD) 95.2 ± 108.0 μg/L and total P1NP 470.9 ± 534.4 μg/L (Table 2). During the haemodialysis, the mean total P1NP increased by 53 μg/L from 471 to 523 μg/L (+11%, P = 0.0002) while mean intact PINP decreased by 6 μg/L from 95 to 89 μg/L (−6%, P = 0.0003). The correlation in geriatric bedridden patients was 0.82 and the both slope and intercept did not contain 1 or 0, respectively (Table 1). In these cases, the differences between total and intact PINP correlated significantly (r = 0.61, P < 0.0001) with serum ICTP concentrations (Figure 2). For this calculation, we excluded two patients who had very high concentration of total P1NP (500 and 597 μg/L) although their intact PINP were not increased (61.8 and 43.1 μg/L, respectively).
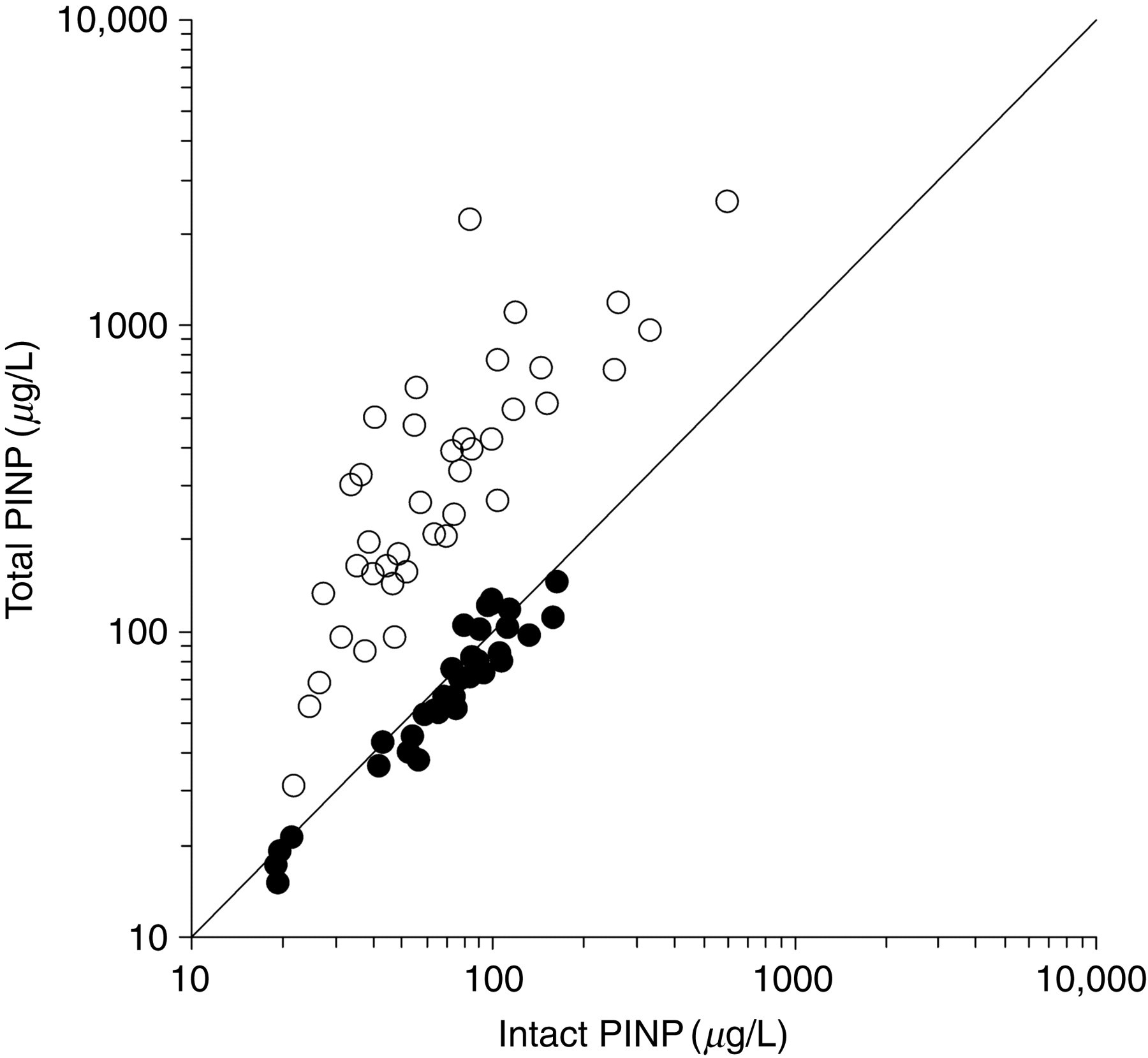
In healthy individuals (solid circles) the correlation between intact and total N-terminal propeptide of type I procollagen (PINP) is rather good (r = 0.89), but in patients with haemodialysis (open circles) the total P1NP is about five times higher compared with intact PINP
