Abstract
Background
In vitro haemolysis is a common occurrence in clinical laboratories and causes a spurious increase in potassium. In the past, haemolysis was sought by visual inspection but is now commonly detected by automated measurement of the haemolytic index (HI). This study compared detection of haemolysis in adult and neonatal samples by inspection and measurement of HI and verified that a single equation is appropriate to correct for the increase in potassium in both haemolysed samples.
Methods
Laboratory staff inspected samples for haemolysis and their observations were compared with the measured HI. The potassium concentrations and haemolytic indices of 613 adult and 523 neonatal samples were correlated to derive equations to compensate for the increase in potassium with increase in HI. These were found not to differ significantly and a single equation for use in both populations was derived.
Results
The presence of icterus was found to decrease ability to detect haemolysis on inspection. The mean (95% confidence limits) potassium increase per unit HI was 0.0094 mmol/L (0.0078–0.0103 mmol/L) for adults and 0.0108 mmol/L (0.0094–0.0121 mmol/L) for neonates. The equation developed to compensate for potassium release in haemolysed samples was: adjusted potassium = measured potassium − (HI in μmol/L × 0.01).
Conclusion
The use of HI rather than visual inspection is particularly recommended in neonates whose serum tends to be icteric. It can be used in the same correction equation as in adults to compensate for potassium released due to haemolysis and facilitate reporting a qualitative comment to assist in immediate clinical management.
Introduction
Haemolysis is defined as the release of intracellular components of blood cells into the extracellular space of blood. 1 It can occur in vitro or in vivo as a result of various mechanisms – for example from pressure through a syringe, blood flow through medical devices, chemical agents such as detergents or immunologically mediated transfusion reactions.
For Clinical Biochemists, haemolysis poses problems as it causes interference in a number of assay systems. This interference may be the result of change in the concentration of an analyte due to its escape from blood cells, for example potassium and aspartate transaminase, chemical interference by free haemoglobin or other intracellular contents in the analytical reaction, or by spectrophotometric interference, particularly at 415, 540 and 570 nm where haemoglobin shows strong absorbance. Even mild haemolysis can cause spurious elevation of serum potassium, although measurement of other analytes may still be measured in the presence of mild to moderate haemolysis.
Haemolysed samples are unsuitable for measurement of analytes which are prone to interference, and must be identified promptly to facilitate repeat venesection.
Haemolysis is characteristically visible as a reddish discolouration after separation of serum or plasma and it can be detected by visual inspection. For many years laboratories have relied on analysts inspecting samples for the presence of discolouration in order to detect haemolysis. More recently automated analysers have incorporated the facility to detect haemolysis spectrophotometrically, displaying the result as a haemolytic index (HI), which can be reviewed in order to make a decision about possible assay interference.
With the introduction of the Roche Modular system in our laboratory we changed practice for the detection of haemolysis switching from visual inspection to the use of an automated HI calculated from the differences in absorption at 570–600 nm and 660–700 nm. The HI at which interference was considered sufficient to cause assay interference was determined according to Roche product literature and local development work. 2,3
After making this change our neonatology team reported an increase in the incidence of haemolysed samples. Anecdotal reports from other hospitals suggested that this was a common finding and that other laboratories had raised the HI used to define haemolysis in neonatal samples as a result of similar concerns.
The aim of this study was to investigate whether there was justification for using different HI cut-offs to define haemolysis in samples from adult and neonatal populations.
Methods
All samples were analysed on the Roche Modular System.
Investigation of visual haemolysis in neonatal samples
For the 2-week period in 2007 all 15 qualified and two trainee biomedical scientist staff working on the Roche modular system were asked to record the presence or absence of visual haemolysis in all samples from the neonatal unit as they processed them according to standard operating procedure. Their observations were compared with the measured HI.
Investigation of visual haemolysis in adult samples
Thirteen samples with a range of HI were shown to analysts who were asked to comment on the presence or absence of haemolysis.
The majority of samples from neonates were icteric. In order to investigate the effect of icterus on the visual detection of haemolysis, an aliquot of blood from a sample with an icteric index of 204 μmol/L was subjected to differing degrees of mechanical trauma by passage through a 23-gauge needle in order to produce a range of haemolysed sera. These were shown to the 17 biomedical scientists and to 11 experienced assistant technical officers for their opinion on the presence of haemolysis.
