Abstract
Background
Neonatal hyperbilirubinaemia is a common treatable cause of brain injury. The treatment for this condition is phototherapy. The decision whether to use phototherapy is currently dependent upon serum bilirubin assay results. However, repeated blood sampling is not only traumatic but may also be a cause of anaemia in neonates. We evaluated a transcutaneous bilirubin assay method to determine whether it was suitable for routine use in preterm infants.
Methods
One hundred and eighty-three transcutaneous bilirubin measurements were taken contemporaneously with blood samples for laboratory measurement of serum bilirubin. The study was carried out with informed parental consent and approval by the local research ethics committee.
Results
The transcutaneous bilirubin method (BiliChek ®) exhibited a consistent positive bias compared with the laboratory bilirubin assay. Consequently, for a given detection rate, the transcutaneous method had a higher screen positive rate, i.e. more neonates would be given phototherapy if transcutaneous bilirubin results were used to decide. There was a margin of safety in the transcutaneous bilirubin assay calibration.
Conclusion
The BiliChek transcutaneous bilirubin assay is a safe alternative to laboratory bilirubin assay in deciding whether to give preterm neonates phototherapy.
Introduction
Neonatal hyperbilirubinaemia is a common occurrence 1 but relatively few affected infants require intervention. Whatever the cause, total serum bilirubin (TSB) levels above defined thresholds warrant phototherapy to prevent the development of kernicterus.
Clinical evaluation of hyperbilirubinaemia involves visual inspection of the skin, sclera and mucous membranes to identify jaundice. However, quantification of TSB based on visual assessment of the depth of jaundice is subjective and inaccurate, and confounded by various factors such as skin colour and haemoglobin. Various methods have therefore been developed to aid non-invasive diagnosis of hyperbilirubinaemia and approximation of TSB levels. They include cephalocaudal staging of jaundice and transcutaneous bilirubin (TcB) reference devices. Previously, due to limited accuracy TcB devices have only been considered useful as screening rather than diagnostic tools for determining hyperbilirubinaemia. They include the Ingram Icterometer, Chromatics Colormate III, Minolta AirShields Jaundice Meter, 2,3 Bilimed and BiliChek ®. 4,5
TSB measurement in the laboratory is considered the gold standard method of bilirubin estimation. It requires a blood sample from the patient and hence a needle prick, which is painful and when repeated can lead to anaemia. Frequent measurement of TSB in babies with bilrubin concentrations below the treatment threshold is inappropriate because it requires unnecessary blood sampling. Furthermore, waiting for results may delay the discharge of both the mother and baby from the hospital.
BiliChek works by directing white light into the skin of the newborn and measuring the intensity of the specific wavelengths that are returned. The light reflected from the skin of the neonate is analysed by the BiliChek using a proprietary algorithm to generate a serum bilirubin measurement. BiliChek is claimed to be accurate regardless of skin colour by compensating for the confounding effects of dermal maturity, melanin and haemoglobin on spectral reflectance. 6 BiliChek is also claimed to have high accuracy and reproducibility across different ethnic groups as well as in term/near-term infants. 7 It has been suggested that the BiliChek device is accurate enough to be a substitute for TSB sampling in these groups of infants and could possibly replace the laboratory measurement of TSB. 8,9 However, there is little of data on its use and efficacy in the preterm infants (especially in babies <30–35 weeks gestation). Two studies 10,11 conclude that transcutaneous bilirubin measurement is safe and can reduce TSB testing. However, there remains confusion in the literature, for example, Thayyil and Marriott 7 cite Rubaltelli et al. 9 as examining infants >30 weeks, when they in fact examined infants >35 weeks. The accuracy of BiliChek at higher bilirubin values (>200–250 μmol/L) with and without phototherapy remains to be seen in this group of infants.
Objective
To look at the agreement between two different methods of measuring total bilirubin using BiliChek (TcB) and TSB in babies <35 weeks gestation with or without phototherapy in order to determine whether TcB measurements would alter clinical practice.
Methodology
Setting
This prospective observational study was carried out at Queen's Hospital, Burton Upon Trent, an average sized district general hospital with approx 3500 deliveries per year. In our neonatal unit (NNU), approximately 150 preterm babies are admitted each year. According to our current policy, all babies born at <35 weeks gestation and/or birth weight of <1.8 kg (or <2 kg if intrauterine growth retarded) are admitted to our NNU. In addition, neonates requiring resuscitation at birth, or those having seizures, breathing/feeding difficulties, etc. are also admitted. A significant number of them develop jaundice during the first couple of weeks of life. The majority (but not all) of these babies require blood tests on admission and then routinely on a weekly basis or based on clinical judgement such as in response to jaundice or infection etc. Most will have TSB measurements at some point during their stay in NNU.
