Abstract
Background
Accurate measurement of sweat chloride concentration is essential for the diagnosis of cystic fibrosis (CF). We surveyed all laboratories enrolled in the Royal College of Pathologists of Australasia Quality Assurance Program (QAP) for Sweat Electrolytes to determine how closely they comply with the Australian Guidelines for the performance of the sweat test for the diagnosis of CF.
Methods
A detailed questionnaire covering most aspects of sweat collection and analysis was sent to all participating laboratories in 2007.
Results
Twenty out of 38 laboratories completed the questionnaire. While adherence to accepted guidelines was noted in many areas, the following main variations were recorded: some laboratories were not doing enough sweat tests to maintain expertise; some were not collecting sweat for the recommended collection time; sweat conductivity was the only test available in some laboratories; there was a lack of agreement between the sweat chloride concentration used to indicate CF or define an equivocal result.
Conclusions
There is room for improvement in the performance of the sweat test in some laboratories in Australasia. The Sweat Testing Working Party of the Australasian Association of Clinical Biochemists is the appropriate body to address the problems involved in sweat testing and to bring about change.
Introduction
The measurement of sweat chloride concentration remains a crucial test for the diagnosis of cystic fibrosis (CF), and needs to be performed to established guidelines to prevent false-positive or false-negative results. 1,2 Sweat collection is generally performed in one of two ways. The Gibson Cooke method 3 uses pilocarpine iontophoresis to stimulate sweat production, with subsequent collection of sweat onto gauze or filter paper for analysis. More recently, many laboratories have changed to using the Wescor apparatus (Wescor Inc, Logan, UT, USA) – this also employs pilocarpine iontophoresis (with the pilocarpine being embedded into a gel) but the sweat is collected into microbore tubing.
Measurement of sweat chloride is the definitive test for the diagnosis of CF. However, some centres also measure sweat electrical conductivity which must only be used as a screening test. 1,2,4 Sweat for conductivity measurement is collected either into a metal cup or into the Wescor microbore tubing.
Since 2000, the Royal College of Pathologists of Australasia (RCPA) Quality Assurance Programs Pty Ltd (RCPA QAP) has carried out a programme for sweat analysis. Each month, laboratories receive two solutions of sodium/potassium chloride which they must analyse according to the principles of their sweat test method.
In 2002, a working party was formed by the Australasian Association of Clinical Biochemists (AACB) in association with the Chemical Pathology QAP group of the RCPA QAP. This working party was established to oversee the performance of sweat testing by Australian laboratories to improve standards and issue guidelines which were published in May 2006. 4 These guidelines include information on the indications for sweat testing, acceptable and non-acceptable tests, suitable patients to test, patient preparation and possible complications, methods of collection and analysis, reference intervals and reporting, quality control and training.
In the past, laboratories in the United Kingdom 5 and New Zealand 6 have been surveyed by a questionnaire regarding their performance of sweat testing. In 2007, the AACB Sweat Testing Working Party carried out a similar exercise. The aim of such a questionnaire was to determine how closely laboratories comply with the published guidelines for the performance of the sweat test. The results are reported here.
Materials and methods
In 2007, the 38 laboratories in Australia and New Zealand participating in the RCPA Sweat Electrolyte Program were sent a detailed questionnaire regarding all the important aspects of the sweat test. It was known that 28 of these laboratories measure sweat chloride concentration, with 14 measuring sweat conductivity, either alone or in combination with sweat chloride quantitation. The questionnaire consisted of eight sections, and a brief summary of the questions in each section follows.
Section A: basic information
This contained questions about the number of sweat tests performed each year, the number of abnormal/equivocal results and how information about sweat testing was conveyed to patients and parents.
Section B: organization
This asked about the number of staff trained to do sweat tests, how many tests each person did per year and any exclusion criteria in relation to the patient's age, gestation and weight.
