Abstract
Understanding the mechanisms of drug metabolism and interactions can help to prevent side-effects. Not only drug interactions, environmental factors, disease processes and ageing are factors in the inter-individual metabolic capacity variance but also genetic factors probably play an important role, as is illustrated in the case presented. Besides therapeutic drug monitoring, genotyping some important cytochrome P450 (CYP450) enzymes was of additional value in explaining why the patient developed severe adverse effects and, moreover, did not experience any therapeutical effect of venlafaxine. Results indicated that the patient was a poor metabolizer for CYP2D6, the most important phase I enzyme to metabolize venlafaxine. This corroborates that polymorphisms in the CYP450 gene influence the metabolic activity of the corresponding enzymes, thus affecting the subsequent serum drug levels and their metabolites. This case highlights the potential benefit of both clinical and genetic risk stratification (pharmacogenetics) prior to treatment, either for setting the individual dose or for making a decision about using a particular drug.
Introduction
Adverse drug reactions (ADRs) pose a serious medical problem and are an important burden on health-care costs. 1,2 In general, the recognition of ADRs has become an increasingly important area in clinical practice. Clinical presentation of ADRs varies and to relate certain reaction with a drug, there must be a temporal relationship, appropriate clinical presentation and, ideally, improvement with discontinuation of the drug. Drug-induced reactions can cause any known pathological pattern. At present, there are no specific tests; rechallenge is unlikely to be helpful and is generally regarded to be dangerous. Drug metabolism is influenced by drug–drug interactions, environmental factors, disease processes, food and ageing. 3 Furthermore, there is increasing evidence that interindividual difference in the genetic profile of phase I and II enzymes account for a substantial portion of the heterogeneity of the response to medication(s) and the development of ADRs. 4,5
Antidepressants are widely used in clinical practice, although the effect is debated. Venlafaxine, for example, belongs to a new generation of antidepressants being a serotonin (5-HT)–norepinephrine (NE) reuptake inhibitor. Venlafaxine and its main active metabolite, O-desmethylvenlafaxine (ODV), inhibit the reuptake of both 5-HT and NE with a potency greater for the 5-HT than for the NE reuptake process. Venlafaxine is metabolized by CYP2D6 into its main active metabolite ODV and to a lesser extent by CYP3A4 into the inactive N-desmethylvenlafaxine, see also Figure 1. 6–8 The suggested therapeutic concentration range lies between 195 and 400 μg/L for the sum of venlafaxine and ODV, and depends mainly on the CYP2D6 activity. 8,9 Decreased CYP2D6 activity increases the risk of side-effects. 7
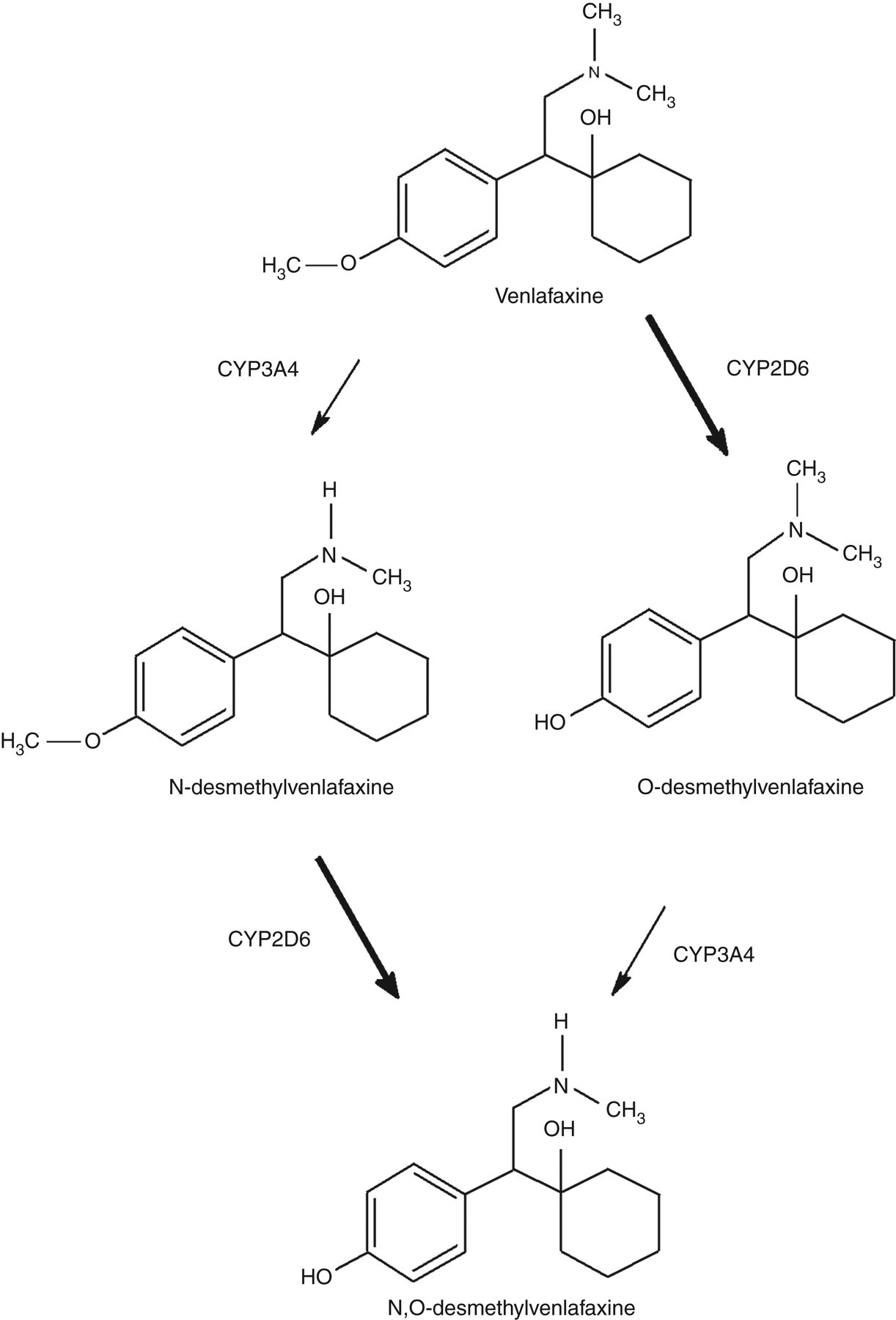
Main metabolic pathway of venlafaxine
Side-effects, due to overdosing, cause only mild symptoms in the majority of patients. However, severe toxicity is reported with the most common symptoms being central nervous system depression, serotonin toxicity, seizure or cardiac conduction abnormalities. Although the exact mechanism of injury is unclear, total serum concentrations of venlafaxine and its major active metabolite ODV of 900 μg/L or more combined are likely to cause toxicity. 6,10 Deaths have been reported following large doses and in combination with other medication and alcohol. 9,11