Time period
The study was conducted over a period of one year (July 2007 to June 2008).
Inclusion criteria
All babies less than 35 weeks gestation admitted to NNU during the study period.
Exclusion criteria
Infants requiring exchange transfusion.
Ethics approval
Both ethics and Research & Development committees approvals were obtained before starting this study.
Consent
Written information leaflets were given to parents of all babies admitted to NNU, which fulfilled the inclusion criteria and were invited to enrol for this study. Subsequently, written consent was obtained from those willing to participate.
Method
Where a clinical decision was made to undertake TSB measurement, a simultaneous TcB measurement using BiliChek was also obtained. The decision to commence phototherapy was made by the clinicians based on the TSB result. The threshold used depends on gestational age [threshold = (Gestation × 10) − 100]; thus, for example the threshold for a 35-week gestation infant would be 250 μmol/L. If the baby did not require phototherapy, he/she was monitored clinically and any further decision to repeat TSB was made on clinical grounds by the medical and/or nursing staff. If, however, the baby required phototherapy, the TSB was repeated in 6–8 h (to ensure a downward trend and improvement in bilirubin levels) and then 12–24 h, till a decision was made to stop phototherapy. TSB was repeated 8–12 h after stopping phototherapy to ensure that the bilirubin levels were still below the treatment level (and there was no significant rebound increase in its level).
To assess precision, BiliChek TcB measurement was assessed 5× in succession in 10 infants.
TSB measurement
TSB was measured in the laboratory using a standard diazo method (Olympus AU640; Olympus Diagnostics, Watford, Herts, UK).
TcB measurement
When samples for TSB measurement were collected, a contemporaneous TcB measurement was also carried out within 15–30 min by applying BiliChek (Respironics Inc., Chichester, UK;
Simultaneous multiple measurements were taken if the neonate required repeated TSB measurements, e.g. during/after phototherapy. The area of forehead used for TcB was not exposed to direct sunlight and/or phototherapy. If an infant required phototherapy, BilEclipse™ (phototherapy protective patch) was positioned over the measurement site prior to starting phototherapy. All phototherapy lights were turned off while a BiliChek measurement was taken. The BilEclipse protective patch was opened and after taking the BiliChek measurement, the flap was closed before recommencing phototherapy. Once phototherapy was discontinued, the BilEclipse flap was removed but subsequent BiliChek measurements were taken from the site on the forehead that had not been exposed to the phototherapy lights. Care was taken to avoid skin areas with bruising, birthmarks, haematomas or excessive hairiness. Gestation, birth weight, postnatal age and ethnicity were recorded.
Legal status of device
BiliChek was approved for use by the Food and Drug Administration (USA) on 22 March 2001. The device is CE-marked, indicating compliance with the Medical Device Directive 93/42/EEC of 14 June 1993.
Sample size
We wished to look at the agreement between two continuous variables (two different methods of measuring total bilirubin: TcB using BiliChek and TSB). Power calculation established that 100 paired results would be a minimum acceptable sample size: allowing estimation of the 95% confidence interval for the limits of agreement with a width of ±0.34 s where s was the standard deviation of the differences between the measurements by the two methods.
Analysis
Analyses were carried out using the full data-set and with a smaller set of the first observation for each infant. The relationship between TcB values and TSB measurements was investigated using X on Y linear regression analysis 12 and difference plots. 13 Usually, X on Y regression would be considered the wrong statistical technique for a method comparison. 14 However, in this situation where the laboratory bilirubin is considered to be the ‘gold standard’ that determines treatment, it is correct because the safety impact of replacing the laboratory TSB assay with the TcB assay is being evaluated. An ROC curve was constructed to investigate the relationship between the bilirubin measurement and phototherapy – essentially, evaluating which measurement (TSB or TcB) had the greatest overall predictive power in babies in whom the blood test had been performed to evaluate clinical jaundice.
Results
Subjects
Gestational age of the subjects ranged from 26 + 0 to 34 + 3 (weeks +days). From 26 to 34 weeks, respectively, there were 1, 2, 2, 8, 7, 4, 14, 10 and 9 infants admitted in each week band.
Precision
Ten separate analytical coefficient of variation (CVs) were estimated on babies with TcB ranging from 80 to 250 μmol/L. CVs ranged from 3.5% to 10.7%, mean 6.33%. CV of laboratory bilirubin estimated from IQC samples at 120 μmol/L is 1.77%. Bias estimated from WEQAS EQA returns is +1.9% against method mean and +6.2% against overall mean at 100 μmol/L.
