Abstract
Assisted reproductive technology has shown rapid advancement since the birth of the first ‘test-tube’ baby in Oldham, UK, in 1978. Since April 2005, women between the ages of 23 and 39, who meet the described eligibility criteria, are able to get one free in vitro fertilization cycle funded by the National Health Service. Private treatment costs anything from £4000 to £8000 for a single cycle of treatment. Almost 15% of the couples in UK are affected by fertility problems and undergo detailed investigations before being offered assisted conception. Assisted reproduction is the collective name for treatments designed to lead to conception by means other than sexual intercourse. These include intrauterine insemination, in vitro fertilization, intracytoplasmic sperm injection and gamete donation. This review is intended to summarize the principles of assisted conception and examine the role of the biochemistry laboratory in: (A) the diagnosis and subsequent management of ovulatory disorders; (B) assessing ovarian reserve before initiating fertility treatment and (C) monitoring fertility treatment. It touches on the screening of potential gamete donors and follow-up of children born after assisted conception. This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association of Clinical Biochemistry.
Introduction
The first attempts at successful in vitro fertilization (IVF) dates back to 1935, when Gregory Pincus claimed to have achieved successful mammal birth as a result of IVF of rabbit eggs. However, in his experiments, the actual process of fertilization took place in the fallopian tube and not in vitro. 1 In 1959, Min Cheuh Chang pioneered the IVF of black rabbits' eggs with black rabbits' sperm, transferred them to a white rabbit and was able to produce a litter of young black rabbits. 1 His work paved the way for the birth of Louise Brown, the world's first baby to be born by IVF in Oldham, UK, on 25 July 1978. 2 Despite being relatively new, the science of assisted conception has had an immense impact on human reproduction. Around the world, more than 200,000 babies are born every year from assisted reproductive technology (ART). 3
Fertility problems affect one in seven couples in the UK. Most couples (about 84 out of every 100) who are trying to have a baby, having regular sexual intercourse (every 2–3 d) and not using contraception, will achieve success within a year. 4 Subfertility is defined as failure to conceive after 1 yr of unprotected regular sexual intercourse. Although this would be the usual time to start investigations, earlier referral may be justified where there are important clinical factors in either partner's history (e.g. pelvic inflammatory disease in the female, previous urogenital surgery in the male or history of sexually transmitted disease in either). 5 A typical primary care trust, health board or strategic health authority may therefore expect to see around 230 new consultant referrals (couples) per 250,000 head of population per year. 4
The detailed investigations of infertility have been discussed elsewhere 5 and will be briefly mentioned here. Table 1 outlines the preliminary investigations of the infertile couple. Further investigations in the females include assessment for patency of the fallopian tubes and the uterine cavity by one or more of the following: (i) hysterosalpingography; (ii) hysterosalpingo-contrast sonography; and (iii) laparoscopic dye test. In the males, if semen analysis confirms azoospermia (or oligozoospermia), further investigations include: (i) serum follicle-stimulating hormone (FSH), luteinizing hormone (LH), testosterone, prolactin, thyroid-stimulating hormone (TSH); (ii) centrifugation of the ejaculate and examination of the pellet for spermatozoa; (iii) karyotyping (if total motile count is <5 × 106/mL); and (iv) testicular exploration or biopsy. 5
Initial investigations of the infertile couple
Adapted from Taylor 5
Assisted reproduction
ART is the collective name for treatments designed to lead to conception by means other than sexual intercourse. ART techniques include intrauterine insemination, IVF, intracytoplasmic sperm injection (ICSI), donor insemination and gamete donation. 6 This review is intended to summarize the principles of ART and examine the role of the biochemistry laboratory in the diagnosis and subsequent management of ovulatory disorders.
