Abstract
Background
A single detectable cardiac troponin predicts mortality in patients treated with dialysis. There are limited data on changes in troponin concentration over time and the clinical implications of serial troponin measurement.
Methods
Serial cardiac troponin T (cTnT) was assayed five times over 12 months in a prospective cohort study of patients with end-stage kidney disease treated with haemodialysis. A concentration of cTnT ≥0.04 μg/L was considered increased. Mortality and cardiovascular events were analysed by survival analysis, according to the serial troponin results.
Results
From 100 patients who provided a baseline sample for cTnT, 81 completed five serial measurements. The analysis of patients who completed serial cTnT measurements demonstrated that 28 patients (35%) had normal cTnT concentrations in all five samples, 20 patients (24%) had between one and four increased cTnT measurements and 33 patients (41%) had increased concentrations of cTnT in all five samples. The 1.7-y patient survival was 100%, 90% and 78% for patients with zero, one to four, or five out of five concentrations of cTnT increased, respectively (P = 0.037), and the corresponding cardiovascular event-free survival was 100%, 91% and 78%, respectively (P = 0.027).
Conclusions
Serial measurements of cTnT concentration were frequently increased in patients receiving haemodialysis. The number of abnormal measurements over time predicted mortality and cardiovascular adverse events.
Introduction
Patients with end-stage kidney disease treated with dialysis have a high mortality from cardiovascular disease (CVD). 1 This mortality is not easily predicted by conventional cardiovascular risk factors and a number of biomarkers for cardiovascular events have been previously suggested. 2 Cardiac troponin I (cTnI) and cardiac troponin T (cTnT) are components of the myocardial contractile apparatus that are released into the circulation in response to myocardial necrosis. Assays on automated platforms are widely available for both cTnI and cTnT. A single detectable cardiac troponin in these patients predicts future cardiovascular events and mortality, despite the absence of symptoms of cardiac disease. 3 Furthermore, increased troponin concentration occurs commonly in end-stage kidney disease, 4 but few studies report serial measurement over time. Changes in troponin concentration over time are likely to be physiologically and clinically important. Initial studies by our group 5 and others 6–9 have examined changes in troponin over time. We have previously demonstrated that 25% of patients with undetectable cTnI at baseline subsequently develop an increased cTnI concentration over a seven-week period. 5 One study has demonstrated an increase in cTnT in 24% of patients after 15 months, 6 and others have identified a substantial proportion of patients on dialysis with persistently increased cTnI 7 and cTnT 8,9 concentrations over time.
The hypothesis underlying this study is that persistently increased troponin concentration in patients on dialysis, who report no symptoms of CVD, might be caused by persistent subclinical myocardial necrosis that is sufficient to cause the release of troponin but has not caused a clinical cardiac event. This may reflect patients at increased risk of future cardiac events or death. The aim of the present study was to determine the prevalence of patients with persistently increased troponin concentration over time, and whether such patients subsequently experience adverse clinical outcomes compared with patients with persistently normal cardiac troponin concentration. This is thus a study of the clinical outcome of those patients who complete serial troponin measurements over an initial 12-month assessment period.
Subjects and methods
Study population
Patients on haemodialysis, who reported no symptoms of CVD, were recruited from dialysis units associated with a single centre (Austin Health) between August 2003 and August 2004. Patients were excluded if they had commenced dialysis (or received a transplant) in the preceding six months, had a cardiovascular event within the previous three months or were expected to survive less than six months. The Austin Health Human Research Ethics Committee approved the study.
Patients undergoing haemodialysis who met the above inclusion criteria were given an invitation to participate at their haemodialysis unit. A total of 204 patients undergoing haemodialysis in the Department of Nephrology at Austin Health were considered for inclusion in the study. Consent was obtained from 103 patients, but two withdrew consent and one received a transplant before the baseline blood could be collected. Seventy-one patients declined to participate, three were not approached and 27 met exclusion criteria (17 were not able to give informed consent, 4 had an active current illness, 2 had a recent cardiovascular event and 4 for other reasons).