Relationship of haemolytic index and potassium concentration
To observe the relationship between potassium concentration and HI, data were obtained on 613 samples from neonates and 523 from adults. This information was obtained from the laboratory APEX computer system and the analyser process systems manager and used to obtain equations to enable compensation for the increase in potassium per unit change in HI. These equations were validated in a further 60 samples from adults and 60 from neonates to confirm that any relationship between the potassium concentration and the HI was eliminated when the derived adjusted potassium result was used in place of the measured potassium.
Statistical analysis
Linear regression analysis, derivation of confidence limits and t-testing were performed using Analyse-it for Microsoft Excel (version 2.12) supplied by Analyse-it Software, Ltd.
To investigate whether there were significant differences between the linear regression data obtained for adults and neonates, it was assumed that the intercepts of linear regression analysis equations were equivalent for both neonates and adults and a ratio was determined using the formula (y-intercept)/x for each data point (
Results
Investigation of visual haemolysis in neonatal samples
The relationship between visually evident haemolysis and the analytical HI (μmol/L) obtained on the Roche Modular system for neonatal samples is shown in Figure 1.
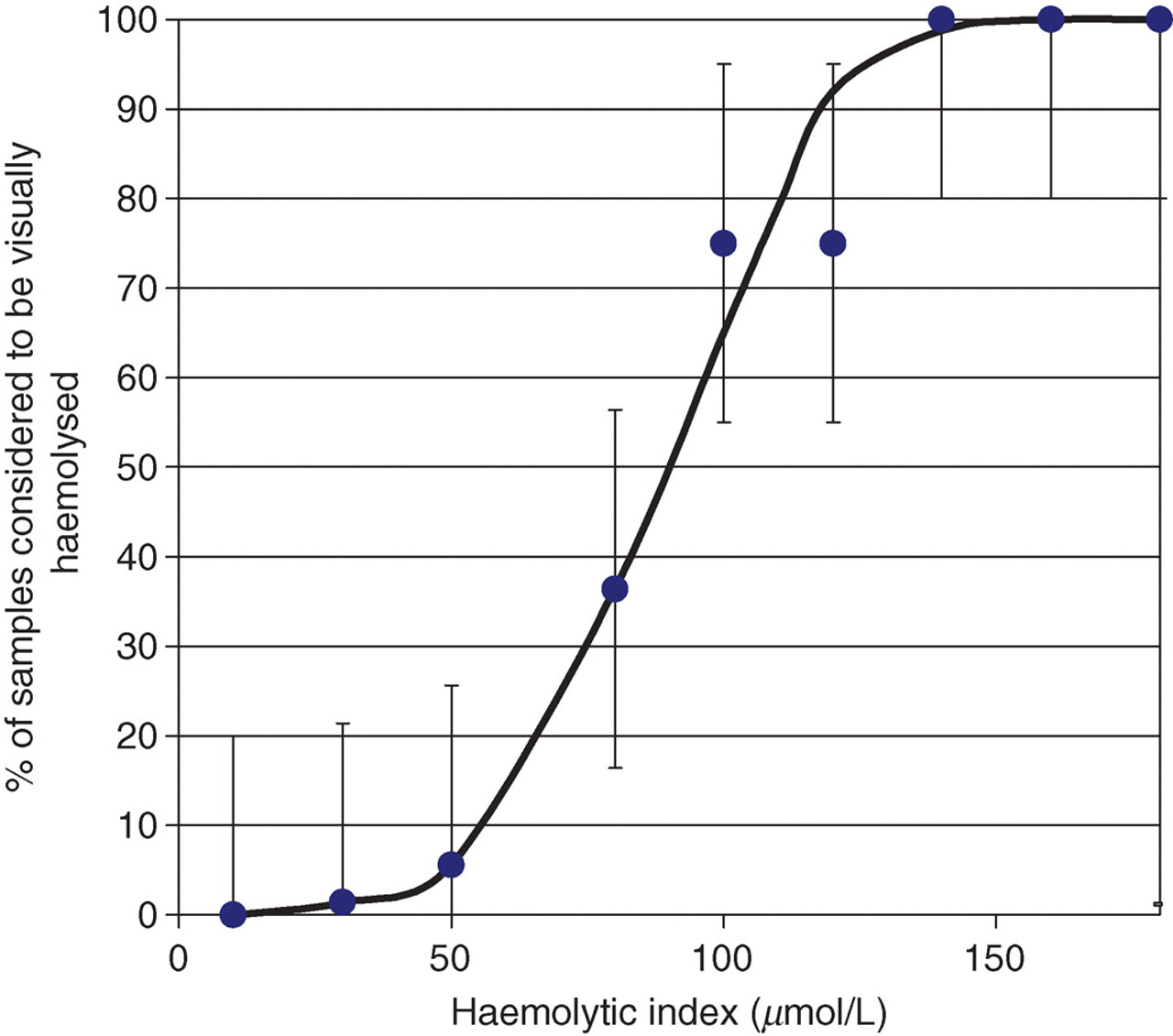
The relationship between visually evident haemolysis and the analytical haemolytic index obtained on the Roche Modular system for neonatal samples
Investigation of visual haemolysis in adult samples
The presence of icterus resulted in underdetection of haemolysis on visual inspection as shown in Figure 2.