Role of the clinical laboratory in monitoring various aspects of the assisted conception process
(A) Assessing ovulation
Regular menstrual cycles in the range of 26–36 d are usually indicative of ovulation. 7 Ovulation involves luteinization of the mature follicle and release of the oocyte. Both are triggered by the LH surge. Patient-monitored basal body temperature charts are not sufficiently reliable for the detection of ovulation. 8 Urinary LH kits may be used as a point of care test (POCT) and are considered a reliable test for predicting ovulation in the ensuing 48 h. 9 In clinical practice, ovulation is confirmed retrospectively by measurement of serum progesterone concentration in the mid-luteal phase, approximately on day 21 of a 28-day cycle. For women with irregular cycles, this test may need to be performed later in the cycle (e.g. day 28 of a 35-day cycle) and repeated weekly until the next menstrual cycle starts, unless the bleeds are so infrequent that ovulation induction therapy would be needed in any case. Historically, values ranging from 16 nmol/L to 28 nmol/L (depending on the analytical platform) have been proposed as the lowest limit indicative of ovulation. 10–12 However, progesterone concentration is dependent on factors other than the phases of the menstrual cycle and the method of analysis: modest physical exercise and calorie restriction may lower progesterone concentrations sufficient to prevent ovulation. 13 Interestingly, similar lower progesterone concentrations in women from deprived areas, suffering from chronic malnutrition and involved in manual labour are not associated with reduced fertility; this is suggestive of significant interpopulation differences in progesterone concentrations consistent with normal ovulation. 14 In a cohort of infertile women undergoing ovulation induction by human menopausal gonadotrophin (hMG), which is a purified extract from human postmenopausal urine and contains both FSH and LH, the minimum progesterone concentration consistent with a full-term pregnancy was found to be about 34 nmol/L. There was no statistically significant difference in the progesterone concentrations between the cycles that resulted in full-term pregnancy and those that ended in miscarriage; full-term multiple pregnancies were associated with a higher mean progesterone concentration than full-term singleton pregnancies. 15
Anovulation and oligo-ovulation are ovulatory disorders that are estimated to cause 21% of female infertility. 4 The World Heath Organization (WHO) classifies ovulation disorders into three groups. 16
Group 1
Hypothalamic pituitary failure (hypothalamic amenorrhoea or hypogonadotrophic hypogonadism)
This group of disorders is characterized by low gonadotrophins, normal prolactin and low oestrogen, and it accounts for about 10% of ovulatory disorders. Failed ovarian follicular development results in hypo-oestrogenic amenorrhoea. This group includes women who indulge in strenuous exercise related to a wide variety of athletic activities, as well as those with low body weight (including anorexia nervosa). Reduction in physical activity and restoration of body weight may help to resume ovulation and restore fertility. Other treatment options include pulsatile gonadotrophin-releasing hormones (GnRH) (which induces the appropriate release of pituitary FSH and LH) and hMG.
Group 2
Hypothalamic pituitary dysfunction
This group, which is characterized by normal oestrogen concentrations, accounts for about 85% of ovulatory disorders. It results in anovulatory oligo/amenorrhoea, predominantly involving women with polycystic ovaries. The definition for the diagnosis of a polycystic ovary 17 (which is usually obtained from an ultrasound scan) requires the presence of at least 12 follicles measuring 2–9 mm in diameter and/or an ovarian volume in excess of 10 cm3. In women who have polycystic ovaries, where there are associated clinical symptoms (such as menstrual cycle disturbances, obesity and hyperandrogenism presenting as hirsutism, acne or androgen-dependent alopecia), this is referred to as the polycystic ovarian syndrome (PCOS). About 30% of the PCOS population is of normal weight. 18
The diagnosis of PCOS is fraught with inconsistencies and the current criteria (see below) are not considered robust enough for either clinical or research purposes. 19,20
The European Society for Human Reproduction and Embryology (ESHRE) and the American Society for Reproductive Medicine (ASRM) define PCOS when at least two out of the following three features are present
21,22
:
Oligo-ovulation and/or anovulation Clinical and/or biochemical hyperandrogenism (see below) Polycystic ovaries, with the exclusion of other aetiologies
Before treatment is started, weight loss and exercise should be recommended in obese women. First-line medical treatment for women with WHO Group II ovulatory disorder, such as PCOS, is ovulation induction with the anti-oestrogen clomiphene citrate for up to 12 months. Clomiphene occupies the oestrogen receptors in the hypothalamus, interfering with the normal feedback mechanisms, increasing gonadotrophin secretion and thereby stimulating the ovaries to produce more follicles.