Section C: sweat collection – method
This covered all the details regarding how the sweat test was performed, what instrumentation was used, the duration of sweat collection and the occurrence of complications.
Section D: adequacy of collection
This section asked how the amount of sweat collected was estimated, the criteria for an inadequate collection and the frequency of repeat testing.
Section E: analytical methods
Details regarding the measurement of chloride and sodium, including the concentration and number of standards and controls were covered here, along with details of instruments used.
Section F: result interpretation
This section included questions about reference intervals for healthy and CF populations, and the addition of interpretative comments to reports.
Section G: action based on results
Important questions in this section related to the reporting of equivocal results and the action necessary to follow up these results. Also in this section were questions about the measurement of sweat conductivity. Laboratories were asked if they followed up abnormal or equivocal conductivity results by performing a sweat chloride measurement.
Section H: any other comments
This gave laboratories the opportunity to add remarks not covered adequately by the rest of the questionnaire.
Results of the survey were kept anonymous so that it was not possible to identify individual laboratories that took part in this survey.
Results
Twenty of the 38 laboratories (53%) completed the questionnaire. A summary of the responses obtained is set out below. Table 1 shows the compliance rate with some of the major aspects of the published guidelines on sweat testing.
Compliance with some of the major aspects of accepted guidelines
CF, cystic fibrosis
Note: Instances of compliance do not equate to the number of laboratories because some laboratories may have recorded more than one instance of compliance to a particular question, e.g. number of collectors performing 10 sweat tests per year. In addition, not every laboratory provided readily interpretable answers to every question, hence the lower numbers for answers to some questions
Section A: basic information
Six centres were performing 200 or more tests annually but two laboratories reported performing only six tests per year. Seven out of the 20 laboratories provided information sheets to parents. The others relied on oral explanation at the time of doing the test. The number of sweat tests performed by each laboratory is shown in Figure 1.
