Abstract
Background
The potential for serum free light chain (sFLC) assay measurements to replace urine electrophoresis (uEPG) and to also diminish the need for serum immunofixation (sIFE) in the screening for monoclonal gammopathy was assessed. A testing algorithm for monoclonal protein was developed based on our data and cost analysis.
Methods
Data from 890 consecutive sFLC requests were retrospectively analysed. These included 549 samples for serum electrophoresis (sEPG), 447 for sIFE, and 318 for uEPG and urine immunofixation (uIFE). A total of 219 samples had sFLC, sEPG, sIFE and uEPG + uIFE performed. The ability of different test combinations to detect the presence of monoclonal proteins was compared.
Results
The sFLC κ/λ ratio (FLC ratio) indicated monoclonal light chains in 12% more samples than uEPG + uIFE. The combination of sEPG and FLC ratio detected monoclonal proteins in 49% more samples than the combination of sEPG and sIFE. Furthermore, the sEPG + FLC ratio combination detected monoclonal protein in 6% more samples than were detected by the combined performance of sEPG, sIFE, uEPG and uIFE. However, non-linearity of the assay, the expense of repeat determinations due to the narrow measuring ranges, and frequent antigen excess checks were found to be limitations of the sFLC assay in this study.
Conclusion
The FLC ratio is a more sensitive method than uIFE in the detection of monoclonal light chains and may substantially reduce the need for onerous 24 h urine collections. Our proposed algorithm for the evaluation of monoclonal gammopathy incorporates the sFLC assay, resulting in a reduction in the performance of labour intensive sIFE and uEPG + uIFE while still increasing the detection of monoclonal proteins.
Introduction
Standard methods for the detection and monitoring of monoclonal proteins include electrophoresis and immunofixation of serum and urine samples. Recently, automated immunoassays using polyclonal antibodies have been developed that can measure serum free light chains (sFLCs) at normal concentrations. 1 The major reported benefits of these assays are increased sensitivity, allowing for greatly improved detection rates of light chain multiple myeloma (LCM), 2 non-secretory multiple myeloma (NSMM) 3 and AL amyloidosis. 4 The assay also enables monitoring these diseases 5 and the ability to detect early response to therapy. 6,7 Other potential benefits of the assay include elimination of the need for 24 h urine electrophoresis (uEPG) 8,9 and use as a prognostic indicator for progression from monoclonal gammopathy of unknown significance (MGUS) to myeloma. 10 Additionally, it has been proposed that the combination of serum electrophoresis (sEPG) and sFLC assay evaluation be incorporated into a screening algorithm for monoclonal gammopathy. 9,11
The majority of the 15% of monoclonal gammopathies that feature the presence of only light chains are not demonstrable by serum electrophoretic methods, and therefore there is a reliance on urine studies for their detection. 8 The elimination of the need for uEPG is highly desirable as 24 h urine samples are frequently not supplied for laboratory evaluation. 12 In our laboratory, of all requests for an sEPG, 80% are accompanied by a request for serum immunofixation (sIFE) whereas less than 20% are accompanied by a request for uEPG. The high frequency of samples analysed by sIFE inadequately compensates for this low frequency of urine electrophoretic determinations. Furthermore, sIFE is a relatively costly and labour-intensive method. Katzmann et al. 8 found that the combination of sIFE and sFLC κ/λ ratio (FLC ratio) in the absence of urine studies would have missed only two (0.5%) patients with a monoclonal protein, and both these cases were deemed not significant enough to warrant further clinical evaluation. Furthermore, this study found no false-positive results when the sFLC assay accompanied an sEPG. In contrast, Hill et al. 9 showed that the combination of sEPG and sFLC assay was sensitive, but in a hospital population of primary care patients the FLC ratio had a positive predictive value of only 39% for B-cell disorders.
A retrospective analysis was performed to evaluate the efficacy of various assay combinations including sFLC assay in order to formulate a cost-effective screening algorithm for monoclonal gammopathy.