4
Second-line options include laparoscopic ovarian surgery or treatment with gonadotrophins.
23
The use of gonadotrophins is associated with increased chances of multiple pregnancies. Laparoscopic ovarian surgery involves making multiple ovarian punctures performed either by diathermy or laser, which may help to correct the endocrine abnormalities and trigger ovulation. On its own it is effective in <50% of the women, but has the advantage of not being associated with multiple pregnancies (thereby not requiring intensive post-treatment monitoring). Although metformin has been recommended by some, recent evidence shows that it is not effective in inducing ovulation.
24
Hyperandrogenism
Androgen sources in women are the adrenals and ovaries and peripheral tissues such as fat and skin. 25 The liver and gut play a minor role in androgen production, particularly in the peripheral conversion of testosterone to the most active form dihydrotestosterone (DHT). Testosterone, dehydroepiandrosterone sulphate (DHEAS), dehydroepiandrosterone (DHEA), androstenedione and androstenediol are the five androgens produced in the body. Testosterone is the only androgen with direct androgenic activity, while DHEAS, DHEA and androstenedione are all precursors of testosterone. Androstenediol has both androgenic and oestrogenic activity. About half of the total testosterone is secreted by the ovaries under the control of LH, which further increases during the mid-cycle LH surge. Nearly one-quarter of the circulating concentrations of testosterone are produced by the adrenal glands. Although testosterone is used as a marker of ovarian androgen secretion, peripheral conversion of androstenedione and DHEA to testosterone may also contribute significantly to the circulating total testosterone. The ovaries also secrete about half of the total androstenedione and small amounts of DHEA.
The adrenal glands produce all the DHEAS and a large proportion of DHEA. The former is a useful marker of adrenal androgen secretion. The adrenals also secrete about half of the androstenedione.
In the circulation, approximately 97% of testosterone is protein-bound, principally to sex hormone-binding globulin (SHBG) and to a lesser extent albumin and other proteins. Only free testosterone is available to tissues. Since measurement of the free testosterone concentration is analytically challenging, calculation of the ratio of testosterone/SHBG concentrations provides an index of free testosterone status (free androgen index). 26
The pathogenesis of PCOS (Figure 1) is almost certainly multifactorial. 27 Both LH and FSH control hormone synthesis by the ovaries, the former regulating androgenic synthesis in the theca cells, while the latter aromatase activity of granulosa cells, which determines conversion of androgenic precursors to oestrogen. In PCOS, the ovarian theca cells increase their ovarian androgen production under the stimulatory activity of the raised LH concentrations, which may, in part, be due to an increased pulse frequency of GnRH. The relatively low FSH results in decreased aromatase activity. The net result is preferential androgen synthesis.