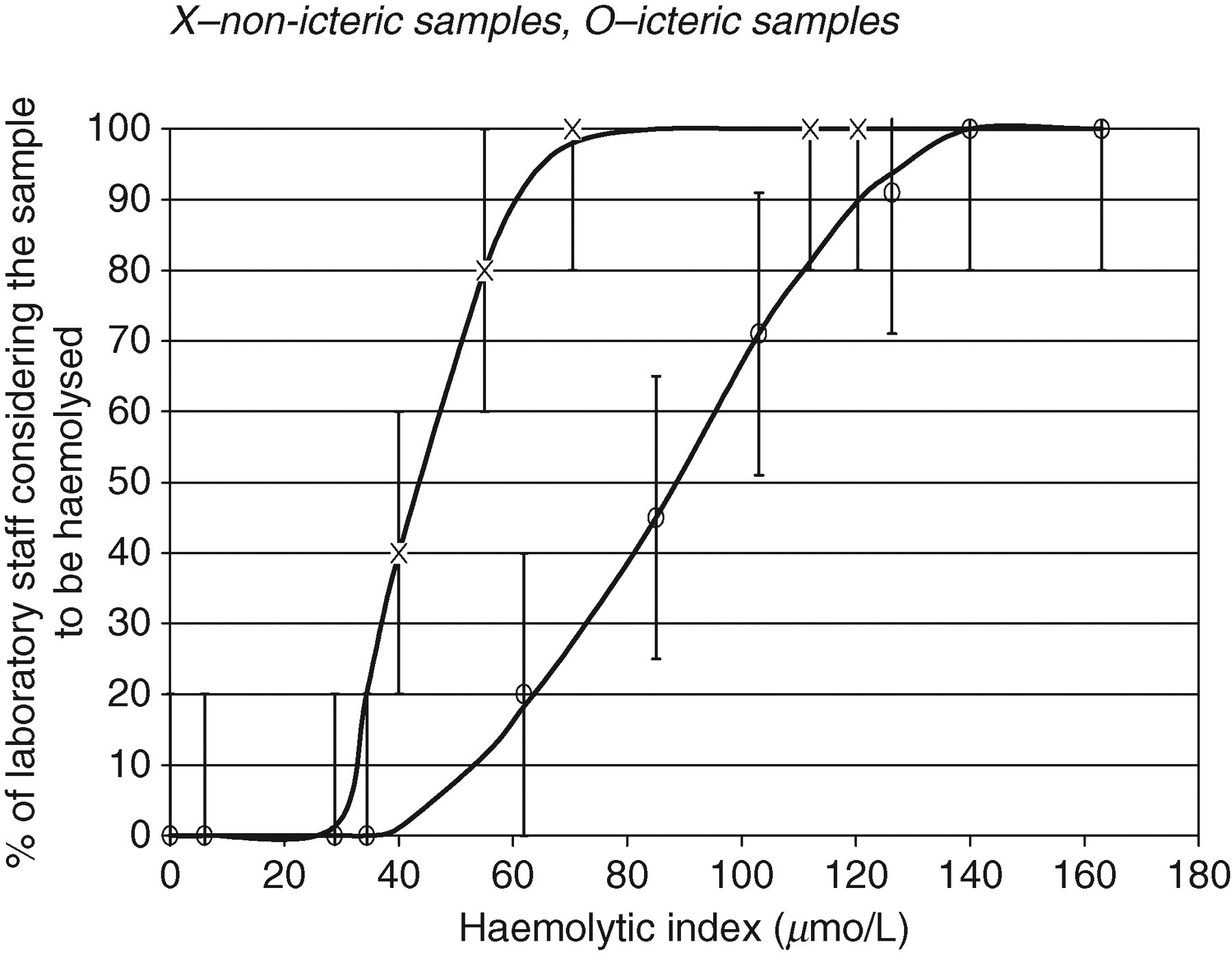
The relationship between visually evident haemolysis and the analytical haemolytic index obtained on the Roche Modular system for icteric and non-icteric adult samples
Relationship between haemolytic index and potassium concentration
The relationship between HI and potassium was compared by plotting the serum potassium concentration against the HI for the 613 adult (Figure 3a) and 523 neonatal samples (Figure 3b). The linear regression equations obtained from this