Abstract
We have documented a unique instance of diabetic ketoacidosis (DKA)-induced stress-related cardiomyopathy (CMP) in a 46-yr-old Caucasian female with type I diabetes mellitus. In times of stress, tissues with high capacity for aerobic metabolism, like myocardium, can preferentially change their metabolic substrate to ketones. The myocyte has a decreased ability to metabolize glucose and free fatty acids in stress CMP. To the best of our knowledge this is the first report of stress CMP complicating DKA. A possible mechanism for the same is hypothesized.
Introduction
Takotsubo cardiomyopathy (CMP) also called stress-induced CMP/stress CMP was first described in Japan and subsequently worldwide. This syndrome can occur after physical and emotional stress, cerebrovascular accidents, gastrointestinal bleeding, acute asthma and non-ketotic hyperosmolar state (NKHS). Stress CMP has not been previously reported with diabetic ketoacidosis (DKA). We describe a case of stress CMP precipitated by DKA.
Case
A 46-yr-old woman with type I diabetes for 25 yr, had been on an insulin pump for 5 yr. She presented with DKA, after running out of insulin. She had epigastric abdominal pain, nausea and vomiting. Her blood chemistry profile values included: sodium – 132 mmol/L, bicarbonate – 4 mmol/L, anion gap – 31 mmol/L and blood glucose – 674 mg/dL. The EKG showed ST-elevations in both anterior leads (V2–V6) and inferior leads (II, III and aVF). Cardiac enzymes peaked at troponin of 7.75 and CPK of 652. An echocardiogram demonstrated a left anterolateral wall hypokinesia and severe apical akinesia and ballooning (Figure 1A and B). The ejection fraction was 30%. Normal coronary arteries were seen on cardiac catheterization (Figure 1E and F). DKA was successfully treated and acidosis rapidly corrected. A follow-up echocardiogram in 12 d revealed a complete return of normal LV function and a correction of the regional wall motion abnormalities (Figure 1C and D).
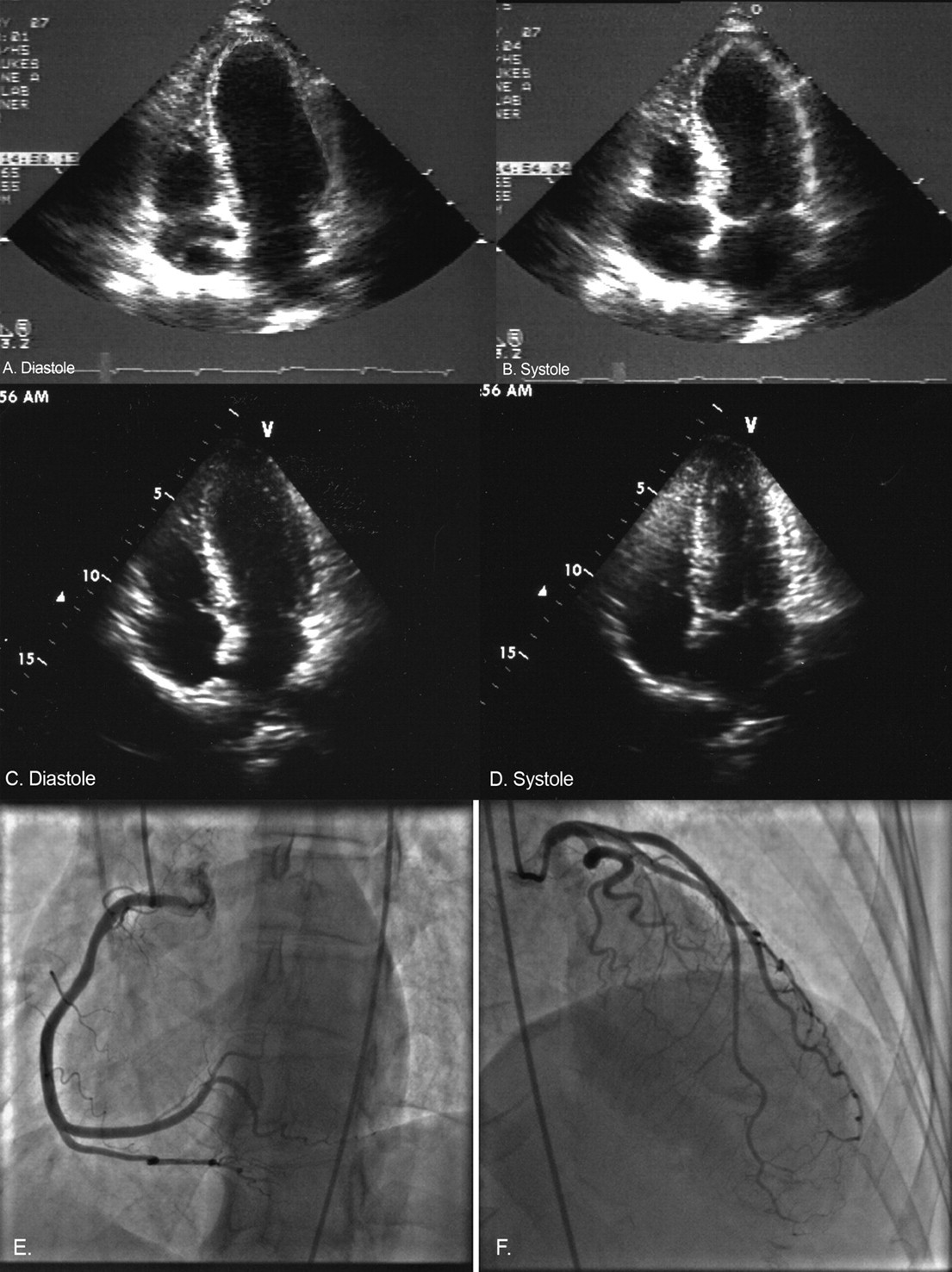
Apical four-chamber view on trans-thoracic echocardiography reveal characteristic apical ballooning of stress cardiomyopathy (A, B) and complete recovery at repeat ECHO subsequently (C, D). Cardiac catheterization reveals normal coronaries without any significant obstruction (E, F)