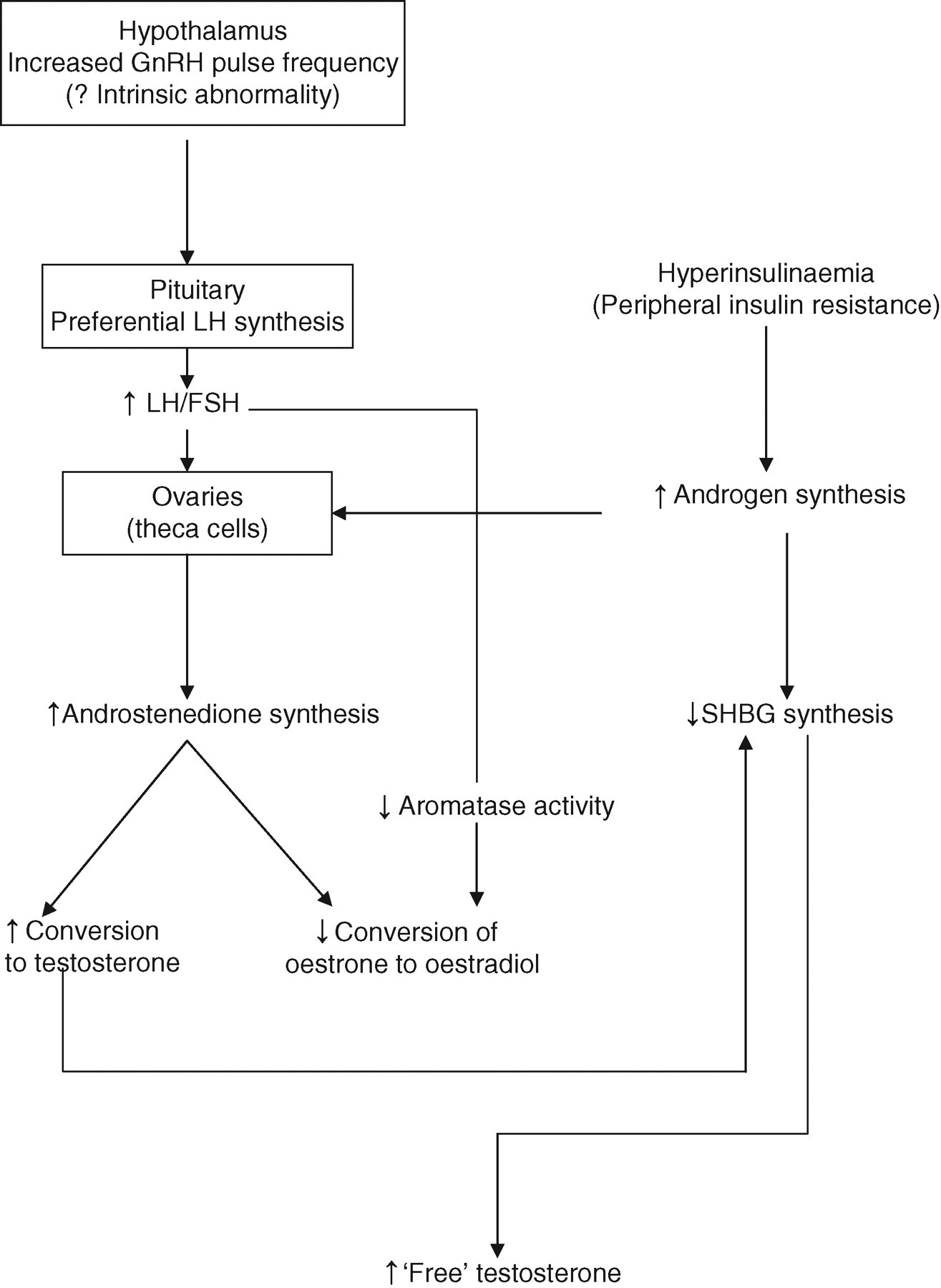
Hyperinsulinaemia due to peripheral insulin resistance is often present in these women and it also promotes hyperandrogenaemia through the binding of insulin to the insulin-like growth factor-1 (IGF-1) receptor. Insulin mimics the action of IGF-1, which augments androgen production by the theca cell in response to LH. Since insulin decreases the concentrations of SHBG, the circulating concentration of free testosterone is also increased.