Materials and methods
Data from 890 consecutive sFLC requests received by the PaLMS Immunorheumatology Laboratory at Royal North Shore Hospital, a university affiliated and tertiary referral hospital, were retrospectively analysed. The results were compared with 549 samples simultaneously evaluated by sEPG, 447 by sIFE and 318 by uEPG combined with urine immunofixation (uIFE). Serum EPG was performed on all samples that were analysed by sIFE. A total of 219 episodes had sFLC assay, sEPG, sIFE and uIFE performed. To determine the specificity of the sFLC assay in a broadly sourced screening population, 73 samples received over a period of one week that displayed no monoclonal protein on sIFE were further assessed by determining their FLC ratio. Comparative validity of results was based on available clinical history, previous electrophoresis results and other relevant laboratory evaluation.
Assay cost modelling was based on data acquired from routine runs and included repeat test rates due to antigen excess checks and dilutions of out of range samples, reagent usage, labour time, positive detection rates, and ratio of monitoring to screening samples.
Electrophoresis and immunofixation procedures were performed on commercial agarose gels (Beckman Coulter, Paragon SPE, Beckman Coulter Inc. California, USA). Immunofixation utilized antisera to gamma, alpha and mu heavy chains and κ and λ light chains (Beckman Coulter). Urine samples were analysed by antisera specific for free κ and free λ light chains (Helena Laboratories, Texas, USA). Urine assays were performed on specimens concentrated by 80× using a centrifugal filtration device (Amicon Ultra, Millipore) with a molecular weight cut-off of 10,000 Da. The sFLC assay was performed by rate nephelometry using the Freelite™ assay (The Binding Site Ltd, Birmingham, UK) on a Beckman Coulter Immage analyser as per the manufacturer's instructions. The concentration of free κ and free λ light chains was measured and the κ/λ ratio was calculated and used as an indicator of clonality. Ratio values outside the reference range of 0.26–1.65 suggested by the manufacturer indicated the presence of a monoclonal free light chain.
Results
Urine IFE versus FLC ratio
A total of 314 samples from 142 patients were evaluated. The uIFE and FLC ratios were concordant for 94 negative and 157 positive samples, respectively. However, uIFE was positive and FLC ratio was normal in 21 samples from 13 patients (Table 1). One of these samples was a false-positive immunofixation result as the patient had no clinical evidence of a monoclonal gammopathy, a normal bone marrow biopsy and no monoclonal proteins detected on subsequent uEPG studies. The remaining 20 samples originated from 12 patients with a monoclonal gammopathy. Eleven of these samples were from seven patients with renal impairment as defined by an elevated serum creatinine and estimated glomerular filtration rate below the individual's age and gender-adjusted reference range. Further analysis of the samples from these patients with renal failure revealed that seven (four patients) had elevations of both free κ and λ levels with a normal ratio, two (two patients) had increased levels of one light chain isotype and two (two patients) had normal sFLC levels with a normal ratio. Five of the patients with renal failure had monoclonal κ bands with the other three harbouring monoclonal λ bands. By contrast, the FLC ratio indicated monoclonal light chains in 42 samples from 25 patients, which were not detected by uIFE. All of these patients had histological confirmation of a plasma cell dyscrasia. The FLC ratio yielded superior sensitivity and equivalent specificity when compared with uIFE (Table 2).