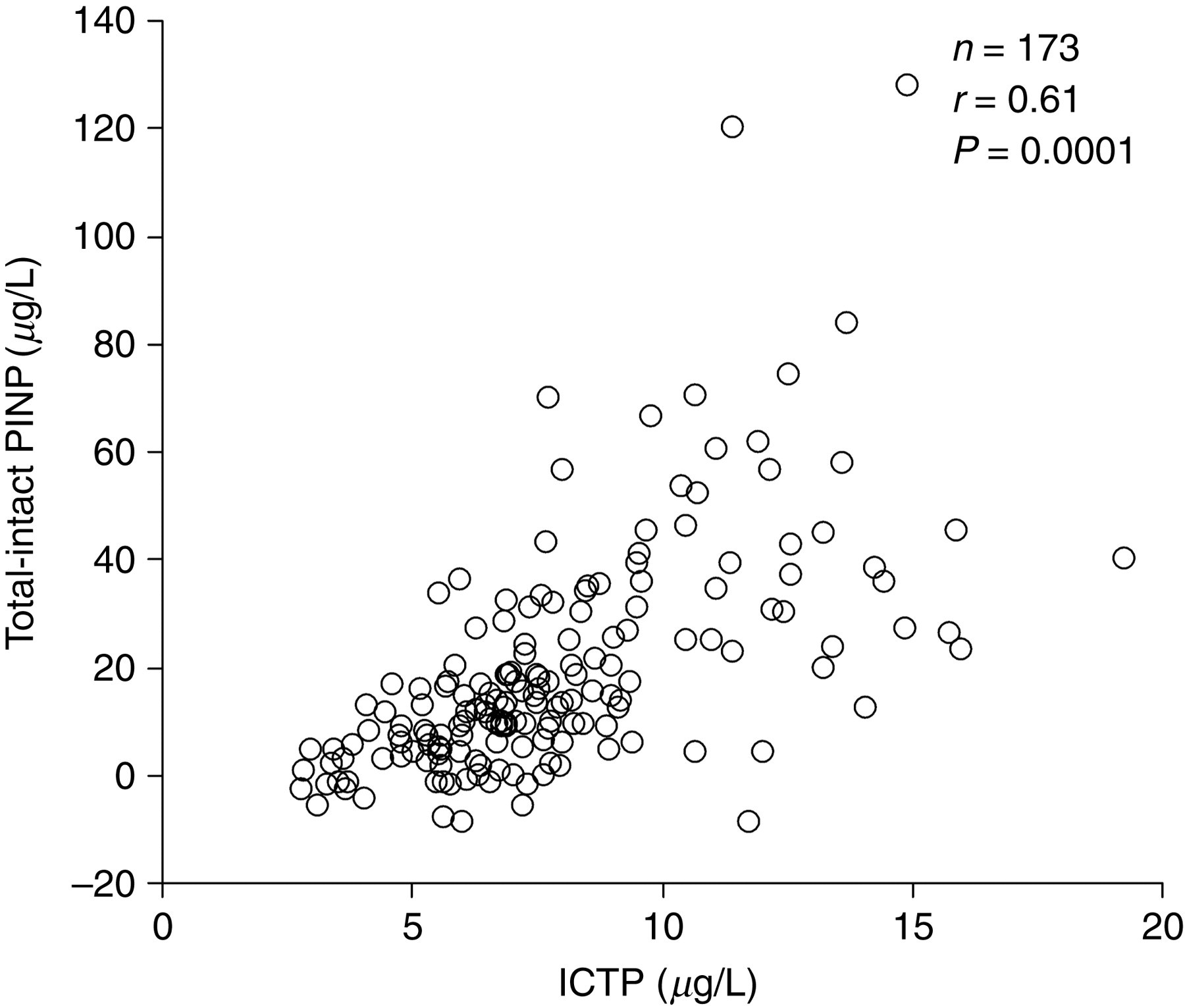
The difference between the total and intact N-terminal propeptide of type I procollagen correlated significantly with the concentrations of degradation products of type I collagen (ICTP) in the bedridden geriatric patients
Means, SDs and medians of the total and intact PINP in different groups
SD, standard deviation; PINP, N-terminal propeptide of type I procollagen
After localization of intact PINP and monomeric forms in gel filtration fractions, samples were lyophilized and dissolved in PBS and then both intact and total PINP were measured in each fraction (Figure 3). In a healthy male serum both assays gave similar results, but in hospital pool serum samples the monomeric form was much increased although the concentration of intact PINP was similar. In the patient with chronic renal failure, the monomeric form was disproportionately increased although the concentration of intact PINP was similar to previous samples (Figure 3).