Group 3
Ovarian failure
This group, which is characterized by high gonadotrophins with hypogonadism and low oestrogen, accounts for about 4–5% of ovulatory disorders. 16 Aetiology is usually idiopathic; known causes include autoimmune disorders, chromosomal abnormalities (Turner's syndrome, androgen insensitivity syndrome) and iatrogenic causes [chemotherapy, radiotherapy or surgical removal]). Diagnosis is based on an elevated FSH (>40 U/L) in two samples more than 1 month apart. 28 Treatment includes hormone replacement therapy to reduce menopausal symptoms and loss of bone density as a complication of the hypo-oestrogenic state. The only fertility treatment available is oocyte donation.
(B) Assessing ovarian reserve
Before embarking on fertility therapy involving complex ovulation induction procedures or participating in ART cycles, it is important to test the ovarian reserve to predict a woman's reproductive potential. 4 Ovarian reserve is defined as the number and quality of follicles in the ovary at any given time. There is an age-related progressive decline in oocyte quality and quantity. The purpose of ovarian reserve tests (ORTs) is to identify women with poor ovarian reserve for their age. These tests may also be useful in the classification and management of younger women who respond poorly to ART cycles. Table 2 lists the currently available ORTs. Most fertility treatment centres have their own protocols for investigating women before assisted conception and there may be considerable variation from one centre to another.
Ovarian reserve tests
Early follicular-phase FSH can be measured, either in all women or selected women over a certain age. There is no consensus as to the lowest cut-off value, above which early-follicular FSH may definitely predict poor ovarian reserve. However, women undergoing IVF with a day 3 FSH of <15 U/L are twice as likely to conceive as women with FSH values between 15 U/L and 24.9 U/L. 29 The sensitivity and specificity of basal (unstimulated – cf. dynamic function tests below) FSH, for the prediction of poor ovarian response and non-pregnancy, is very variable; on both counts, accuracy of basal FSH, in regularly cycling women, is adequate only at very high threshold concentrations. However, such high FSH values are rarely seen in practice; therefore the test has hardly any clinical value. 30 FSH, like many of the other ORTs, may only be useful as a screening test for counselling purposes and further diagnostic steps. 31 Similarly, a low (<290 pmol/L) day 3 oestradiol concentration, independent of FSH concentrations, has been associated with improved stimulation response and higher pregnancy rates. 32 However, it has a modest likelihood ratio (4–5) for predicting ovarian response. At the same time, it is difficult to identify a clear threshold concentration of basal oestradiol, for prediction of non-pregnancy. Therefore, it has very limited clinical applicability as an ORT. 30
Other less widely available hormonal markers include inhibin-B and anti-Müllerian hormone (AMH). Inhibin-B is secreted by antral follicles in response to FSH and is the major marker of follicular growth (and ovarian function); it inhibits FSH release by negative feedback. It is also involved in the control of feto-maternal communication required to maintain pregnancy (in males, Inhibin-B expression and secretion are positively correlated with Sertoli cell function, sperm count, and spermatogenic status and are negatively correlated with FSH). 33 Although present in ovulating women, it is not normally found in postmenopausal women. It has been observed that day 3 inhibin-B concentrations of <45 pg/mL were associated with a poorer response to fertility treatment and reduced pregnancy rate. 34 However, even very low concentrations of inhibin-B are associated with only modest accuracy (at a likelihood ratio of approximately 4) for the prediction of poor ovarian response or non-pregnancy, thus limiting its clinical application. 30
Embryologically, both male and female fetuses have two pairs of ducts (Wolffian and Müllerian), which ultimately differentiate to form the internal genitalia. In males AMH influences the regression of the Müllerian duct, which appears to be an important physiological function. In adult females, AMH is produced by the granulosa cells of the preantral and small antral follicles and its serum concentration is a marker of the growing follicle pool. Declining number of ovarian follicles with increasing age is associated with falling serum concentrations of AMH making it a novel measure of ovarian reserve. 35 Serum AMH concentration seems to be a better predictor of ovarian reserve and ART outcome than FSH, inhibin-B or oestradiol. 36 Using a cut-off of <1.4 μg/L it has been found to have a sensitivity and specificity of 76% and 86%, respectively, for prediction of poor ovarian response for IVF treatment. Although this equates to a modest performance, its main potential advantage is the ability to be measured throughout the cycle, in contrast to the other parameters, which can only be determined in the early follicular phase. This may also lead to its wider application in primary care. 37
In addition to single hormone measurements, various dynamic tests have also been used as ORTs.