Method comparison
At the end of the data collection period, there were 183 paired results. Regression analysis showed r = 0.8965, P < 0.005 (full data-set), and r = 0.8775, P < 0.005 (first observation data-set). Figure 1 shows least-squares X on Y regression plot for transcutaneous bilirubin versus laboratory bilirubin (intercept = 17.7, slope = 1.059 (full data-set); intercept = 27.8, slope = 0.997 (first observation data-set)). Figure 2 shows the absolute difference plot and Figure 3 shows the percentage difference plot, with lab bilirubin as the x-axis because in this case this is ‘the gold standard’. It is clear from these graphs that the transcutaneous bilirubin method tends to give a consistently higher estimate for the bilirubin concentration than the laboratory method. The difference increases in the absolute difference plot and remains roughly constant in the percentage difference plot, indicating a fixed calibration bias. Figure 4 is a plot of differences between TSB and TcB, versus gestation age. Regression analysis gave r = 0.055; P = 0.46, indicating no significant association between differences in measurement with gestation age.
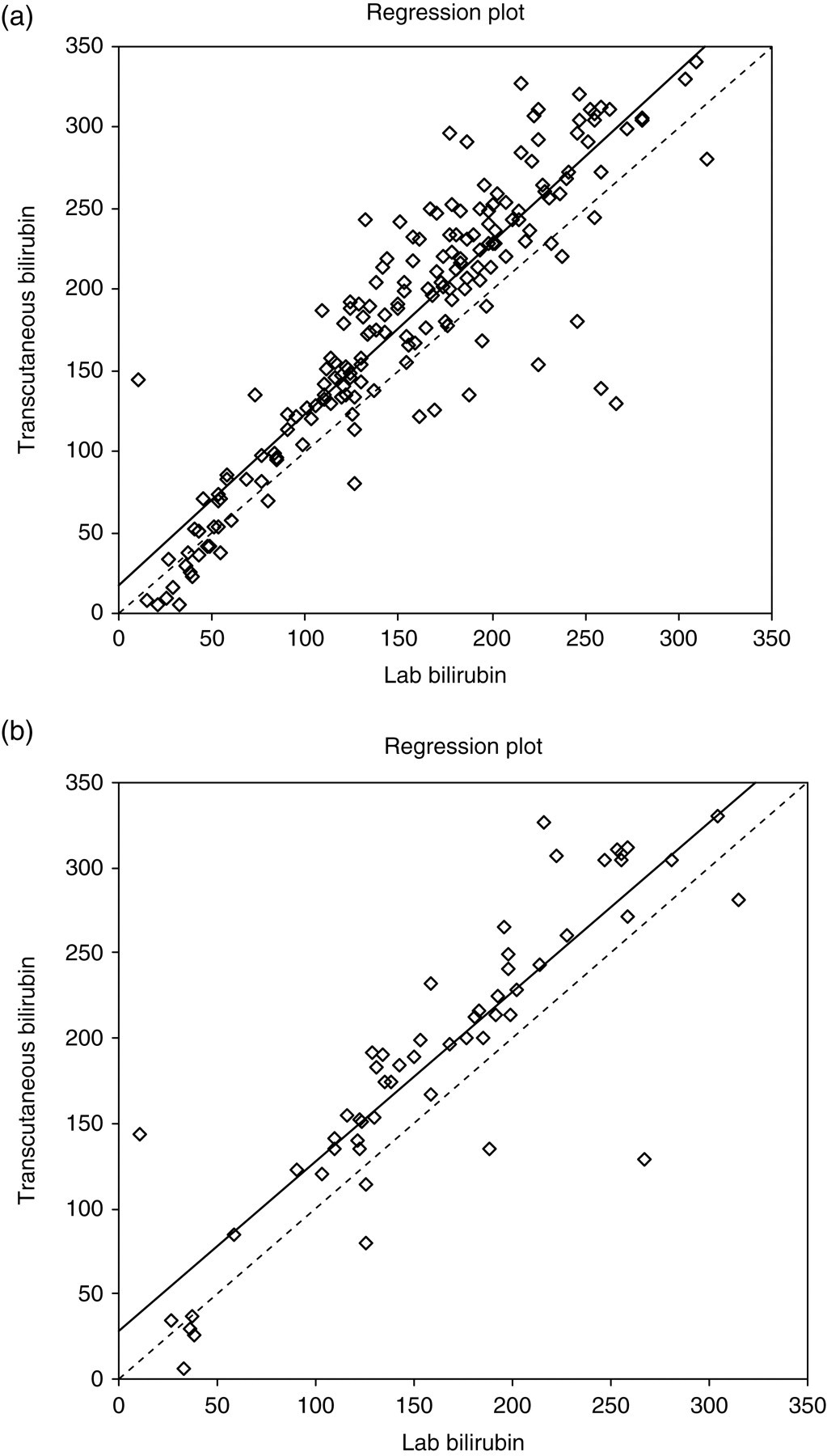
Least squares X on Y regression plot of laboratory bilirubin (μmol/L) versus transcutaneous bilirubin (μmol/L): (a) whole data-set; (b) first result from each infant only