Comparison of FLC ratio with uIFE for the detection of monoclonal light chains
FLC ratio, serum free light chain κ/λ ratio; uIFE, urine immunofixation
Comparison of sensitivity, specificity and positive and negative predictive values for the detection of monoclonal light chains between sFLC assay and uIFE*
*Assumes that abnormal FLC ratio is a true positive if the patient has histologically confirmed monoclonal gammopathy
FLC ratio, serum free light chain κ/λ ratio; uIFE, urine immunofixation
Serum IFE versus sEPG combined with FLC ratio (EPG + FLC ratio)
A total of 447 samples from 176 patients were analysed (Table 3). The EPG + FLC ratio combination was considered positive if either test indicated the presence of a monoclonal protein. Serum IFE and EPG + FLC ratio were concordant for 107 monoclonal negative and 213 monoclonal positive samples. However, sIFE detected intact immunoglobulin monoclonal bands in nine samples from five patients, which had a normal EPG + FLC ratio. Four of these samples were from a post autologous transplant myeloma patient with renal impairment. This patient had an IgG κ paraprotein band migrating in the β region on sEPG. One sample was from a post autologous transplant NSMM patient that demonstrated an IgG κ paraprotein band migrating within the γ region. This band was indicative of immune reconstitution and not evident on analysis of follow-up samples. Another sample originated from a patient with MGUS and revealed the presence of an IgA paraprotein within the β region. Two specimens were from a patient with peripheral neuropathy and featured a weak β-migrating IgA λ monoclonal band. Another sample was from a patient with a plasmacytoma and an IgA λ band present in the γ region. Hence, the majority of these samples had β-migrating monoclonal proteins. Serum IFE did not increase the detection rate of monoclonal light chain bands beyond that of the sFLC assay.
Comparison of sEPG + FLC ratio and sIFE for the detection of monoclonal protein
*sEPG + FLC ratio considered positive if either assay detects monoclonal protein
sEPG, serum electrophoresis; FLC ratio, serum free light chain κ/λ ratio; sIFE, serum immunofixation
The EPG + FLC ratio combination indicated monoclonal proteins, which were not detected by sIFE in 118 samples from 24 patients. Thirty-two samples from 10 patients featured a monoclonal protein also found by urine IFE. However, 19 samples from nine patients with a histological diagnosis of a plasma cell dyscrasia had a normal uIFE. Sixty-seven serum samples were not accompanied by a urine sample and 66 of these were from patients with a histological diagnosis of a plasma cell dyscrasia. Only one sample originated from a patient without a plasma cell dyscrasia on bone marrow biopsy.
Serum IFE combined with uIFE (sIFE + uIFE) versus EPG + FLC ratio
A total of 219 samples from 110 patients were analysed (Table 4). Test combinations were considered positive if any component assay demonstrated the presence of a monoclonal protein. Monoclonal proteins were detected by both test combinations in 154 samples from 75 patients and were absent by either test combination in 37 samples from 31 patients. The sIFE + uIFE detected a monoclonal protein in nine samples from five patients where the EPG + FLC ratio was normal. All had a confirmed histological diagnosis of a monoclonal gammopathy. Six samples from four patients were found to have a monoclonal protein by uIFE only and all of these samples contained a trace of Bence Jones protein (BJP). Three samples from one patient with renal impairment revealed a serum IgG paraprotein in the gamma region and a BJP with the same electrophoretic mobility. By comparison, the EPG + FLC ratio indicated monoclonal proteins in 19 samples from nine patients where the sIFE + uIFE was normal. All had a confirmed diagnosis of a plasma cell dyscrasia. Three of these patients, comprising seven samples, had renal impairment.
Comparison of sIFE + uIFE with *sEPG + FLC ratio for the detection of monoclonal protein
*sEPG + FLC ratio considered positive if either assay detects monoclonal protein
†sIFE + uIFE considered positive if either test detects monoclonal protein
sIFE, serum immunofixation; uIFE, urine immunofixation; sEPG, serum electrophoresis; FLC ratio, serum free light chain κ/λ ratio
Specificity of the sFLC assay
Seventy-three samples received in one week that did not contain a monoclonal protein on sIFE were assayed for sFLCs. These samples represented a cross section of in- and outpatient samples screened with an sEPG from various hospital departments including haematology, nephrology, rheumatology, hepatology, endocrinology and emergency. Approximately 50% of the requests were derived from the haematology department.