Study procedures
Background clinical and anthropometric data were obtained at the time of giving informed consent. Patients provided blood at baseline and subsequently provided four further samples over the next 12 months. All blood was taken before the middle dialysis session of the week. The results of baseline troponin concentrations have been reported previously. 10 Those who died, experienced a cardiovascular event, withdrew from the study or underwent renal transplantation during the 12 months of blood collection were excluded from subsequent analysis of serial measurements. Only patients with five complete serial troponin measurements over 12 months were included in this final clinical outcome analysis.
Laboratory procedures
cTnT was measured by electro-chemiluminescence immunoassay using an Elecsys E170 analyser (Roche Diagnostics, Indianapolis, IN, USA). A concentration of 0.04 μg/L was the cut-off used to define an increased cTnT as it represents the lowest concentration corresponding to a 10% coefficient of variation (CV) for this assay. 11 While cTnT concentrations below 0.04 μg/L and above the 99th percentile may also have prognostic implications, the 10% CV cut-off was chosen for this study because this is what is recommended for clinical practice to avoid false-positive results. 12
Outcomes
Patient outcomes were followed for up to 2.8 y. The primary outcome measure was all-cause mortality. The secondary endpoint was a combined outcome of cardiac death, myocardial infarction, coronary revascularization procedure, stroke, peripheral revascularization and gut ischaemia. The cardiovascular events were adjudicated by an independent clinical event committee.
Statistical analysis
Baseline characteristics were compared using analysis of variance for parametric data, presented as mean and standard deviation, or the Kruskal–Wallis test for non-parametric data, presented as median and inter-quartile range. Proportions were analysed by chi-square, with Fisher's exact test where appropriate. Kaplan–Meier survival curves were constructed to compare patients with none of the five measurements of cTnT increased, to patients with one to four out of five measurements of cTnT increased, and patients with increased cTnT concentrations on all five measurements. The date of the fifth blood sample was the beginning of the analysis time. In addition, patients with the baseline cTnT concentration above the cut-off as a single measure were compared with patients with a normal cTnT concentration at baseline analysed from the date of this baseline sample, and patients with two consecutive cTnT measurements above the cut-off were compared with patients with zero or one of two consecutive cTnT measurements above the cut-off analysed from the date of the second sample. Patients were censored at the first clinical endpoint event, receipt of a kidney transplant or on 30 June 2006. Results are expressed as the survivor function (%) with 95% confidence interval in brackets. Comparison between the different troponin groups was assessed by the log-rank test.
Statistical analysis was performed using Stata version 8.0 (Statacorp, College Station, TX, USA).
Results
Baseline characteristics
Of the 100 patients receiving haemodialysis who provided a baseline blood sample, the major causes of end-stage kidney disease were glomerulonephritis (40), diabetes (14), renovascular disease (10), reflux nephropathy (9), and other causes or unknown (27). The type of vascular access in use was native radiocephalic fistula (58), native brachiocephalic fistula (24), upper limb prosthetic fistula (12), and either a catheter or other fistula (6). Patients underwent thrice weekly dialysis for 12 h (50), 13.5 h (23) or 15 h (16), with the remaining 11 having either a different number of sessions or different weekly hours. Most patients utilized either a polyamide membrane (69) or cellulose triacetate (23), and 34 patients used a hi-flux membrane.
Of the 100 patients who provided blood at baseline, 19 did not provide five serial samples for the following reasons: nine died, three received a transplant, three moved away and one withdrew consent before a fifth sample was collected. In addition, two patients had insufficient serum in one sample for cTnT analysis and one did not provide the fifth sample. Thus there were 81 patients, on whom the full 12-month initial data were available, in the final study cohort.
Serial troponin concentrations
The number of measurements per patient with increased cTnT concentration demonstrated a bimodal distribution, with 28 patients (35%) having zero out of five measurements ≥0.04 μg/L, 20 patients (24%) having one to four of the five measurements of cTnT ≥0.04 µg/L and 33 patients (41%) having all five measurements ≥0.04 μg/L. The patients with one to four of the five measurements of cTnT ≥0.04 μg/L consisted of seven, six, five and two patients with one, two, three and four cTnT measurements ≥0.04 μg/L, respectively. Patients with no increased cTnT concentrations were significantly younger than those with one to four measurements of cTnT ≥0.04 μg/L and had a significantly higher serum albumin than this group at the time of the first sample. Cardiovascular medication use did not differ between groups (Table 1).