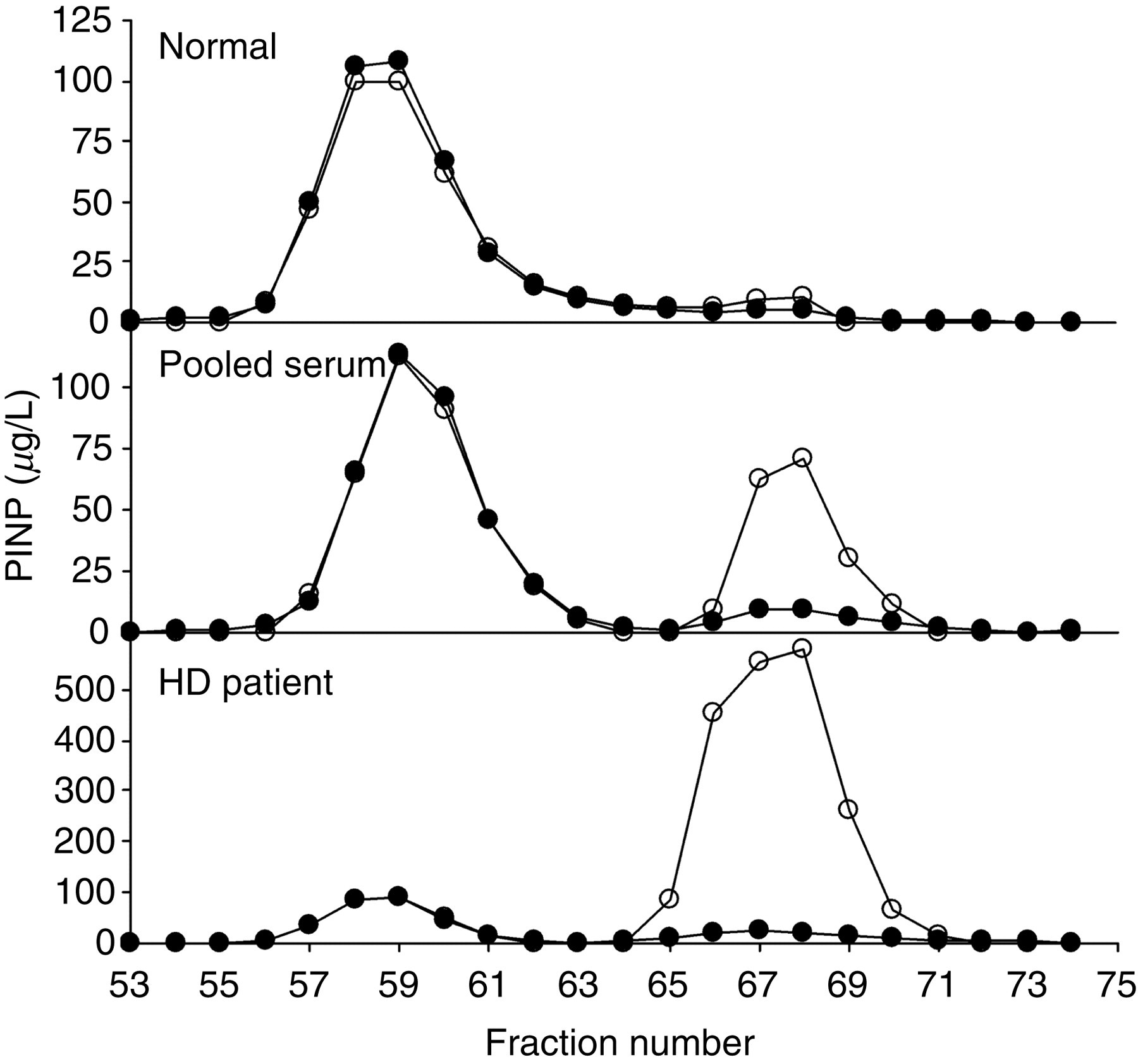
Serum samples of a normal men, a pooled serum sample from Oulu University Hospital and the patient serum before haemodialysis treatment were fractioned by gel filtration. In each fraction the concentrations showed the results with the total P1NP (open circles) and the intact PINP (solid circles)
Discussion
Two different assays for aminoterminal propeptide of type I procollagen are commercially available. The total P1NP assay measures both trimeric and monomeric forms of PINP antigens while the intact PINP measures only the trimeric form. It was previously reported that the intact form is readily denaturized by heat into monomeric form. 10 These authors purified the intact PINP propeptide from human amniotic fluid where the propeptide was very easily found in the monomeric form. They draw a conclusion that the propeptide itself is instable in biological fluids. We have a similar experience when we first isolated trimeric PINP from amniotic fluid by gel filtration. The partially purified trimeric PINP was soon found in the monomeric form. Only when we isolated the trimeric PINP from human ascitic fluids using several protease inhibitors, we were able to purify intact stable propeptide. It is possible that the amniotic fluid collected during delivery contains a lot of connective tissue degradation enzymes that can easily degrade the propeptide itself during purification process.
Previously, our laboratory has developed a similar assay for aminoterminal propeptide of type III procollagen (PIIINP) and in this case the concentration of monomeric form is much larger than that of trimeric form. 16 However, trimeric form of PIIINP is covalently bound to each other by disulphide bridges and it is not possible to fabricate monomeric form by the simple heat denaturation. Another explanation was that this monomeric form is derived from the degradation of tissue pN-collagens which is abundant in soft tissues. 17 Collagens are synthesized as procollagens which contains at both ends large propeptides, PINP in the aminoterminus and PICP in the carboxyterminus. 1 PICP is usually liberated first during entering to the extracellular space but PINP could stay for a while on the collagen proper. This partially processed procollagen is called pN-collagen. Based on this reasoning when we developed intact PINP assay, extra attention was directed not to measure the monomeric form.
