Abstract
Background
Australian guidelines on the diagnosis of diabetes mellitus (DM) recommend performing an oral glucose tolerance test (OGTT) in people with fasting plasma glucose (FPG) values of 5.5–6.9 mmol/L.
Aim
To evaluate indications, outcomes and confounding factors of OGTTs performed at a large teaching hospital and to compare them with Australian DM guidelines.
Method
A retrospective audit of OGTTs performed over an 18-month period in a teaching hospital in a major Australian city. Information gathered included co-morbidities; medications; risk factors for type 2 DM; indication for OGTT; results of OGTT and previous glucose tests.
Results
All 129 OGTTs identified were included in the audit. Eighty-nine (69%) were male, with a median age of 57 years (range 19–86), and 3% were of Australian Aboriginal ethnicity. An indication for OGTT was identified in 93%, including FPG 5.5–6.9 mmol/L (36%) and random plasma glucose (RPG) 5.5–11.0 mmol/L (19%). Other indications for OGTT identified included polycystic ovary syndrome or metabolic syndrome (8%), peripheral neuropathy (3%) and as part of a research protocol (12%). Forty-two (35%) were inpatients at the time of OGTT, of which 35 (30%) were admitted for acute medical or surgical illnesses such as stroke. Nineteen percent were taking medications known to affect plasma glucose (e.g. oral corticosteroids).
Conclusion
Only 55% of OGTTs had a previous FPG or RPG value warranting OGTT using current Australian DM guidelines. Other valid indications for OGTT were identified in the majority of the remainder. In addition, 41% were performed in the presence of confounding factors (such as acute illness or medications known to affect plasma glucose). Many of the OGTTs that are currently being performed are in the presence of confounding factors that could cause misleading results.
Introduction
Australian guidelines on the diagnosis of type 2 diabetes mellitus (DM) recommend an oral glucose tolerance test (OGTT) in people with fasting plasma glucose (FPG) 5.5–6.9 mmol/L or random plasma glucose (RPG) 5.5–11.0 mmol/L. 1 FPG is the preferred initial test for DM as well as the preferred confirmatory test for a previous abnormal glucose result. The Australian guidelines differ from those of the American Diabetes Association, which do not recommend routine use of OGTT to diagnose DM, and the World Health Organization, which recommend OGTT for FPG 6.1–6.9 mmol/L. 2,3 Despite these differences, there have been no studies in the published medical literature evaluating OGTTs on adherence to current DM guidelines in non-pregnant patients. In this study, we evaluate the indications, outcomes and confounding factors of OGTTs performed at a large teaching hospital over an 18-month period.
Methods
The study was registered as a clinical audit and was conducted retrospectively on all OGTTs performed at Royal Perth Hospital (RPH) over an 18-month period (01/01/2004–30/06/2005). RPH is a large teaching hospital in Perth, Western Australia. It treats a largely adult, non-pregnant population, providing both inpatient (∼70,000 patients/yr) and outpatient (∼225,000 visits/yr) services. 4 OGTTs at RPH are performed using a standard protocol that involves a 75 g oral glucose load with 0, 1 and 2 h plasma glucose collections.
Subjects who underwent OGTT in the study period were identified by electronic search of the laboratory database. Data were then extracted (including a copy of the request form) and patients' medical notes were individually reviewed. The data collected included age, gender, Australian Aboriginal ethnicity, inpatient/outpatient status, co-morbidities, medications, type 2 DM risk factors, indication for OGTT and results of previous glucose tests. DM risk factors collected were age >55 yr, aboriginality, hypertension, cardiovascular disease (including ischaemic heart disease, peripheral vascular disease or stroke), polycystic ovary syndrome, impaired fasting glucose (IFG) and any family history of DM. 1 Inpatients also had admission diagnosis and length of stay recorded.
The indication for OGTT was collected from the pathology request form, medical notes or previous abnormal glucose results. Where more than one indication was identified, a previous abnormal glucose result was taken as the primary indication. Outcomes of OGTTs were analysed based on FPG and 2 h plasma glucose results.