Recently, we reported two cases of interstitial pneumonia with cardiac failure developing in patients treated with venlafaxine. 12 A strong relationship between the development of patients' illness and the initiation of venlafaxine treatment was identified. It was hypothesized that alterations in metabolic drug clearance as a result of genetic factors or drug–drug interactions might, in part, be responsible for the drug-induced toxicity. Unfortunately, no DNA was available for these two cases and therefore CYP450 variants could not be confirmed. However, this observation prompted us to evaluate a possible role of this genetic variant in the presented case. We report a case with a severe refractory depression not responding to venlafaxine in which therapeutic drug monitoring (TDM) in combination with pharmacogenetics was performed in the diagnostic follow-up.
Case report
A 42-year-old woman was visiting the outpatient hepatology clinic because of, since six weeks existing, progressive fatigue, dyspnoea, paresthesias of the fingers, hands and legs. She felt agitated and had an elevated heart rate in rest of 110 beats per minute. She was known with morbid obesity (body mass index [BMI] 46 kg/m2), arterial hypertension, left ventricle hypertrophy with a decreased left ventricle ejection fraction (40% of predicted), depression and non-alcoholic steatohepatitis. The medication she used is summarized in Table 1. At that time haematological and biochemical laboratory investigations revealed no abnormalities. After her last suicide attempt, two months before presentation at the outpatient clinic, the psychiatrist started venlafaxine 75 mg to treat her depression. As no mental improvement occurred, the venlafaxine dose was increased gradually to 225 mg daily, but still no improvement was established. Moreover, her mental situation worsened and so did the other complaints she presented with at the outpatient clinic. The fore-mentioned adverse effects (especially the tachycardia and agitation) coincided the most with the side-effects listed for venlafaxine, see also Table 1. This prompted us to measure the serum venlafaxine level and of its main active metabolite ODV, which were 1300 μg/L and less than 100 μg/L, respectively, well above the recommended combined concentration range of 195–400 μg/L. 8 At the same time, three common polymorphisms in the CYP450 genes (CYP2C9, CYP2D6 and CYP2C19) were analysed, as described previously. 13 These tests were performed to rule out or establish a genetic component. Moreover, the genotyping was used to investigate whether venlafaxine was the most likely cause for the ADR, or one or more of the prescribed co-medication not being metabolized properly by the investigated enzymes. For accumulation of other drugs might be causing additional drug–drug interaction problems. A homozygote variant of CYP2D6 (CYP2D6*4/*4) was found indicating that no functional CYP2D6 enzyme and, therefore, no metabolic activity, was present. Furthermore, a heterozygote variant of CYP2C9 (CYP2C9*1/*2) and a wild-type genotype of CYP2C19 (CYP2C19*1/*1) were found. Mainly the fact that the patient appeared to be a poor metabolizer of CYP2D6 substrates explained the high toxic serum levels of venlafaxine without the presence of its main active metabolite ODV. Venlafaxine was stopped immediately and so was metoprolol, also metabolized by CYP2D6, see also Table 1. 14 Gradually, her complaints of agitation, dyspnoea and tachycardia disappeared.