In healthy individuals, the intact and total PINP gives similar results as reported previously. 11 However, in haemodialysis patients the results were very different. We found out that the increase was due to increase of the monomeric forms that cumulate in the serum when the kidneys are not functioning. Accumulation of the breakdown products caused by decreased kidney function is a normal phenomenon. Haemodialysis treatment did not have much affect on the intact or total PINP concentration as reported previously for both assays. 8,9 Indeed, haemodialysis cannot totally replace kidney since many aspects of normal kidney functions are missing such as degradation of small peptides in the primary urine. Also in hospital serum pool, we found out that the monomeric form was increased compared with healthy individuals. Most likely this is due to the fact that many patients in which this pool was collected have an illness that will affect connective tissue metabolism just as various cancers, wound healing, fibrotic conditions, severe sepsis, 18 etc. Could the accumulation of monomeric form be harmful in other conditions than haemodialysis patients as well? In this study, we tested bedridden old patients who most likely have an increased catabolism of connective tissue. We measured serum ICTP concentration in the same samples and observed that many of the tested patients had increased ICTP concentrations as reported previously in our laboratory. 19 ICTP concentration is an indication of increased catabolism of type I collagen. By substracting intact PINP value from total P1NP value, we should get an estimate of the circulating monomeric PINP. This difference between the two PINP assays correlated significantly to ICTP values indicating that monomeric form may originate from tissue collagen breakdown similarly to monomeric form of PIIINP.
Type I procollagen is synthesized both in bones and soft tissues and so also PINP are produced in these tissues. In bones, PINP is liberated before mineralization since there is no PINP in mineralized matrix. However, in soft tissues it is possible that there are some pN-collagens remaining. At least we have stained with immunohistochemistry PINP-positive cells in fibrotic conditions in skin. 20 ICTP reflects matrix metalloproteinase pathway of type I collagen degradation that occurs in soft tissue and also to minor extent in bones. Our conclusion is that the monomeric form reflects the degradation product of tissue pN-collagen rather than heat denaturation product of the circulating propeptide. This conclusion is based on the fact that monomeric form is increased both in patients with increased catabolic breakdown of type I collagen and in patients with reduced clearance of the monomer in the kidneys.
DECLARATIONS