The clomiphene citrate-challenge test (CCCT) involves measurement of basal FSH and oestradiol on day 3 of the cycle, administration of 100 mg of clomiphene citrate daily from day 5 to day 9 (both inclusive), followed by measurement of FSH concentration on day 10. The test is interpreted as being abnormal if either basal or day 10 FSH is greater than 10 U/L. Clomiphene citrate acts as an oestrogen antagonist on the pituitary gland, where it blocks the action of circulating oestrogen. The rationale behind the CCCT is that patients with adequate ovarian reserve would demonstrate rising concentrations of oestradiol and inhibin-B in the early follicular phase, which would restrict the clomiphene-induced FSH rise.
38
Depending on the FSH threshold concentrations, the sensitivity of CCCT varies from 35% to 93% and the specificity from 47–98%. Although it was found to have good specificity in most studies, it is not used routinely now due to the low sensitivity. Although recent evidence suggests that it may be a good predictor of poor responders (non-pregnancy) to IVF therapy,
39
it does not seem to add significantly to the simpler antral follicular count (AFC) ultrasound test in the prediction of poor ovarian response.
40
The exogenous FSH ovarian reserve test (EFORT), involves injecting 300 IU of purified FSH intra-muscularly on day 3 of the menstrual cycle. Blood samples are collected just before the injection to measure plasma FSH and oestradiol; 24 h later another blood sample is collected for measurement of plasma oestradiol to determine the post-stimulation increment in oestradiol concentration. In one study,
41
a ‘normal’ response (basal FSH <11 U/L and oestradiol increment >110 pmol/L) was associated with adequate response to ovarian stimulation in 90% of the women. Despite good predictive value for pregnancy, the specificity was still low at 60%. Gonadotrophin agonist stimulation test (GAST) evaluates the rise in serum oestradiol from day 2 to day 3 of the cycle after administration of a GnRH agonist; the latter causes a transient increase in pituitary secretion of FSH and LH. At a specificity rate of approximately 90%, GAST appears to have a sensitivity of approximately 80% and a positive predictive value of approximately 82% for poor responders to IVF.
42
However, on comparing the predictive accuracy and clinical value of the GAST with a day 3 AFC on ultrasound and serum inhibin-B concentration, neither a single nor a repeated GAST performed better. In addition, it had no statistically significant relationship with ongoing pregnancy.
42
It is beyond the scope of this review to discuss the AFC, ovarian volume (OVVOL) and ovarian blood flow, which are measured ultrasonographically. A recent systematic review
30
demonstrated that individual ORTs have only modest-to-poor predictive properties, and though mostly inexpensive, are unsuitable for routine clinical use. An ideal ORT should be able to identify a substantial percentage of patients being considered for IVF, who have virtually no chance of becoming pregnant in a series of treatment cycles, due to the loss of ovarian reserve. These patients can be advised against entering an ART programme, due to the high costs and minimal expected results.
37
As yet there is no single test available to accurately predict ovarian reserve. A combination of ultrasonographic and hormonal tests is likely to have more predictive accuracy and clinical value.
43,44
Predictably, accuracy of testing for poor ovarian reserve is better than that for occurrence of pregnancy, since a successful pregnancy after IVF depends on many other factors. It has been suggested that as poor ovarian response would provide some information on ovarian reserve status, the first cycle of IVF should be entered without any formal prior testing.