Characteristics, at the time of the first sample, of patients on haemodialysis who underwent five serial measurements of cTnT
Patients are divided according to whether zero (no cTnT+), some (1–4 cTnT+) or all five (5 cTnT+) measurements of cTnT were ≥0.04 μg/L. Data are expressed as mean ± standard deviation, number (%) or median (interquartile range). The P value is for the test across the three groups
CVD = cardiovascular disease; Hb = haemoglobin; cTnT = cardiac troponin T; SBP = systolic blood pressure; DBP = diastolic blood pressure; PP = pulse pressure
*P < 0.05 versus patients with ‘No cTnT+’; Antiplatelet agents include aspirin, clopidogrel, dipyridamole or ticlopidine; RAS (Renin Angiotensin System) blockade includes both angiotensin converting enzyme inhibitors and angiotensin receptor blockers; ESA = erythropoiesis stimulating agent (either erythropoietin or darbepoietin)
Clinical outcomes
All-cause mortality
After the 12 month period collecting the initial troponin data, the 81 study patients were followed for up to 1.8 y. There were no deaths in patients with none of the five measurements of cTnT ≥0.04 μg/L, two deaths in patients with one to four measurements of cTnT ≥0.04 μg/L and seven deaths in patients with all five measurements of cTnT ≥0.04 μg/L. The survival of these patients at 1.7 y was 100%, 90% (95% confidence interval: 66–97) and 78% (60–89), respectively (P = 0.037, Figure 1). The adjudicated causes of death in the seven patients with persistently increased cTnT concentrations who died were definite CVD (3), possible CVD (2) and inconclusive (2).