Confounding factors evaluated were hospitalization at the time of OGTT and medications known to affect plasma glucose (i.e. increase or decrease) as defined in a prominent Australian medicines information text. 5 If the effect of a medication on plasma glucose was uncertain (e.g. beta-blockers) or present only at high doses (e.g. thiazide diuretics), the medication was not classed as potentially interfering. 5 The dosage of medications and time of last dose were not recorded.
Statistical analysis was performed using Microsoft Office Professional Edition 2003 (Excel). No external funding source was used.
Results
All 129 OGTTs identified from the laboratory database were included. Eighty-nine (69%) were male and the median age was 57 years (range 19–86). Four subjects were of Australian Aboriginal ethnicity. In seven patients ethnicity was not documented. The majority of patients had multiple medical co-morbidities (median 3 [range 0–7]), including hypertension (50%), cardiovascular disease (40%) and dyslipidaemia (32%). Most (84%) were taking at least one medication (median 4 [range 0–12]), of which anti-hypertensives (49%), lipid-lowering agents (44%) and anti-platelet drugs (40%) were the most common. In eight patients information on medications was not available (including four referrals from other laboratories).
DM risk factors were identified in 110 (85%) subjects. The most prevalent were age ≥55 years (58%), hypertension (50%), cardiovascular disease (40%), IFG (22%) and a family history of DM (13%).
An indication for OGTT was identified in 120 patients (93%), including FPG 5.5–6.9 mmol/L in 36% and random plasma glucose (RPG) 5.5–11.0 mmol/L in 19% (Figure 1). The OGTT was used to ‘confirm’ a diagnosis of DM (based on previous abnormal FPG or RPG) in 11% and as a part of a research protocol in 12%. Of those with no identifiable indication for OGTT, six patients had no record of previous FPG or RPG and three had a previous RPG <5.5 mmol/L.
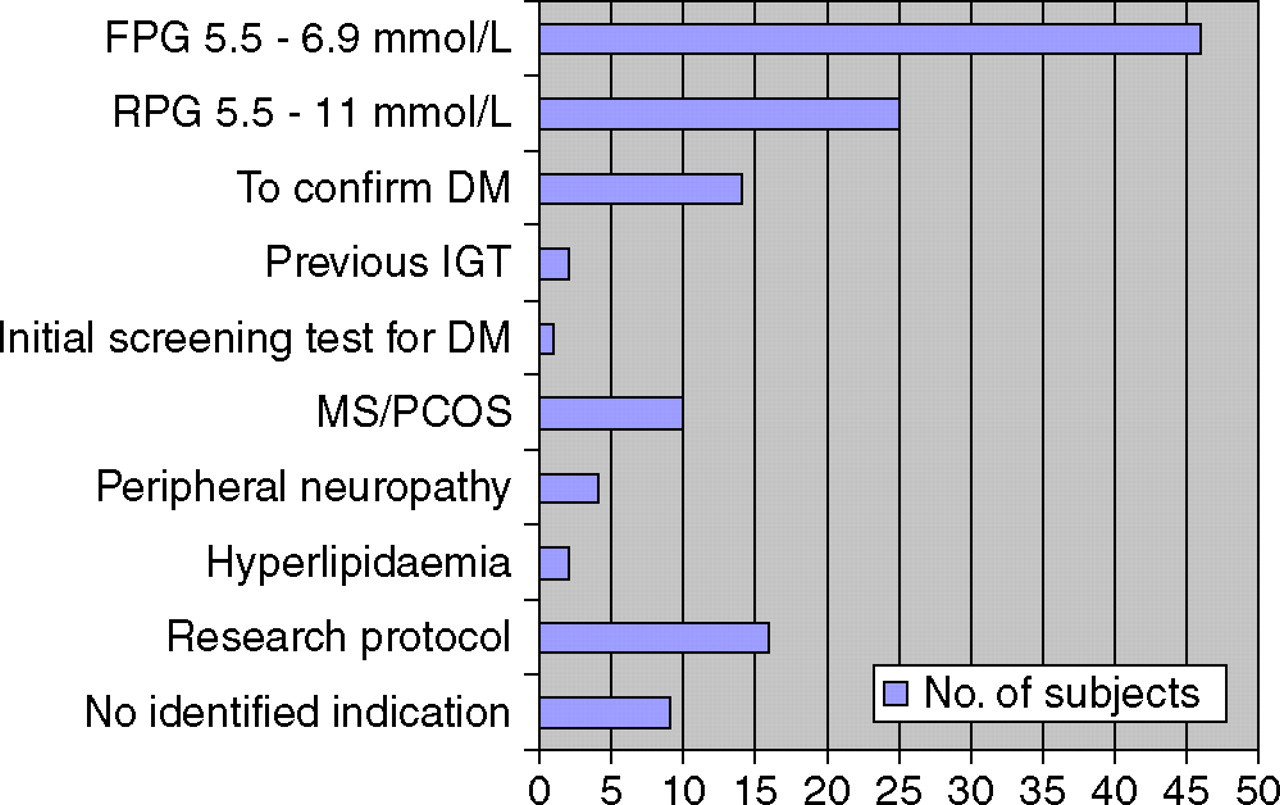
Indications for OGTT from 129 patients who underwent OGTT in the study period. DM, diabetes mellitus; FPG, fasting plasma glucose; IGT, impaired glucose tolerance; MS, metabolic syndrome; OGTT, oral glucose tolerance test; PCOS, polycystic ovary syndrome; RPG, random plasma glucose
Potentially confounding factors were identified in 53 patients (41%). Forty-two subjects (33%) were in patients at the time of OGTT. Of these, 35 patients had been admitted for acute medical or surgical illnesses, including stroke (22 patients), myocardial infarction (4 patients) and acute infection (3 patients). The median length of stay for inpatients was 13 days (range 1–57). Twenty-four patients (19%) were taking medications known to affect plasma glucose, including oral corticosteroids (8%) and tricyclic antidepressants (5%, Table 1). 5