Discussion
This patient's clinical presentation, EKG changes, echocardiographic abnormalities, angiographic findings and subsequent complete resolution of LV apical ballooning met the diagnostic criteria for stress CMP. Hypoglycaemia and NKHS are among the reported known causes. 1 However, to date, DKA has not been a reported association or cause of stress CMP.
Myocardial glucose utilization and fatty acid metabolism are both impaired in stress CMP. 2,3 Ketones become the preferred metabolic substrate of the myocardium. In DKA, during stress there is always an abundance of ketones. This readily available energy substrate for the myocardium may account for the rarity of stress CMP in such patients. During physical activity, free fatty acids (FFA) and ketones are preferably utilized by muscles. This spares glucose for other vital functions. Acetoacetate is preferred over FFA by the myocardium. The hierarchy of cardiac energy usage occurs in the following order during stress – ketones, short chain fatty acid, other FFAs and glucose. At any point, all of these energy sources will be utilized in the stated order of preference. 4
High concentrations of ketones inhibit glucose utilization in tissues by numerous pathways. The oxidation of FFA and ketones produce acetyl CoA, which enters the citric acid cycle to cause an increase in intracellular concentration of citrate. Citrate acts as a negative allosteric modulator for the enzyme phosphofructokinase. The activity of phosphofructokinase is central in regulating the rate of glycolysis. This inhibits further metabolism of glucose along the glycolytic pathway. Elevated fructose phosphate and its isomer glucose phosphate (which accumulates in the cytosol) now inhibit the hexokinase-mediated conversion of glucose to glucose phosphate. The accumulated intracellular glucose impedes the further entry of glucose inside the cell (Figure 2). High concentrations of acetyl CoA obtained from ketone and FFA metabolism also prevent oxidative decarboxylation of pyruvate by inhibiting pyruvate dehydrogenase.