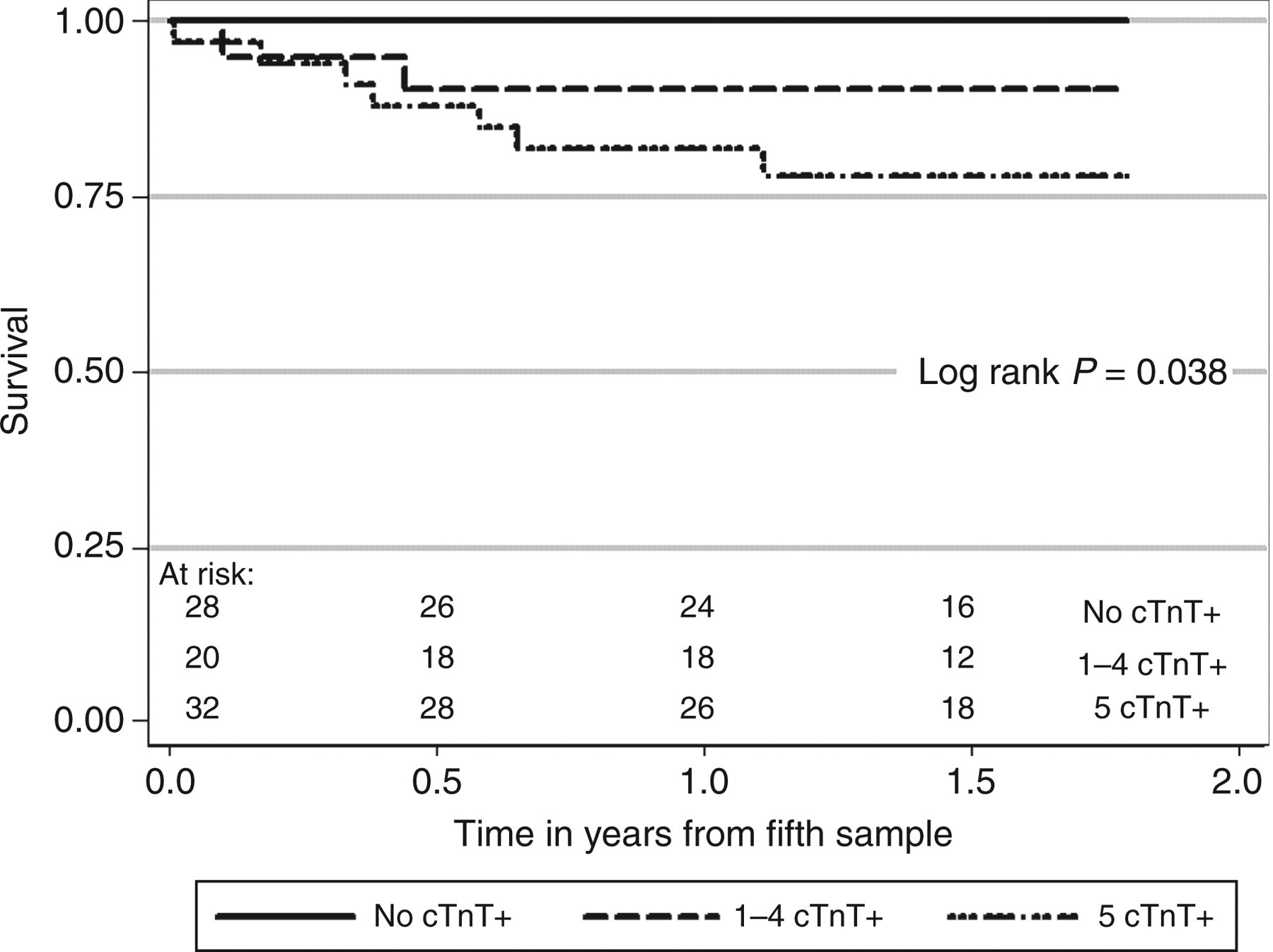
The survival of patients receiving dialysis who had none of the 5 measurements of cTnT ≥ 0.04 μg/L (No cTnT+) compared to 1–4 measurements of cTnT ≥ 0.04 μg/L (1–4 cTnT+), compared to all five measurements of cTnT ≥ 0.04 μg/L (5 cTnT+). For pairwise comparisons: Log rank P = 0.013 for No cTnT+ versus 5 cTnT+, P = 0.096 for No cTnT+ versus 1–4 cTnT+, and P = 0.298 for 1–4 cTnT+ versus 5 cTnT+
The survival at 2.5 y of patients with a single baseline cTnT ≥0.04 μg/L was 73% (58–84) compared with 88% (76–95) if the baseline cTnT was below this value (log rank P = 0.109). However, patients with two consecutive measurements of cTnT ≥0.04 μg/L demonstrated a 2.5 y survival of 71% (56–81) from the date of the second sample compared with 95% (82–99) in patients who did not have two consecutive measurements of cTnT ≥0.04 μg/L (log rank P = 0.005).
Combined cardiovascular events
Seventy-two patients provided five samples without experiencing an event before the fifth sample was collected, and were followed for up to 1.8 y. Cardiovascular events occurred in zero of 26 patients with no cTnT concentrations ≥0.04 μg/L, one of 17 patients with one to four cTnT concentrations ≥0.04 μg/L and six of 29 patients with all five cTnT concentrations ≥0.04 μg/L. The event-free 1.7 y survival of these three groups was 100%, 91% (51–99) and 78% (57–90), respectively (log rank P = 0.027, Figure 2). The adjudicated events in the patients with persistent cTnT elevations were: myocardial infarction (2), cardiac death (2), coronary artery bypass graft surgery (1) and femoro-popliteal bypass surgery (1).