Medications affecting plasma glucose in study subjects
Medications known to affect plasma glucose that were identified in 24 of 129 study participants who underwent OGTT in the study period. OCP, oral contraceptive pill; TCA, Tricyclic antidepressants
*These medications may lower plasma glucose 5
The outcomes of OGTTs were analysed based on FPG and 2 h plasma glucose results (Table 2). In subjects with a baseline FPG of 5.5–6.0 mmol/L, 32% gave an abnormal 2 h glucose compared with 60% and 24% in those with FPGs of 6.1–6.9 and <5.5 mmol/L, respectively.
Outcomes of OGTTs based on FPG and 2 h plasma glucose
Results of OGTTs from 129 study subjects – interpreted using current Australian guidelines 1
ADA, American Diabetes Association; DM, diabetes mellitus; FPG, fasting plasma glucose; IFG, impaired fasting glucose; IGT, impaired glucose tolerance; OGTT, oral glucose tolerance test; PG, plasma glucose; WHO, World Health Organisation
*ADA criteria classify FPG 5.6–6.9 mmol/L as IFG 2
†WHO guidelines do not recommend OGTT in this group 3
Discussion
The AusDiab study demonstrated the alarming rates of DM (7.4% prevalence) and impaired glucose tolerance (IGT, 10.6% prevalence) in Australian adults, many of which are undiagnosed. 6 OGTTs are an important part of the stepwise approach to the diagnosis of DM and ‘prediabetes’ (IGT and IFG) recommended in the Australian DM guidelines. 1
The audit of OGTTs performed at a large teaching hospital found a discrepancy between current DM guidelines on OGTTs and clinical practice. There are no studies in the published medical literature evaluating OGTTs on adherence to current DM guidelines in non-pregnant patients. One Australian audit of OGTTs from a suburban Sydney laboratory in 1990 compared baseline FPG to DM guidelines; however, including pregnant patients. 7 In our study, a non-pregnant adult population was used and OGTT indications and confounding factors were compared with current Australian guidelines. Australian guidelines on the diagnosis of type 2 DM recommend OGTT in people with FPG 5.5–6.9 mmol/L or RPG 5.5–11.0 mmol/L. However, only 55% of OGTTs in the study had previously documented FPG 5.5–6.9 mmol/L or RPG 5.5–11.0 mmol/L. Some patients may have had previous glucose estimations performed at other laboratories, which, unless documented in patients' notes, were not available to this study. OGTT is an unpleasant procedure that is potentially dangerous in patients with uncontrolled DM, and therefore should be undertaken only when it is appropriately indicated. Measurement of FPG would obviate the need for OGTT in many cases by either diagnosing or ruling out DM based on FPG itself. It is not the current practice of this laboratory to ascertain these facts (or to vet OGTT requests) before performing an OGTT when requested by clinicians.