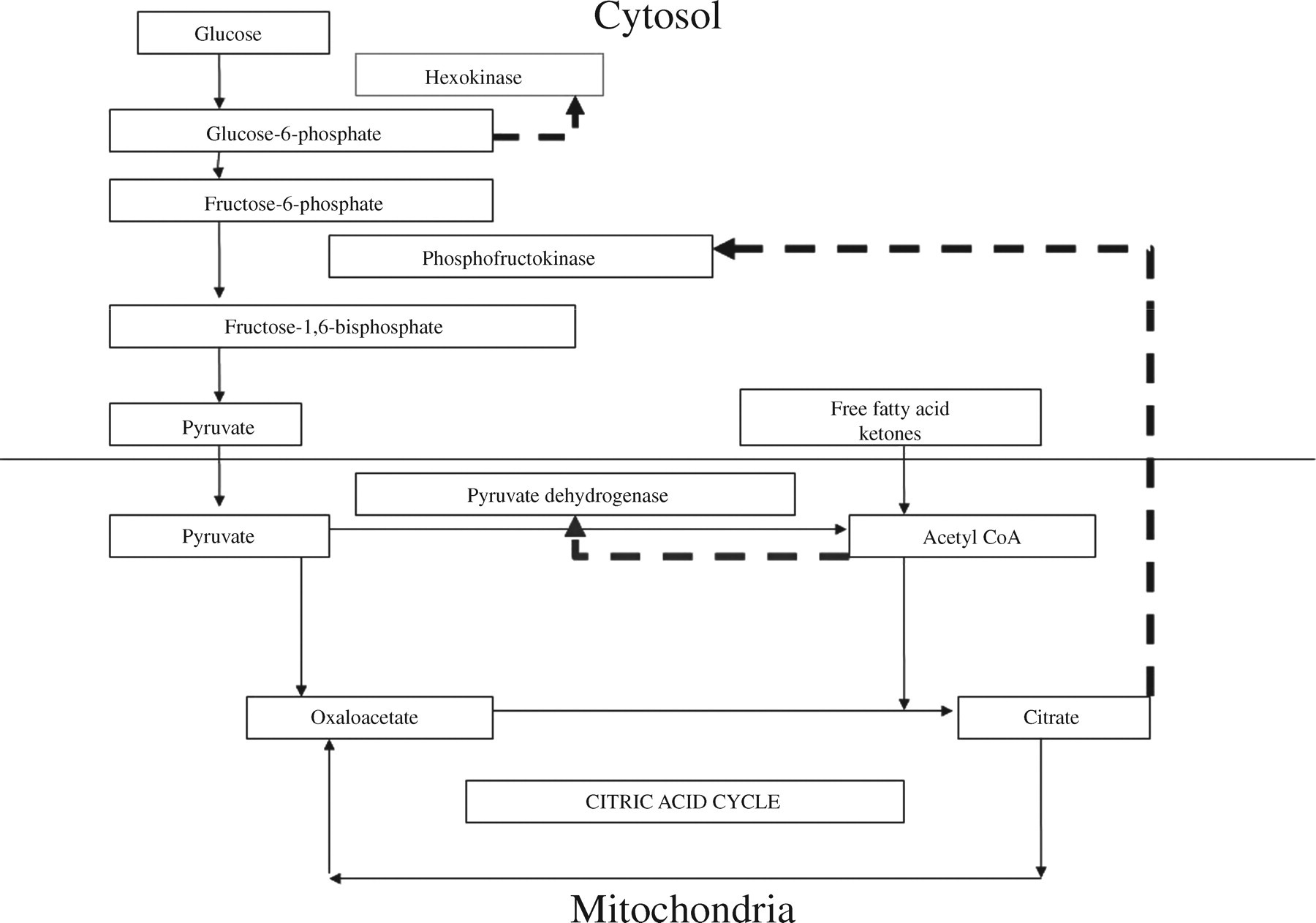
Metabolic pathway in a cardiac myocyte during diabetic ketoacidosis. High concentrations of ketones produced by the liver are metabolized by extrahepatic tissues. In the mitochondria they produce acetyl CoA. Acetyl CoA is utilized by the citric acid cycle producing citrate. Citrate diffuses out to the cytosol inhibiting phosphofructokinase and the glycolytic pathway of glucose utilization. Glucose phosphate that accumulates upstream inhibits hexokinase leading to cellular overload with glucose. This prevents entry of glucose into the cell. The broken lines (- - -) indicate inhibition of specific enzymes by metabolites
In DKA, the high rate of fatty acid oxidation in the liver produces ketones like acetoacetate and hydroxybutyrate. Hydroxybutyrate is mostly converted to acetoacetate. Acetoacetate is converted to acetoacetyl CoA, which is then cleaved by enzyme thiolase to acetyl CoA. Acetyl CoA is metabolized by the citric acid cycle. 5 Acetoacetate also spontaneously decarboxylates to acetone. Ketone bodies that are produced are primarily metabolized in extrahepatic tissue and utilized in preference to glucose and FFA in tissues like the heart. As the plasma concentrations of ketones increase, so does their utilization by several extrahepatic tissues like skeletal muscles and myocardium. The oxidative pathways in these tissues become overwhelmed when plasma ketone concentrations exceed 12 mmol/L. At this point ketones begin to accumulate and ketoacidosis ensues.