30
(C) Monitoring ovulation induction
The aim of ovulation induction therapy is to stimulate the ovaries to produce one or more eggs. Ovarian monitoring provides information about the ovarian response to ovulation induction agents by ascertaining the number and size of the developing follicles. Ultrasonography is regarded as a safe, accurate and efficient method of monitoring follicular development in response to ovulation induction, 45 and in helping to reduce multiple pregnancy rates, especially in women with PCOS. 46 Oestrogen monitoring provides no additional information compared with ovarian ultrasound. 47 It is useful in detecting the ovarian hyperstimulation syndrome (OHSS), though ultrasonography has equally good predictive potential. 48 A recent Cochrane review 49 concluded that there was no evidence to support that the monitoring of stimulated cycles in IVF and ICSI by a combination of ultrasound and serum oestradiol concentrations was more effective than ultrasound alone on outcomes of pregnancy rate and live births. However, since it would be very difficult to design a randomized trial of sufficient sample size to compare the two monitoring strategies, and there was a small but definite degree of inter-observer variability in interpreting ultrasound, the review suggested that the current practice of combined monitoring should be continued. 49
Ovarian hyperstimulation syndrome
Ovulation induction carries the risk of overstimulation and OHSS- an exaggerated response to ovulation induction therapy. OHSS is typically associated with exogenous gonadotrophin stimulation and is rare with the use of other agents. 4,50 It is usually a self-limiting disorder lasting for a few days. There is a broad spectrum of clinical features. Mild illness manifests as lower abdominal discomfort, nausea, vomiting and diarrhoea requiring outpatient clinical observation. Severe OHSS is a potentially fatal condition leading to rapid weight gain, hypotension, tachycardia, tachypnoea, renal impairment, ascites, pleural and pericardial effusion, haemoconcentration and coagulopathy. 50 Severe OHSS affects around 3% of cycles when hMG is used 51 and 0.2–1.0% of all ART cycles. 52
The characteristic pathological feature of OHSS is increased capillary permeability resulting in a fluid shift from the intravascular space to third space compartments. 53,54 The cause of this altered capillary permeability is not clear. Vascular endothelial growth factor (VEGF) has been implicated in the pathophysiology of OHSS. 55,56 VEGF plays an important role in follicular growth and ovarian angiogenesis and its concentrations correlate with the severity of OHSS. 55,56 Other factors which have been implicated in the genesis of OHSS include IGF-1, angiotensin-mediated altered capillary permeability, increased follicular fluid concentrations of prorenin and renin and increased secretion of protein-rich fluid from enlarged ovaries or peritoneal surfaces. At the cellular level, various cytokines and growth factors may be involved either directly or indirectly via VEGF. 55,57
Table 3 lists the risk factors for the development of OHSS. Oestradiol concentrations and ultrasound follicular tracking are used to help predict which patients will develop OHSS. The severity of OHSS is classified by a combination of clinical and laboratory findings, with features of severe OHSS including clinical ascites, oliguria, raised haematocrit, hypoproteinaemia and ultrasound scan measurement of ovarian size over 12 cm. Laboratory investigations (serum creatinine and electrolytes, liver function tests, haemoglobin and haematocrit) are used not only to determine the severity, but also to track the progress of the condition. Treatment of mild/moderate OHSS is mainly supportive – analgesia and adequate hydration. However, in more severe OHSS, inpatient care is required, with treatment options including intravenous fluid management, paracentesis and thromboprophylaxis. 58
Risk factors for developing ovarian hyperstimulation syndrome 74
(D) Assessing and monitoring in vitro fertilization
It is recommended that prior to undergoing IVF, individuals (both male and female) should be screened for blood-borne viruses (BBV) – Hepatitis B, Hepatitis C and HIV. Women should be offered screening for Chlamydia trachomatis and if found positive, both partners should be referred for treatment and contact-tracing. 4
IVF involves switching off the natural ovulatory cycle to facilitate controlled ovarian stimulation using gonadotrophins to encourage the development of several follicles. This is followed by administration of human chorionic gonadotrophin (hCG) (as a surrogate LH surge) to mature eggs ready for collection. The collected eggs undergo IVF and the resulting embryos are transferred to the uterus. Luteal support is provided by the administration of hormones to aid implantation of the embryos and the stimulated cycles are carefully monitored.