analysis were used to derive formulae to compensate for the increase in potassium caused by haemolysis using the method described by Dimeski et al. 4
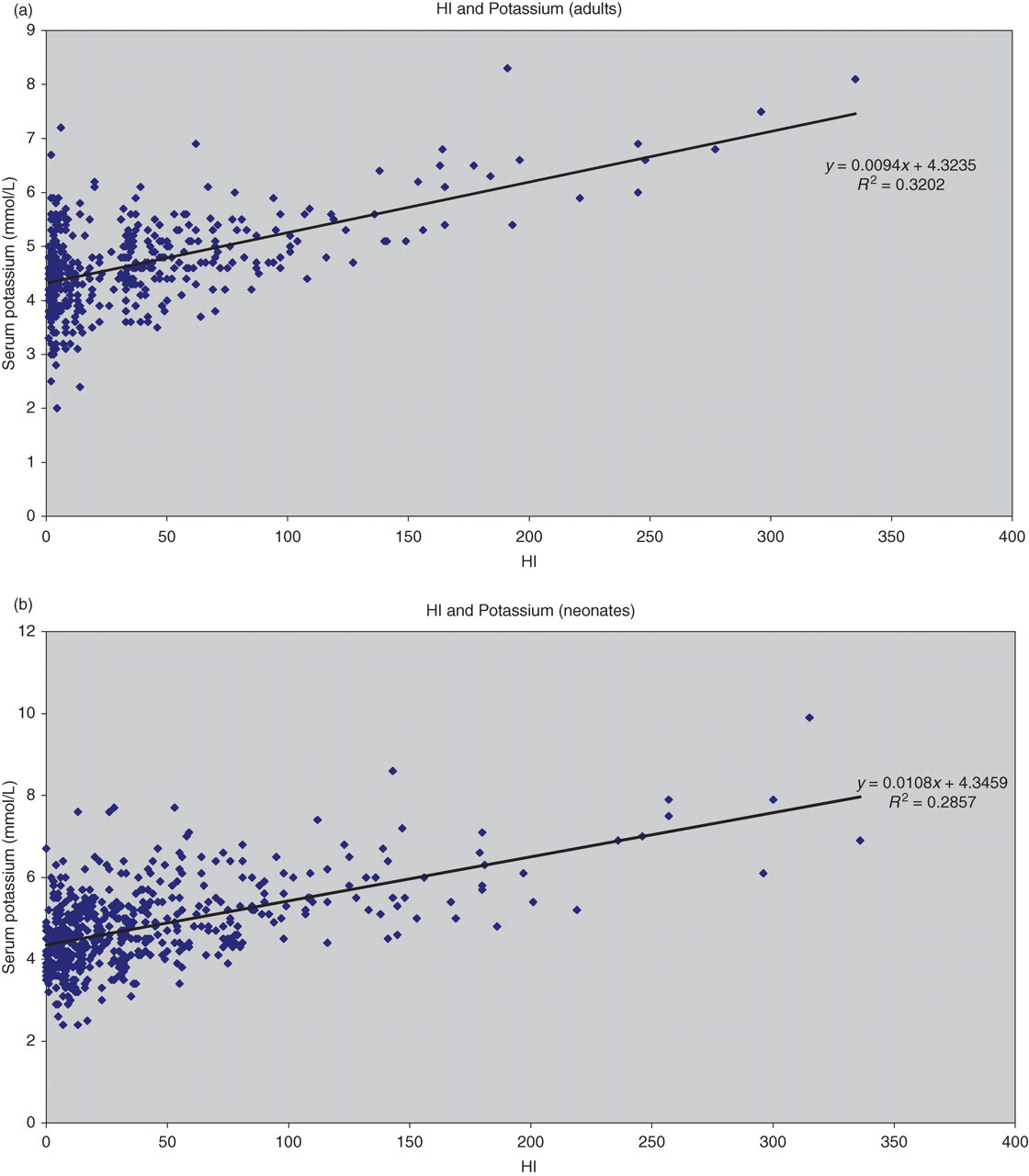
Relationship between serum potassium (mmol/L) and the analytical haemolytic index (µmol/L) obtained on the Roche Modular system for samples from 613 adults (a) and 523 neonates (b)
The mean (95% CI) potassium increase per unit increase in HI was 0.0094 (0.0078–0.0103) for adults and 0.0108 (0.0094–0.0121) for neonates.