Our patient developed stress CMP in spite of abundant concentrations of ketones for metabolism. We propose that severe acidosis is a mechanism that precipitates acute CMP. The severe acidosis was evidenced by a plasma bicarbonate of 4 mmol/L and a anion gap of 31 mmol/L. The patient was re-admitted 4 weeks later with DKA, but without stress CMP, and her plasma bicarbonate was 15 mmol/L. The hypothesis that severe acidosis induces cardiac dysfunction is supported by these findings.
Cardiac contraction involves a series of well-synchronized steps. With depolarization of the cell membrane, L-type Ca2+ channels are opened. There is a small increase in intracellular calcium concentration. This causes the ryanodine receptors (RyRs) of sarcoplasmic reticulum to open, resulting in a significant increase of the cytosolic calcium from 10−7 mol/L to 10−5 mol/L. Calcium binds to troponin C (TpC) forming TpC.4Ca2+, which interacts with troponin I and troponin T. These three, in concert with tropomyosin lead to actin-myosin interaction, resulting in cardiac contraction. When the sarcoplasmic calcium concentration falls below 10−7 mol/L cardiac muscle undergoes relaxation. The decrease in cytosolic calcium results from a time-dependent closure of RyRs, resequestration of calcium by the sarcoplasmic reticulum calcium pump (SERCA) and calcium efflux from the cell.
The reduction of cardiac myocytic inotropy is the result of acidotic inhibition of most steps of the excitation-contraction coupling. A fall in pH results in a decreased calcium release from the sarcoplasmic reticulum, reduced sensitivity of TpC to calcium, displacement of calcium from TpC-binding sites and inhibition of SERCA. 6
Cardiomyocytic damage is also attributed to acidosis-induced apoptosis. There is a direct correlation of low pH and induction of apoptosis in patients undergoing cardiac surgery. 7 Diabetic patients have a higher predisposition to cardiomyocytic apoptosis than non-diabetics. 8 In non-ischaemic myocardial acidosis, LV ejection fraction is impaired with a drop in pH from 7.2 to 6.2. There is a synchronous increase in caspase-3 activity. 9
The tissue kallikrein system is a natural defence against apoptotic injury. All components of the kallikrein system are present in the heart. 10 Tissue kallikrein cleaves kininogen to produce kinin. Kinin binds to kinin-B2 receptor. The latter activates various second messengers like cyclic adenosine monophosphate (cAMP), cyclic guanosine monophosphate (cGMP), nitric oxide (NO) and prostacyclin. These second messengers influence various protective pathways in the diabetic myocardium. 11 Akt signalling is crucial to cardiomyocyte survival. Akt phosphorylates Bad, a member of the Bcl family. Bad promotes apoptosis through its interaction with Bcl-xL. Phosphorylation of Bad prevents this interaction. Instead, Bad binds to 14-3-3 protein. Akt-Bad-14-3-3 pathway is anti-apoptotic. Another effect of Akt activation is the phosphorylation of glycogen synthase kinase-3 (GSK-3), the inactive form of GSK. The resulting Akt-GSK-caspase-3 keeps caspase-3 inactivated. 12 The kallikrein system protects the heart from acidosis-induced cardiac injury. We hypothesize that in our case cardiac muscle damage, extensive enough to cause persistent heart failure, was prevented by the rapid correction of acidosis.
The rarity of stress CMP in DKA is due to the protective effect of abundant ketones. To the best of our knowledge this is the first case of stress CMP precipitated by DKA as a result of the concomitantly severe acidosis.
Footnotes
DECLARATIONS