One of these 73 samples had abnormal FLC ratios yielding a specificity of 99% for plasma cell dyscrasia. This false-positive result consisted of a borderline abnormal ratio of 0.24 (reference range 0.26–1.65) from a patient with positive anti-SSA and anti-SSB antibodies but no subsequent diagnosis of a plasma cell dyscrasia.
Linearity of the sFLC assay
Serum FLC results outside the normal range were checked for antigen excess by repeat testing at a higher dilution as advised by the manufacturer (The Binding Site Ltd, Birmingham, UK). We reviewed the antigen excess checks from three assay runs for evidence of non-linearity. There were 25 checks in total, 13 for excess κ and 12 for excess λ, and none of these found to be in antigen excess. None of the dilutions used to check for κ excess yielded a result within 50% of the target value that was obtained with the standard screening dilution. Seven of these checks were more than double the target value, including two that were more than treble the target value. The checks for λ excess were more consistent, with seven of 12 yielding results within 50% of the target and only two over 100% greater than the target value.
Cost analysis
This study population had a high frequency of plasma cell dyscrasia and a low sample size with 30 samples analysed on average per week. The cost per sFLC assay was Aus$ 42 per sample. However for the sample population selected from our routine EPG requests according to the proposed protocol (Figure 1), the lower frequency of positive samples associated with screening samples results in less repeat testing, which combined with greater economies of scale, reduce the cost per test to Aus$ 33 (Table 5).
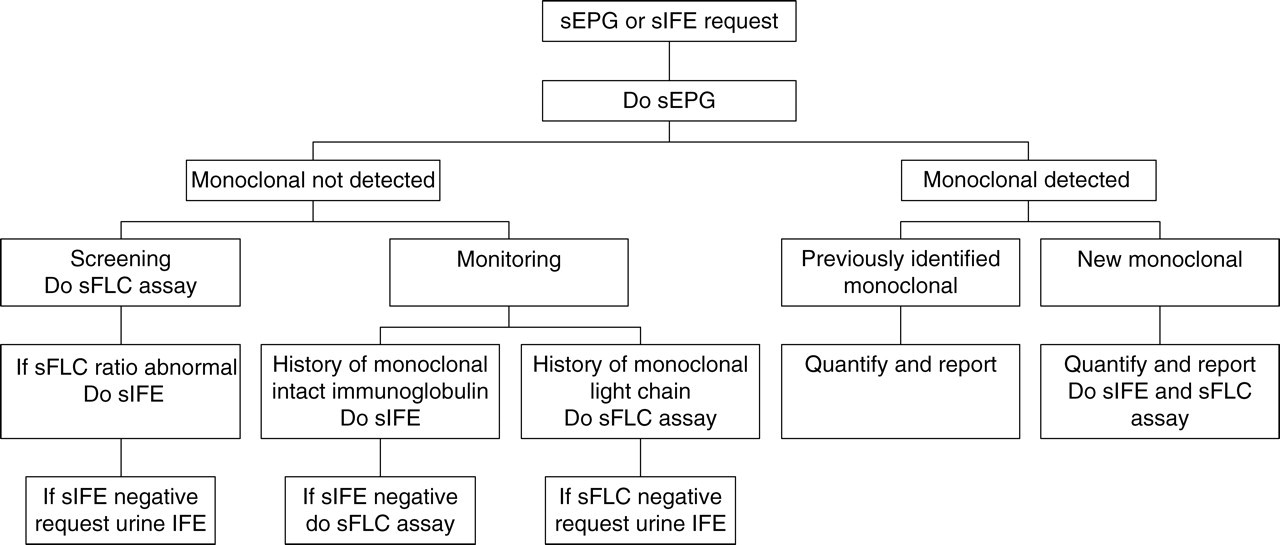
Proposed protocol for laboratory evaluation of monoclonal gammopathy. sEPG, serum electrophoresis; sFLC, serum free light chain; sIFE, serum immunofixation
Comparison of the estimated costs* per week between the current and proposed protocols
*Costs include reagents, controls and calibrators, labour and consumables, weekly calibration for sFLC
†Current protocol is to perform specific assays as requested by clinician
sEPG, serum electrophoresis; sIFE, serum immunofixation; uEPG, urine electrophoresis; sFLC, serum-free light chain
The costs modelled on our laboratory's request patterns, positive detection rates and the requirement of repeat testing indicate that a change in protocol would incur similar reagent costs and save approximately five hours of labour per week.