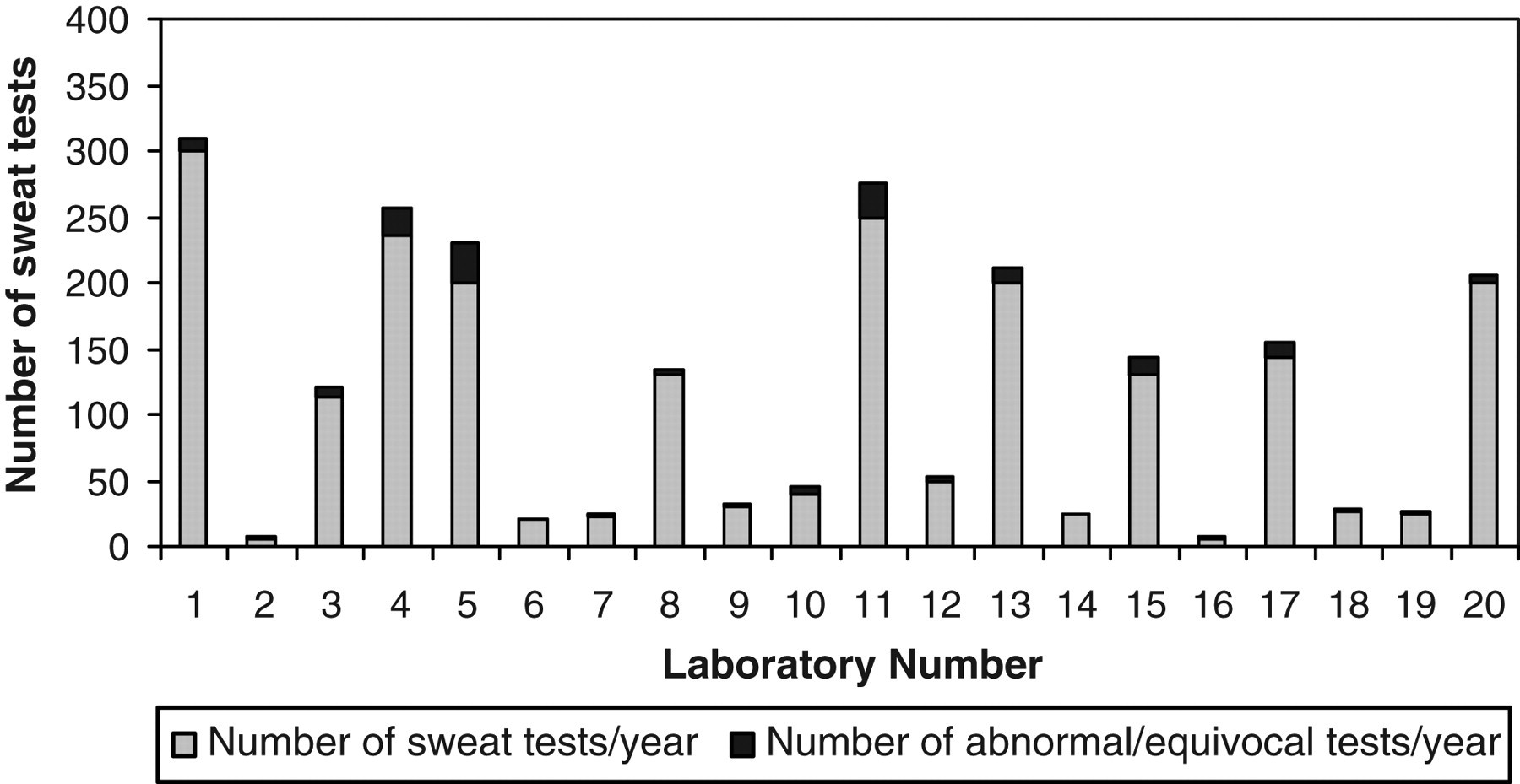
Number of sweat tests performed per year in each laboratory
Section B: organization
Sweat collection was performed by specimen collection staff (which included nurses) in eight centres and by laboratory technical staff in 14. Two centres employed a mixture of specimen collection staff and laboratory staff to do this work. The total numbers of staff carrying out sweat collections in each laboratory ranged from two to 12 and the number of collections per year done by each sweat collector ranged from one to 104. One laboratory collected sweat on site and then sent it to another central laboratory for analysis.
Exclusion criteria
Some laboratories did not document a minimum age for sweat collection. The minimum age for others ranged from two days to four months. Only two centres took gestational age into account. Eleven laboratories did not stipulate a minimum weight of the subject. One laboratory had the minimum weight as >1.5 kg, while four had minimum weights of 3 kg or more.
Adolescent/adult tests
There was wide variation in the percentage of subjects tested in the adolescent and adult age groups, ranging from <2% to 40%.
Section C: sweat collection – method
Nine laboratories used the Gibson Cooke method for collecting sweat, 10 used the Wescor apparatus and two employed an ‘Advanced Instruments collecting cup’ (one of these used the cup only for measuring conductivity, not chloride). Pilocarpine was universally employed at the anode, except for one laboratory, which stated that it used potassium bicarbonate (possibly an error in reporting). At the cathode, most used pilocarpine as well, but potassium nitrate, dipotassium hydrogen phosphate, potassium sulphate, magnesium chloride, magnesium sulphate, sodium nitrate and potassium bicarbonate were also used there.
Site
All collectors used the forearm, but the lower leg and the thigh were also used on occasion. The majority of laboratories performed only one sweat test at a time, but some did two on opposite limbs in agreement with the Clinical and Laboratory Standards Institute (formerly NCCLS) guidelines. 1
Current
No laboratory reported using a current greater than 4 mA, although one centre stated that they used a low current of 0.2 mA for 10 min. Another centre employed a current of 1.5 mA over 15 min.