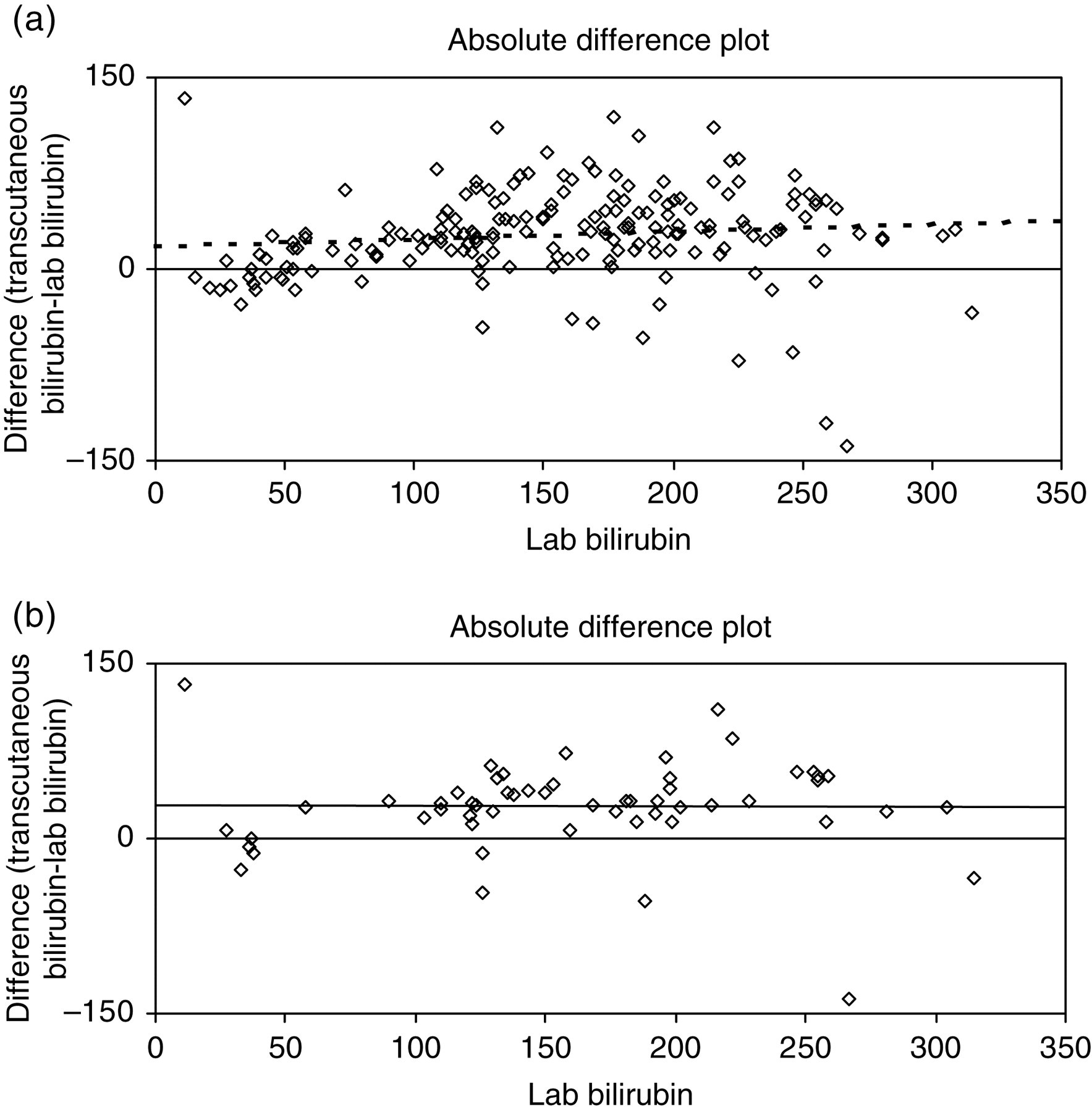
Absolute difference plot for difference (μmol/L) versus lab bilirubin (μmol/L): (a) whole data-set; (b) first result from each infant only
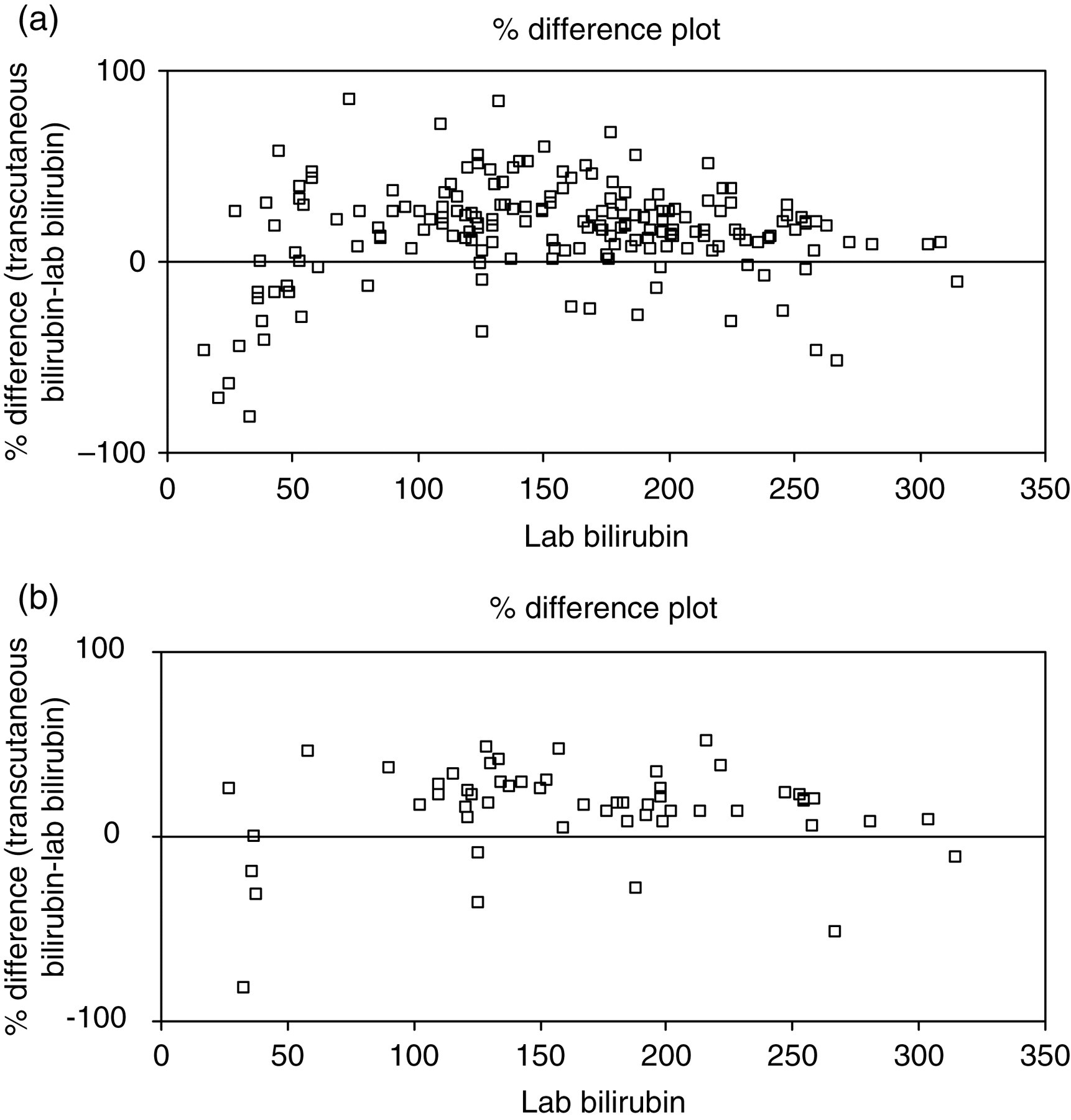
Percentage difference plot for % difference versus lab bilirubin (μmol/L): (a) whole data-set; (b) first result from each infant only
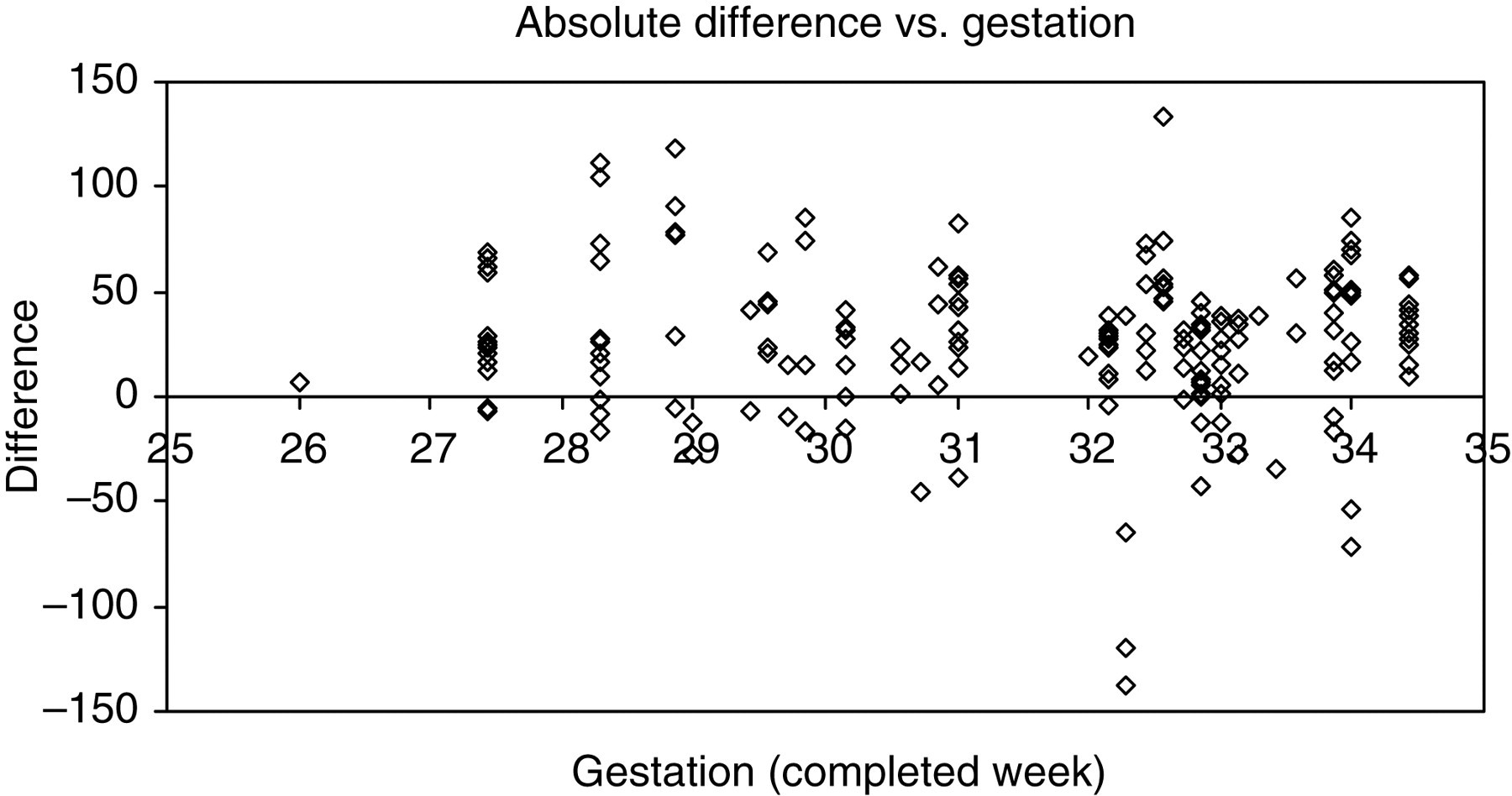
Absolute difference plot for difference (μmol/L) versus gestation age
The important issue in evaluating the utility of the new method is the diagnostic accuracy. Figure 5 shows ROC plots for laboratory and transcutaneous bilirubin. The ROC plot was constructed by ordering the data in two columns (‘received phototherapy’ and ‘did not receive phototherapy’). A series of cut-offs were applied (from 20 to 300 μmol/L, at 20 μmol/L intervals) and the number of true and false, positive and negative results in each column were evaluated for each cut-off. This allowed sensitivity and specificity to be calculated for each cut-off to generate the ROC curve. The area under the curve (AUC) for TSB was 0.8423, and for TcB was 0.7713 (P < 0.5); i.e. there is a statistically greater likelihood that phototherapy would be indicated by TcB measurement.