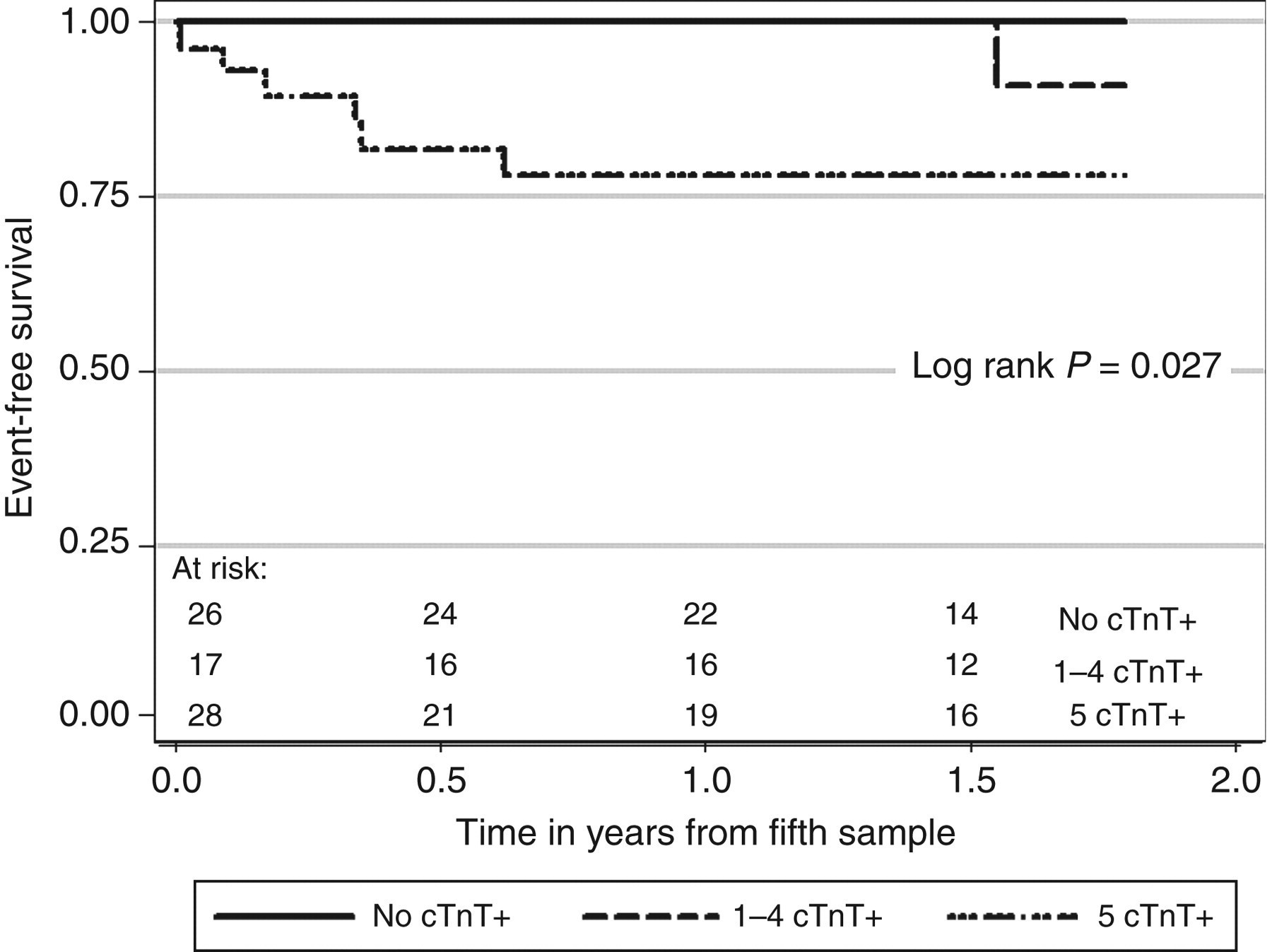
The cardiovascular event-free survival of patients receiving dialysis who had none of the 5 measurements of cTnT ≥ 0.04 μg/L (No cTnT+) compared to 1–4 measurements of cTnT ≥ 0.04 μg/L (1–4 cTnT+), compared to all five measurements of cTnT ≥ 0.04 μg/L (5 cTnT+). For pairwise comparisons: Log rank P = 0.014 for No cTnT+ versus 5 cTnT+, P = 0.259 for No cTnT+ versus 1–4 cTnT+, and P = 0.165 for 1–4 cTnT+ versus 5 cTnT+
Event-free survival at 2.5 y was 69% (54–81) if the single baseline cTnT ≥0.04 μg/L compared with 86% (74–93) if it was not (log rank P = 0.120), and was 74% (59–84) if two consecutive values of cTnT were ≥0.04 μg/L compared with 88% (70–96) if no two consecutive measurements of cTnT were ≥0.04 μg/L (log rank P = 0.049).
Discussion
This study of serial measurements of cTnT demonstrates that a significant population of patients on dialysis, who report no symptoms of CVD, have persistently increased concentrations of cTnT over the course of a year. Thereafter, they have more cardiac events and greater mortality than patients without persistently increased cTnT concentrations. Importantly, patients with all serial concentrations of cTnT <0.04 μg/L over a period of 12 months had an excellent clinical outcome, with no events over the subsequent follow-up period.