The relationship of the outcome of OGTT to fasting glucose is similar to those of previously published Australian OGTT results. 8,9 Subjects with a normal FPG <5.5 mmol/L have only a 5% chance of being diagnosed with diabetes as a result of an OGTT. Given that these are selected cases with a high clinical suspicion of DM, the yield in the general population for the diagnosis of DM in those with FPG <5.5 mmol/L would be very low and therefore not cost-effective to recommend OGTT. At the other end of the spectrum those with FPG >7.0 mmol/L have a very high likelihood of being diagnosed with diabetes, supporting the use of an FPG cut-off of 7.0 mmol/L for the diagnosis of DM. On the other hand, subjects with a FPG values of 5.5–6.0 mmol/L and 6.1–6.9 mmol/L have a 9% and 26% likelihood, respectively, of being diagnosed with DM at OGTT. These data support the current Australian DM guideline recommendation to perform OGTT in those with an FPG between 5.5 and 6.9 mmol/L. 1
One third of OGTTs were performed on inpatients, many admitted with acute illnesses (e.g. stroke). Both acute illness and hospitalization are stressors that are known to affect plasma glucose adversely. 1,10 Australian DM guidelines (from NHMRC) recommend avoiding confounding factors when performing OGTT. 1 The following is stated in the document: ‘Plasma glucose measurement may be affected by acute intercurrent illness (e.g. myocardial infarction …). These situations may introduce additional uncertainties in making a diagnosis of diabetes. Minor elevations of plasma glucose should be interpreted with caution and testing should be repeated after recovery from the acute episode.’ (page 79). 1 The prolonged length of stay of many of the inpatients in the study may have allowed OGTT to be performed when less acutely unwell (e.g. rehabilitation phase following stroke). In our audit, 19% of patients were taking medications that can also affect plasma glucose (e.g. corticosteroids), although dosage and time of last dose were not recorded. Further research in this area is indicated to study the effects of confounding factors that are listed as causing misleading OGTT results, in order to determine if patients who have a diabetic OGTT result in the presence of these confounding factors in fact have clinical outcomes similar to patients with DM. In some clinical situations, undertaking OGTT while on potentially interfering medications may be appropriate (e.g. psychiatric patients on long-term atypical antipsychotics). 11
One limitation of the study design was that clinical information was collected retrospectively from medical records. Incomplete data collection was minimized by reviewing each patient's medical notes, OGTT request form and previous glucose results. While this study was conducted in a hospital setting, the findings on confounding factors for OGTTs are applicable to general practice as well (where the majority of OGTT requests originate).
In conclusion, only just above half of the subjects in the audit had a previous FPG or RPG value warranting OGTT using current Australian DM guidelines (although other valid indications for OGTT were identified in the majority of the remainder of the patients). Furthermore, the OGTTs were frequently performed in the presence of potentially confounding factors such as hospitalization for acute illness and diabetogenic medications. Laboratories may consider vetting requests for OGTTs to check adherence to guidelines for their performance if this is not currently done.