The purpose of monitoring ovarian response by ultrasound is two-fold. First, to optimize the timing of luteinization before oocyte retrieval and second, to ensure safe practice in reducing the incidence and severity of OHSS. It is common practice to do three ultrasound scans: at the start of ovarian stimulation in GnRH agonist-controlled cycle, to assess follicular growth and development of OHSS at day 7 to day 9, and to determine timing of hCG administration at days 11–14. 4
It has been suggested that in spontaneous pregnancies, luteal and placental progesterone production may be inadequate for many days before a spontaneous abortion or ectopic pregnancy is detected; serum progesterone has been advocated as a biochemical marker for the early detection of potentially non-viable pregnancies. 59 Although on its own this cannot be extrapolated to assisted conception, there is evidence that in women undergoing IVF/ICSI, elevated serum progesterone concentrations predict viable intrauterine pregnancies; exogenously supplemented progesterone is unable to rescue a pregnancy destined to result in a miscarriage. Therefore, in early gestation, serum progesterone measurement may potentially be a very useful marker of pregnancy outcome in women undergoing IVF or ICSI. 60
The diagnosis of pregnancy following IVF is done by hCG measurements (urine or serum) around 10 d after embryo transfer followed by ultrasonography at 4–5 weeks. 61
If the test is done too early, a false-positive pregnancy test may result from the administered hCG. As for a spontaneous pregnancy, serial hCG measurements and ultrasonography are carried out if ectopic pregnancy is suspected.
(E) Screening gamete donation
The laboratory plays an important role in screening for genetic and infective disorders during the process of gamete donation. The Human Fertilisation and Embryology Authority (HFEA) recommends that the screening process should be carried out in accordance with current professional guidance produced by relevant professional bodies (British Andrology Society and British Fertility Society). This includes testing for BBV (see above), cytomegalovirus (CMV), chlamydia and syphilis. Karyotyping, and depending on the donor's family history and ethnicity, screening for cystic fibrosis, β-thalassaemia, sickle-cell disease and Tay-Sach's disease are also recommended. 62 All licensed clinics are now required to inform couples whether or not a donor has been tested for cystic fibrosis and of the risks for any child who may be born from fertility treatment.
(F) Follow-up of children born as a result of assisted reproduction
The number of children born as a result of ART is rapidly increasing. In 2004, ART was responsible for 1% of all children born in the USA. 63 In England and Wales, there were approximately 8000 babies born as a result of IVF and/or ICSI in 2001, accounting for 1.3% of all live births. 64 There is developing concern about the potential to transmit genetic abnormalities and the long-term consequences on later development of children born as a result of assisted conception. A systematic review 65 analysed five clinical outcomes: congenital malformations, growth disturbances, neurological development disturbances, chromosomal abnormalities and transmission of subfertility to male offspring. Overall, no increased risk of major birth defects, including chromosomal abnormalities, was found in children resulting from ICSI when compared with those conceived by standard IVF or naturally. Similarly, a small increase found in congenital malformations in children born after IVF was attributed to maternal characteristics and not to any aspect of the IVF procedures. 66 However, more recently, a multicentric cohort study found higher risk of congenital malformations, especially those involving the urogenital system in boys, in children born after ICSI. IVF and ICSI children were also more likely to have a significant childhood illness, surgical operation or hospital admission. 67 Interestingly, children born after assisted conception may have a lower risk of infantile autism. 68 There is conflicting evidence about increased risk of development of retinoblastoma following IVF. 69,70 Current literature suggests that there is no evidence to link any increased cancer risk with ART. 71,72 Long-term longitudinal follow-up of children resulting from ART is needed. With the ever-expanding cohort of children born out of assisted conception, the frequency of genetic testing is likely to increase.