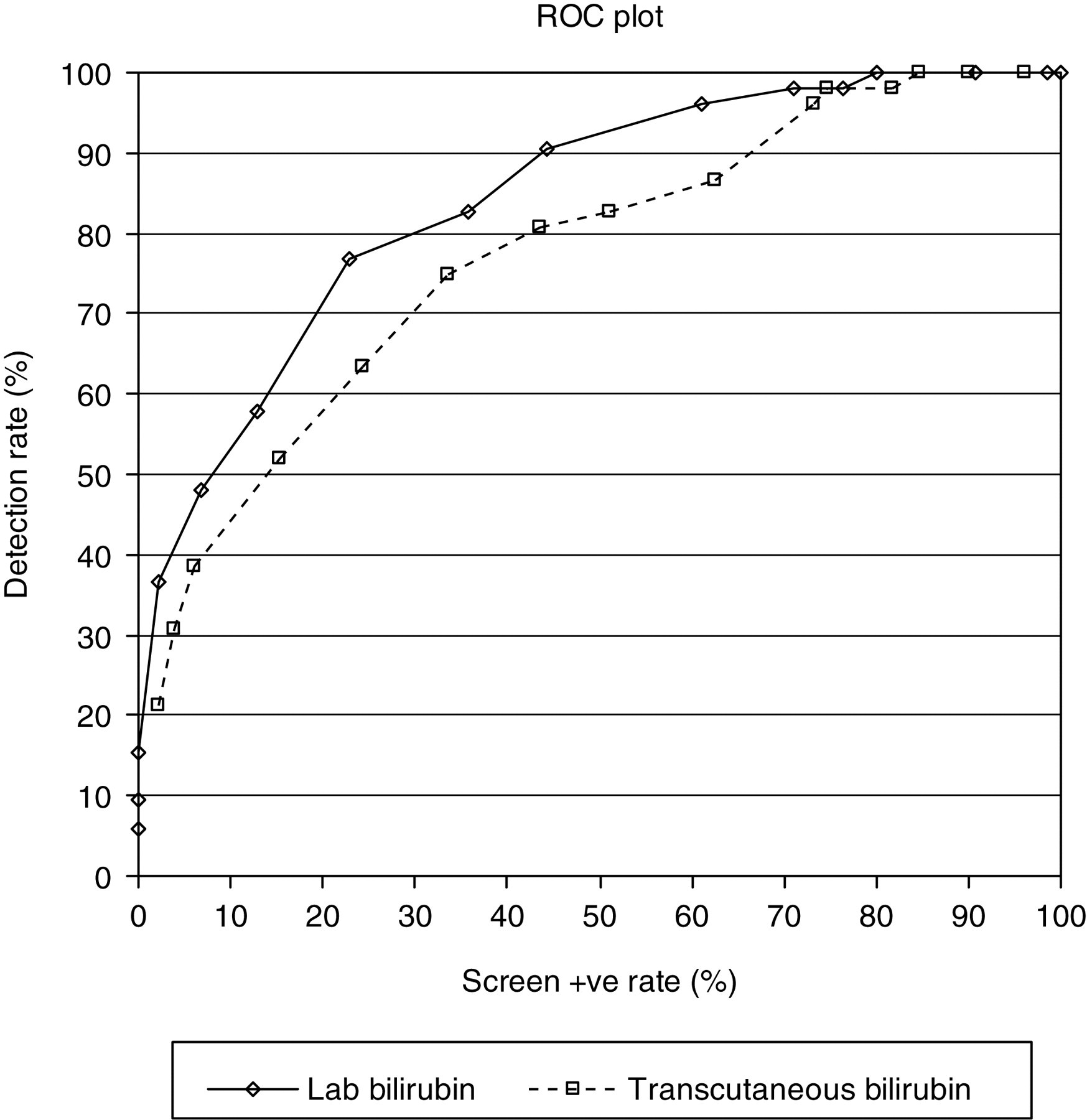
ROC plots for lab bilirubin and transcutaneous bilirubin
Ethnicity
The 183 paired results came from 57 infants, of whom 50 were Caucasian, four were Indian subcontinent and three were mixed ethnicity. Fourteen paired measurements were made on non-Caucasian infants. To evaluate whether skin pigmentation affected the performance, the difference between laboratory and transcutaneous bilirubin measurements was calculated and a Mann-Whitney rank sum test was carried out to assess whether ethnicity was significant. Rank sums were: non-Caucasian 1207; Caucasian 15,629; P = not significant.