Duration of collection
Reported collection times ranged from 10 to 60 min, and only eight out of 19 laboratories were using a collection time of 20–30 min. 1,2,4
Section D: adequacy of collection
Those laboratories using the Wescor microbore tubing for collection decided adequacy of sweat collection on volume of sweat obtained, while those using the Gibson Cooke method used weight of sweat. Reported minimum volumes or weights were checked to see if they corresponded to the minimal sweat rate stipulated by the guidelines (1 g/m2/min). 1,2 It was found that some laboratories quoted minimum acceptable volumes or weights of sweat, which were too low to give reliable results (see Table 1).
Section E: analytical methods
Measurement of chloride
There was equal use of colorimetric techniques (which included titrimetry), ion selective electrodes (ISEs) and coulometric methods (which included use of a chloridometer). Concentrations of standards ranged from 0 to 200 mmol/L. Most laboratories used a single standard (100 mmol/L) plus a ‘blank’, but several used multiple standards. The most commonly used internal controls (8 laboratories) were the commercially available Quantimetrix QC (Quantimetrix, CA 90278, USA) with chloride values of approximately 26, 52 and 106 mmol/L.
Measurement of sodium
Eight laboratories employed a flame photometer for measuring sodium, while five used an ISE. One laboratory used atomic absorption spectrophotometry for measuring sodium. There was wide variation in the ways standards were run. Some centres used a single standard, while others employed multiple standards.
To a specific question regarding the addition of quality control material to filter paper or gauze (for those laboratories using the Gibson Cooke method), seven centres replied ‘Yes’ and two centres replied ‘No’.
Section F: result interpretation
Table 2 sets out what laboratories quote as reference intervals for sweat chloride for a healthy population and a CF population. For those laboratories measuring sweat conductivity, reference intervals for healthy and CF populations are also quoted.
Reference (Ref) intervals quoted by laboratories for sweat chloride for a healthy population and a cystic fibrosis population
CF, cystic fibrosis
*Laboratory measures sodium only
†Ref intervals not quoted
Section G: action based on results
Most centres now report an equivocal or intermediate range of 40–60 mmol/L for sweat chloride. This is in line with published guidelines. Three centres that carry out conductivity measurements stated that they do not perform sweat chloride quantitation when they obtain abnormal or borderline conductivity results. Unfortunately, the questionnaire was not specific enough to determine if these laboratories referred patients with abnormal or borderline conductivity results to another centre for definitive measurement of sweat chloride.
Discussion
It is apparent from this survey that some laboratories in Australasia are performing very few sweat tests each year, while others perform large numbers. Given that the Australian Guidelines recommend that each person trained to carry out sweat tests perform at least 10 tests each year, those laboratories performing small numbers of tests, and finding difficulties in maintaining expertise in sweat testing, should give consideration to referring patients to larger, more experienced centres. While this may have implications for regional centres, patients can receive financial support for travel to sweat test centres where a sweat test can be reliably performed.
The survey shows wide variation in the exclusion criteria for sweat testing. The Australian Guidelines state that sweat tests are generally not performed until the subject is more than two weeks of age and weighs more than 3 kg. While in exceptional circumstances the test can be attempted in smaller, younger infants, 7 the guideline that the subject be older than two weeks (corrected for gestational age) and weigh more than 3 kg should be followed, to prevent inadequate collections and the risk of burning the skin. Sweat tests should never be performed on babies under 48 h of age, as falsely high values may be obtained. 8
The NCCLS (now renamed CLSI) guidelines state that the potential for causing a burn increases with the magnitude and duration of the iontophoretic current. 1 These guidelines also state that the current should be brought up slowly to a maximum of 4 mA and maintained there for 5 min. Therefore, very low currents, such as 0.2 mA, as employed by one laboratory, should probably not be used, and prolonged applications of the current should be avoided.