The formulae derived for adjusted potassium were as follows:
For adults:
Adjusted K = Measured K − (HI in μmol/L × 0.0094).
For neonates:
Adjusted K = Measured K − (HI in μmol/L × 0.0108).
Using these compensation equations, the relationship between HI and compensated potassium showed no correlation in the 60 subsequent samples received from adults and 60 from neonates (R 2 = 0.0038 adults; R 2 = 0.024 neonates).
On comparing the linear regression data for adults and neonates they were found not to differ significantly (P = 0.3114) and a single equation was derived for use in both populations from the pooled data:
Adjusted K = measured K − (HI in μmol/L × 0.01).
Discussion
Haemolysis is reported to be detectable as a reddish brown discolouration of plasma or serum at haemoglobin concentrations above 0.3 g/L (18.8 μmol/L). 1 and historically haemolysis has been detected by visual inspection of samples. However, visual inspection has been noted to be inaccurate in the detection of haemolysis 5 and the detection limit has been postulated to be higher in icteric sera. 6 A change from visual inspection to use of the HI would therefore be likely to increase the reported incidence of haemolysis, in line with the observation of our neonatologists.
Laboratories vary in the way in which potassium results from samples known to be haemolysed are reported to neonatal units. 3 Options include not analysing the sample, not issuing a result or issuing a result and indicating the presence of haemolysis. 1,4 Equations to correct for the degree of haemolysis have been derived in a simulated environment 4 and using authentic data from adult patients and have been used as the bases of systems, which issue qualitative comments on the corrected potassium result. 7
We have derived a formula with the data from the Roche Modular system from our own laboratory to enable us to compensate for elevations in potassium concentration due to haemolysis in samples from neonates and adults. Issuing the potassium concentration of a haemolysed sample, even with an indication of its nature may be open to misinterpretation and is not recommended. 1 Whilst neonatologists are adept at dealing with patients with concentrations above the quoted adult reference range some are less familiar with the possibility of haemolysis masking hypokalaemia – a rarer biochemical disturbance in this patient population. HI is not subject to rigorous quality control and hence there are concerns about issuing a quantitative potassium using HI as the basis for a mathematical correction factor. 7
We found the potassium increase per unit of HI to be higher than that reported by Dimeski, perhaps due to the difference between derivation from simulated and actual patient data. It is of note that Dimeski quotes units for HI as grams of Hb/L, our Roche system is configured to equate the HI to μmol/L of haemoglobin. Laboratories should ensure that they are familiar with the way their own platform derives the index prior to introducing it into clinical service. While we have validated the equation for use on the Roche Modular system, we would suggest that other users consider its applicability to their own laboratory settings.
In conclusion, haemolysis is a common clinical problem resulting in an inability to interpret potassium measurement. Visual detection of haemolysis tends to underestimate haemolysis in the presence of icterus and use of the automated HI is particularly recommended in the neonatal population. We have derived a correction formula for use in adults and neonates which we plan to use in our laboratory in order to provide a qualitative comment to help prioritize the need for repeat potassium measurement and so aid immediate clinical management.
DECLARATIONS