Specifics of the medication and dosages administered at time of presentation
Type of medication (group), main adverse reactions (ADRs) and CYP route metabolism is depicted
Discussion
This case highlights the importance of CYP450 genotyping in combination with serum drug monitoring to establish a relationship between ADR and drug metabolism. High toxic serum levels of venlafaxine without a detectable concentration of its main active metabolite ODV strongly indicated that no CYP2D6 enzyme activity was present. This was confirmed by genotyping. The complaints with which patient presented herself at the outpatient clinic could be attributed to high levels of venlafaxine and were a direct consequence of the rate of CYP metabolism. 7,15,16 Furthermore, this drug is known to cause serotonin syndrome in some cases. 17 The complaints were not related to metoprolol, which is also metabolized mainly by CYP2D6, see also Table 1. Metoprolol, a selective adrenergic beta-1-blocking agent, was prescribed to the patient because of hypertension. The drug competes with adrenergic neurotransmitters such as catecholamines, resulting in a decreased heart rate, cardiac output and blood pressure. Toxic effects include bradycardia, hypotension, bronchospasm and cardiac failure, which were not observed in our patient. Another rare side-effect of metoprolol therapy can be depressive symptoms, but as the patient was prescribed metoprolol years before the onset of the depression, this was unlikely. 18,19 A more plausible explanation is the fact that patients can also be non-responders to venlafaxine therapy, not linked to a certain CYP450 genotype, but in combination with a poor metabolizer the risk of a maintained depression and acquiring ADRs is increased substantially. 8,20 Other more rare ADRs include pulmonary (e.g. interstitial pneumonia) and cardiovascular (e.g. agina pectoris, sinus arrythmia) problems and even sudden deaths have been reported, suggesting venlafaxine and/or its metabolites to be a possible proarrhythmic agent. 21–23 Direct toxic effect or hypersensitivity is likely to be the mechanism.
Venlafaxine is mainly metabolized by CYP2D6 and only for a very small amount by CYP3A4, see also Figure 1. 8,24 In particular, CYP2D6 variants may manifest phenotypically as poor, intermediate, extensive or rapid metabolizers. 25,26 Administration of venlafaxine to poor metabolizers places them at risk of accumulation of the drug to toxic concentrations. 7 Moreover, poor metabolizers are more likely to experience ADR. 7 Also, co-administration of venlafaxine with drugs that inhibit the activity of CYP2D6, such as some antiarrhythmic agents, could provoke accumulation of the drug and predispose patients to drug-induced damage. 27 However, the venlafaxine datasheet states that co-prescription of venlafaxine and metoprolol, although it should be exercised with caution, poses no problem in a normal situation when checking the patient's blood pressure regularly.
Genotyping prior to administration of the drug probably prevents unnecessary medication for at least six weeks (period needed for evaluating the effect of the medication). Furthermore, and possibly even more important, it warns the psychiatrist that increasing the dose is no option, since the active metabolite will not be formed due to the inappropriate metabolism of venlafaxine. Moreover, it exposes the patient to an increased ADR risk, because of decreased clearance. The risk for development of drug toxicity, including drug-induced lung damage and cardiopulmonary effects, differ depending on a variety of host (including CYP450 polymorphisms) and drug factors. 23,28 Increased serum levels of drugs can be observed, not only after overdose administration of the drug, but also because of decreased clearance in some subjects, due to, as in this case, an incapability to metabolize or drug–drug interactions. 25
The promise of pharmacogenetics, the study of the relationship between variants in a large collection of genes and variable drug effects, lies in its potential to identify the right drug and dose for each individual patient. 29 Thus, genotyping can provide therapeutic and pharmaco-economic benefits and should be considered in patients using (multiple) drugs. 29,30 The affected CYP450 enzyme, causing an inappropriate effect of the antidepressant, can also influence other drugs metabolized by the same CYP450 system. Therefore, physicians should prescribe combinations of certain drugs with caution.
In conclusion, introduction of venlafaxine in our patient resulted in tachycardia and agitation without improvement of the depressive symptoms. It appeared that the patient was a poor metabolizer for CYP2D6, the most important phase I enzyme to metabolize venlafaxine. Discontinuation of this drug resulted in resolving of symptoms. This case highlights the potential benefit of both clinical and genetic risk stratification (pharmacogenetics) prior to treatment, either for setting the individual dose or for making a decision about using a particular drug. To minimalize ADRs, genetic screening prior to prescription of certain ‘risk drugs’ and TDM during treatment would be an ideal situation.
DECLARATIONS