Proposed protocol for monoclonal gammopathy
A significant proportion (5–15%) of patients with monoclonal gammopathy, including LCM, AL amyloidosis and NSMM, produce monoclonal light chains only. 8 These monoclonal light chains may not be detectable in the absence of a urine sample. In our laboratory less than 20% of screens include a urine sample. The sFLC assay could potentially increase our detection rate simply by allowing us to test all samples. The cost, however, of introducing sFLC assay for all sEPG requests would be prohibitive (>Aus$ 110000 p.a.). We have devised a cost-effective strategy for maximizing the detection of monoclonal proteins by utilizing the sFLC assay and restricting the use of immunofixation to the typing of new monoclonal proteins, the clarification of EPGs that are difficult to interpret and the classification of the therapeutic response (Figure 1).
Reducing the use of sIFE poses the risk of not detecting a small number of monoclonal proteins with β or α-2 electrophoretic mobility. We found 28 samples from nine patients in whom the sEPG failed to detect a β-migrating monoclonal protein that was detected by sIFE, the largest of which was an IgA paraprotein quantified at 3.8 g/L. The sFLC ratio indicated monoclonality in 21 of these samples comprising six patients. These included three patients with IgA paraproteins on immunofixation and three with light chain bands only.
Discussion
These data support previous studies that have demonstrated increased sensitivity of sFLC assay compared with uEPG + uIFE. 13 This however assumes that an abnormal FLC ratio is a true indicator of monoclonal light chains in patients with histologically confirmed monoclonal gammopathy. This assumption is strengthened by the high specificity found in our monoclonal gammopathy negative population. Our study also supports those of others that suggest the combination of sEPG and FLC ratio is an effective screening strategy for monoclonal gammopathy. 9 This strategy increases the detection of monoclonal proteins compared with immunofixation of both serum and urine. Our results suggest that the FLC ratio is a more sensitive method for the detection of monoclonal light chains than uIFE. Similarly the combination of FLC ratio and serum EPG was more sensitive than either sIFE or sIFE combined with uIFE, and this supports its proposed utility in the detection of minimal residual disease. The combination of sEPG and FLC ratio fails to detect a small number of monoclonal proteins found by immunofixation. The monoclonal proteins not detected by the combination of sEPG and FLC ratio are likely to be small bands within the β and α-2 regions. Patients harbouring such bands are likely to have MGUS. Of the 28 samples from nine patients with β-migrating monoclonal protein not detected by EPG, 21 were flagged as having a monoclonal protein by FLC ratio, which in our proposed protocol would trigger the performance of an sIFE. The β-monoclonal proteins in the remaining seven samples would not be detected without the addition of immunofixation to the screening protocol.