Discussion
The average CV for TcB measured using the BiliChek method was 6.33% (range 3.5–10.7%). According to Westgard (
The purpose of neonatal bilirubin assay is to identify those infants in need of phototherapy. The decision is essentially between the risk of bilirubin-induced brain damage and a treatment (phototherapy), which is safe but which may delay the patient's discharge from hospital. Phototherapy is initiated according to a cut-off calculated as
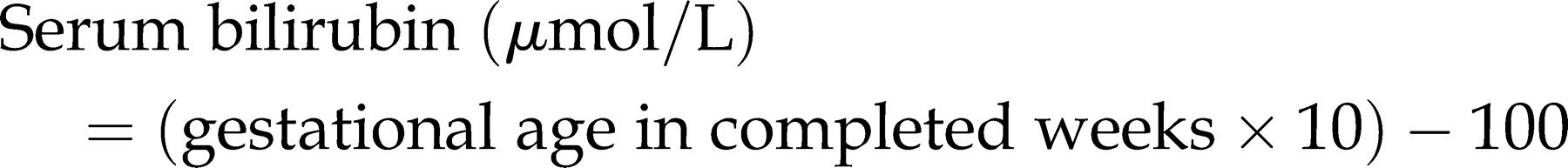
The %difference plot (Figure 3) shows there is a significant increase in scatter and bias below 100 μmol/L. For the gestation range studied, the phototherapy cut-off varies from 160 to 240 μmol/L. Therefore, this bias is of no clinical significance.
Consequently, a change in the decision tool (the bilirubin assay) must be based on the principle that the new method must not increase the risk of the adverse event. However, it is not essential that the alternative method should always give exactly the same result as the ‘gold standard’ laboratory method because it is not only the absolute bilirubin concentration, but also the trend in concentration that is used to inform clinical decision-making – if there is an upward trend which will soon take bilirubin above the treatment threshold then clinical judgement applies and phototherapy may be initiated. Furthermore, the fact that some transcutaneous bilirubin measurements are significantly below the laboratory method is not a major deficiency because currently paediatricians are inhibited from taking blood samples because they aim to minimize trauma to infants. Use of a transcutaneous measurement will allow more frequent bilirubin assessments to be carried out with a lower threshold of suspicion. Thus occasional low readings would be rechecked and discrepancies identified before there are any possible clinical consequences.
From the regression plot (Figure 1) it can be seen that using only the first measurement results in a significant decrease in the number of data points at the low end of the bilirubin range – this is because bilirubin measurement is usually first prompted by a clinical impression of jaundice – so the missing data points are those from when bilirubin is falling. The obvious good agreement between TSB and TcB bilirubin data at the low end of the scale in the full data-set indicates that there is quite good agreement between TSB and TcB when bilirubin is decreasing and therefore that TcB can be used when infants are being treated with phototherapy.
In Figure 5, it is clear that the AUC for the transcutaneous bilirubin method is smaller than the AUC for laboratory bilirubin. Our findings are similar to a previous study of 25–35-week infants that also showed that transcutaneous measurement of bilirubin gave higher estimates, 10 and a study in a neonatal intensive care unit that demonstrated a potential to avoid 40% of TSB measurements, with consequent reduction in trauma to infants. 11
For a given detection rate, the TcB method tends to have a higher screen positive rate, meaning that at any ‘true’ bilirubin concentration the transcutaneous method will identify a higher proportion of infants as needing phototherapy than would the laboratory method. Furthermore, the initial investigation of hyperbilirubinaemia involves collection of samples for other investigations as well as bilirubin, so initial bilirubin assessments are confirmed by the laboratory and under-measurement by a transcutaneous test would therefore not be an issue. Consequently, we believe that the transcutaneous method meets the acceptability criteria for safety result because the threshold for phototherapy is lowered (i.e. more infants receive phototherapy), thereby lowering the risk of causing brain injury by inaction. Furthermore, the reduction in blood sampling requirement is an advantage because it reduces trauma and the likelihood of developing anaemia. The phototherapy protocol includes reassessment of bilirubin at least every 12 h: it has been demonstrated that many cases of anaemia on NNU are iatrogenic. 17,18
Therefore, the only remaining safety consideration is whether the small increase in phototherapy treatment that would result is likely to cause injury to any infants, or would increase workload on the NNU to unacceptable levels. Phototherapy is safe and effective 19 and therefore does not constitute any risk to the infant. We would, however, recommend that very high TcB readings (approaching exchange transfusion values) should be cross referenced by TSB measurements. We therefore conclude that the Bilicheck® represents a safe and acceptable alternative to laboratory bilirubin. Use of this method will reduce unnecessary blood sampling in premature neonates with consequent clinical benefits.
DECLARATIONS