The high proportion of patients on dialysis with persistently increased cTnT concentration is consistent with previous studies. Using a similar cTnT assay to that used in our study, 35% of patients on dialysis without baseline CVD had all of four measures of cTnT above the upper reference limit of 0.04 μg/L, 8 and other investigators have demonstrated that 19 of 58 patients (33%) had all of three measurements of cTnT above 0.04 μg/L. 9 In this study, patients who had every cTnT measurement above 0.04 μg/L did not have an increased risk for developing ischaemic heart disease, as distinct from patients who demonstrated an increase in cTnT over time.
Our study provides new information on the subsequent patient follow-up in those individuals with persistently increased concentrations of cTnT, and we suggest that cardiac pathology, and not altered renal elimination alone, is the most likely explanation for these persistently increased concentrations of cTnT. Although studies have demonstrated an independent association of troponin concentration with glomerular filtration rate in patients not on dialysis, 10,13 the degree of renal elimination remains a controversial issue. One group has demonstrated immunoreactive fragments of cTnT, small enough to be excreted by the kidney, that may explain the increased cTnT in patients on dialysis 14 but others have not confirmed this finding. 15
The poorer outcomes of patients with persistently increased concentrations of cTnT, when compared with the favourable outcomes of patients with no increased cTnT on serial measurement, suggest an underlying cardiac abnormality. The heterogeneous nature of CVD in end-stage kidney disease was demonstrated in a study of 126 prospective kidney transplant candidates. 16 In this study, a cTnT concentration ≥0.04 μg/L was associated with more severe coronary artery disease on angiography, elevated left ventricular volumes and left ventricular mass index, reduced left ventricular ejection fraction and raised left ventricular filling pressures. No single process can necessarily be implicated in the poorer outcome. Since the majority of ruptured plaques in coronary arteries do not result in clinical events, the chronically increased cTnT concentrations may be a marker of repeated subclinical plaque rupture in diffusely atherosclerotic vessels. The presence of myocardial scar using late gadolinium enhancement on cardiac magnetic resonance imaging was evident in four of 13 patients on haemodialysis with cTnT ≥0.07 μg/L and absent from 13 patients with cTnT ≤0.03 μg/L. 17 Increased cTnT concentration may also be a marker of myocardial stress, such as myocyte hypoxia caused by the perfusion mismatch occurring in a hypertrophied ventricle that is prone to myocardial necrosis and interstitial fibrosis. 18 Because the half-life of cTnT is short, 19 persistently increased concentration of cTnT suggests chronic ongoing myocardial injury rather than discrete plaque rupture and consequent acute thrombosis, and such patients could be more likely to succumb when other physiological stresses occur.
The implications of our study for current clinical practice are that serial cTnT measurements may be used to define patients with an excellent outcome compared with patients with persistently increased concentrations, who should be considered at high risk. The appropriate diagnostic and therapeutic intervention in these patients with persistently increased cTnT concentrations compared with those without increased concentrations requires future studies. Considering two consecutive increased concentrations is a more practical clinical approach than waiting for five measurements over 12 months before initiating clinical action. Having two measurements of cTnT ≥0.04 μg/L strengthens the association of cTnT with mortality, and thus serial measurement of cTnT may be useful for surveillance of an individual's risk over time, with reference to previous values. A limitation of this study is that the number of events was small and did not allow for further analysis to demonstrate whether these associations were independent of other factors. The major research question that remains to be answered, with respect to cardiac troponins and patients on dialysis, is how best to use them to improve clinical outcomes and thus complete ‘the evolution of a clinical test’. 20
In conclusion, persistently increased concentrations of cTnT in patients receiving dialysis are associated with increased cardiovascular events and mortality, whereas patients with no increased concentrations have excellent outcomes. Defining risk according to increased cTnT concentration is the first step in developing management algorithms that may guide the intensity of cardiovascular investigation and treatment according to individual risk in this population.
Declarations
This study was supported by unrestricted grants from Amgen, Bristol-Myers Squibb, Janssen-Cilag and Servier. Abbott Diagnostics, Roche Diagnostics Australia, and Medtec Products Australia provided assay kits at various levels of discount for the biochemical assays.