We found only one false-positive result in our sFLC request cohort, the majority of whom were evaluated by clinical haematologists. This supports the Mayo study, which found no false-positive results in 121 individuals without monoclonal gammopathy. 14 Another false-positive result was detected in our smaller sEPG screening population, providing a specificity of 99%. In contrast, Hill et al. 9 found more false positives when analysing samples sourced from a broad range of clinical disciplines and concluded that the false-positive rate is problematic in a laboratory with a relatively low frequency of requests for sIFE. The false-positive FLC ratio introduced a significant increase in the need for sIFE. In our laboratory 80% of sEPG samples are also assayed by sIFE. In our proposed testing protocol the reduction in sIFE due to the introduction of sFLC assay far outweighs the modest increase due to false-positive FLC ratio results. Five patients in our study had traces to low levels of BJP detected by uIFE but had a normal FLC ratio. A low concentration of urine monoclonal proteins in patients with normal sFLC concentrations and κ/λ ratios has been reported to be of doubtful clinical importance. 15
The sFLC assay, however, has a number of analytical problems, including non-linearity in a significant number of samples, a risk of antigen excess, batch to batch variation, narrow measuring ranges and build-up of latex in cuvettes, which can disturb the performance of other assays. 15–18 Antigen excess checks and narrow measuring ranges result in up to 50% of samples in our assay population being re-tested at further dilutions, which is expensive and time consuming. Non-linearity, antigen excess and batch variation may lead to inconsistency with patient monitoring. 16,17 In contrast to previous reports, we found non-linearity to be the consistent finding rather than the exception, despite adhering to the manufacturer's recommendations. Hence, for patients that are monitored by sFLC assay, results obtained from sequential samples at different dilutions may include an artefactual shift. If possible, a previous sample should be retested at the same dilution in parallel. 17 This procedure also guards against the effects of batch variation. Although non-linearity may cause inconsistencies in monitoring patients, it is unlikely to affect diagnosis provided standard screening dilution is used.
Renal impairment affects the FLC ratio and potentially results in both false-positive and false-negative FLC ratios. 17 Although twice as many κ as λ light chains are produced in humans, 19 there is a faster rate of clearance for κ light chains, resulting in a bias in the κ/λ ratio towards λ. 20,21 With renal impairment, the ability to clear κ faster than λ is lost with a subsequent rise in the κ/λ ratio. 22 It would therefore be expected that false-negative results due to renal impairment would occur in the presence of λ light chain monoclonal proteins. However, most of our suspected FLC ratio false negatives were of κ type, suggesting only a limited impact from renal impairment. It is possible that alterations in renal function are mirrored in changes in sFLC from both malignant and benign sources so that the κ/λ ratio is not significantly altered. 12
The reliance of quantitation of BJP in 24 h urine samples is also troubled by technical problems, including underestimation of light chains by total protein assays, concentration artefacts, electrophoretic smearing due to polymerization and protein binding, staining variability and proper collection. 15,23 Impaired renal function, especially in advanced renal failure, may also affect excretion of Bence Jones as a result of oliguria or anuria. The major limitation as found by our laboratory and that of others is the difficulty of obtaining a urine sample for analysis. 8,12 A 24 h urine collection is still required, however, for the estimation of proteinuria. 24
Limited data on sequential samples from unique patients (not shown) that compared changes in concentrations of 24 h urine BJP with sFLC measurements showed acceptable correlation coefficients in the range of 75–99%. A more detailed prospective study is required to determine the utility of the sFLC assay in the monitoring of patients with monoclonal gammopathy. A recent study by Dispenzieri et al. 23 found that the sFLC assay enabled monitoring of disease in the absence of a quantifiable M-spike. Bradwell et al. 2 showed previously that following chemotherapy, 32% of patients were considered to be in remission as assessed by urinary FLC compared with 11% when assessed by the sFLC assay. The modest correlation between serum and urine monoclonal FLC observed in some studies 2,25 can at least partly be explained by the renal metabolism of FLC, which is affected by renal failure in up to 50% of myeloma patients at diagnosis. 26
Our study demonstrates the utility of the sFLC assay in combination with sEPG for the screening of monoclonal gammopathy. An algorithm has been formulated incorporating the sFLC assay, which is a cost-effective and efficient means of detecting monoclonal proteins, especially in a population where a low percentage of urine samples are submitted for evaluation. The advantage of our study in devising our algorithm is the practical use of the sFLC assay in a laboratory and the use of histology and IFE as standards instead of clinical diagnosis. 11,27 A larger prospective study is, however, required to confirm our results. Analytical problems affecting the sFLC assay include the frequent finding of non-linearity, the necessity for frequent assay repeats due to narrow measuring ranges and antigen excess detection. These analytical shortcomings should be addressed by the manufacturer.
DECLARATIONS