For the process of sweat collection, the appropriate chemicals that avoid contamination were generally employed at the anode and cathode. One centre was using magnesium chloride at the cathode, but this laboratory was only measuring sweat sodium and not sweat chloride. In accordance with the published guidelines, sweat should be collected for no more than 30 min and no less than 20 min, as the concentration of electrolytes is dependent on sweat rate. Extending the sweat collection period beyond 30 min will produce little, if any, additional sweat. 2 Also, it has been reported that prolonging the collection beyond 30 min can result in a sample from less than maximally stimulated sweat glands and can lead to a false-negative result. 1 Laboratories collecting sweat for periods outside the recommended time of 20–30 min should review their practices.
The decision limits regarding the adequacy of collection must be based on the sweat secretion rate, which should not be less than 1 g/m2/min. Approximately one-third of the laboratories employing both the Gibson Cooke and the Wescor collection systems reported accepting sweat collections below these minimal weights/volumes.
Methods employed for analysis of sweat generally fell within the accepted guidelines. For those laboratories using the Gibson Cooke method, internal quality control material must first be added to, and then eluted from, the gauze or filter paper, i.e. the control must be handled in the same fashion as a patient. Two laboratories were not performing this step. One laboratory only measured sweat sodium, without chloride. As measurement of sweat chloride is the definitive test for the diagnosis of CF, measurement of sweat sodium alone is no longer acceptable. 1,2,4,9
There is general consensus among Sweat Test Guidelines published in the USA, the UK and Australia, and the United States Cystic Fibrosis Foundation consensus statement, 9 that a sweat chloride concentration >60 mmol/L strongly supports the diagnosis of CF. Those centres using a higher cut-off value for diagnosis of CF should lower it to 60 mmol/L. All laboratories should also quote an equivocal reference interval for sweat chloride that will alert clinicians to the possibility of CF. This interval is set between 40 and 60 mmol/L in the Australian and other guidelines, but it is possible that the lower limit of this equivocal range may change to 30 mmol/L. 9 It is known from studies in infants without CF that the upper two standard deviations limit for sweat chloride is about 30 mmol/L. 10
Laboratories performing sweat conductivity measurement alone must regard this as a screening test. Sweat conductivity >80 mmol/L (expressed as sodium chloride equivalent) is very likely to be due to CF. 4 However, in accordance with the Australian Guidelines, all patients with sweat conductivities >50 mmol/L should have measurement of sweat chloride.
There is a strong case for standardizing the reporting of sweat test results, including interpretative comments, across Australasia. This should benefit clinicians and ensure that both diagnostic and borderline results are followed up appropriately. This has been problematic up until now in the absence of reliable reference intervals. A recently completed study of sweat chloride values from truly healthy controls screened for a CF phenotype and the common CFTR gene mutation (ΔF508) is now available. 11 This shows that sweat chloride concentration increases with age. Therefore, it seems appropriate that age-adjusted reference intervals should be part of reporting sweat test results in the future. This is particularly appropriate, as this survey has shown that in some centres in Australasia, high numbers of sweat tests are performed on adolescents and adults who may have milder forms of CF.
In conclusion, this survey has demonstrated that most sweat tests performed in Australasia conform to the published guidelines. However, this conclusion is not certain, given that there was only a 53% response rate to the questionnaire. The questionnaire has, however, served a useful purpose in raising awareness of the published guidelines and alerting the Sweat Testing Working Party to the current deficiencies in sweat testing in Australasia. The results of the RCPA Sweat Electrolyte Program indicate that sweat analysis in Australia is universally performed at a high level but the all-important collection procedures cannot be tested in a QAP. Because of the technical aspects of sweat collection, it is important to ensure that all staff performing sweat collection and analysis are properly trained, and maintain their expertise by performing sweat tests regularly. Improving compliance with the collection component of sweat testing should lead to improved consistency and reliability of results between laboratories. The use of conductivity is only valid as a screening test, and in the case of abnormal or equivocal conductivity results, sweat chloride must be measured. Finally, laboratories across Australasia need to develop consensus with regard to those sweat chloride results that definitely signify CF, and those that are equivocal and require further follow-up and investigation.
DECLARATIONS
