Abstract
Background
Elevated cardiac troponin (cTn) concentrations predict an increased mortality in patients suffering from end-stage renal disease (ESRD). This study compares the performance of a precommercial high-sensitive cTnT assay (hs-cTnT) with two contemporary cTn assays in detecting cTn elevations in ESRD patients during a six-month follow-up.
Methods
Thirty-two ESRD patients were followed for six months, during which cTn concentrations were assessed every two months. Baseline biomarker concentrations were compared with those in a simultaneously measured reference population of 501 healthy subjects.
Results
During follow-up 26 (81%), 32 (100%) and 9 (28%) of the patients showed elevated cTn concentrations according to the current cTnT, the hs-cTnT and the cTnI assays, respectively. The range of concentrations measured in each patient had a median (interquartile range) magnitude of 0.03 μg/L (0.02–0.06), 0.017 μg/L (0.011–0.029) and 0.011 μg/L (0–0.017) according to the aforementioned assays.
Conclusion
According to the hs-cTnT assay, all of the ESRD patients had elevated cTnT concentrations at least once during the follow-up. As elevated cTn concentrations are highly prognostic of adverse events, the use of serial measurements has thus identified additional patients at risk for such events. The fact that we find cTn concentrations to be higher in patients with a history of cardiac disease is in line with this. Additional studies in ESRD patients are needed to investigate the added diagnostic and prognostic value of the very low cTnT concentrations and variations detected only by the hs-cTnT assay.
Introduction
Patients suffering from end-stage renal disease (ESRD) have a markedly reduced lifespan. Overall, the life expectancy of patients undergoing dialysis is about four times as low as for the general population. 1 The main cause of death in these patients is due to cardiovascular events and over 55% of patients on dialysis suffer from congestive heart failure. 1,2 Considering this high incidence of cardiovascular complications, there is a need for accurate and sensitive biomarkers that can be used for diagnosis and risk stratification in ESRD patients. Over the years, the cardiac troponins (cTns) have proven to be accurate and sensitive markers for assessing ischaemic cardiac damage and predicting cardiovascular death in ESRD patients. 3,4 Nonetheless, cTn concentrations can be elevated in the absence of apparent cardiac damage or clinical symptoms (as reviewed by Kanderian and Francis 5 and Lamb et al. 6 ) and might be influenced by a decreased renal clearance in patients with severe renal disease. 7,8 As the diagnosis of acute myocardial infarction (AMI) has been defined as an increase and/or a decrease of cardiac markers – preferably cTnT or cTnI – detected with at least one value above the 99th percentile limit of the reference population and should be associated with clinical, electro-cardiographic or imaging findings, 9,10 such cTn elevations can impede the diagnosis of an AMI (e.g. when ESRD patients are presenting with clinical symptoms). Recent National Academy of Clinical Biochemistry (NACB) guidelines have addressed this issue and suggest that for patients with chronically elevated concentrations of cTn, changes in cTn (>20%) 6–9 h after the onset of clinical symptoms are indicative of an AMI. 11 However, such changes in cTn concentrations might also occur in the absence of clinical symptoms. 12,13 To date, little is known about the biological variation in cTn concentrations, although some studies have performed serial measurements to investigate the cTn serum concentrations over longer periods of time. 12–16
The advent of new, more sensitive cTn assays could improve the usefulness of both single and serial cTn measurements. To date, most commercial cTn assays lack a sufficient analytical performance to accurately detect cTn concentrations in healthy subjects, either because the limit of detection (LOD) of the assay is higher than the reference concentrations or because the assay imprecision, expressed as the coefficient of variation (CV) is higher than 10% at the 99th percentile of the healthy subjects. 17–19 Assays with sufficient sensitivity to determine the reference concentrations will improve the detection of ‘abnormal’ cTn concentrations in ESRD and other patient populations. More precise assays will also be able to detect temporal changes in cTn concentrations, which could not be detected to date. Such increasingly confident measurements will not only identify patients with abnormal cTn concentrations and variations, but could also shed light on the discrepancy between cardiac troponin T (cTnT) and cardiac troponin I (cTnI). Until recently, the number of ESRD patients showing increased cTnT concentrations was about three times as high as for cTnI (53% versus 17%). 5 With the advent of new, more sensitive cTnI assays, the difference has decreased, but the number of patients with cTn concentrations above the 10% CV limit of the assay is still higher for cTnT (59%) than for cTnI (32%). 20
In this study we investigated the performance of two recently developed cTn assays, the precommercial high-sensitive cTnT assay (hs-cTnT) (Roche Diagnostics, Mannheim, Germany) and the Architect i2000SR cTnI assay (cTnI) (Abbott Diagnostics, Wiesbaden, Germany), in measuring cTn concentrations in a population of haemodialysis patients. For comparison, cTnT was also measured using the current fourth-generation cTnT immunoassay (Roche Diagnostics, Mannheim, Germany). In addition, we assessed the intraindividual variation in the cTn concentrations during a six-month follow-up.
Materials and methods
Reference population
Baseline cardiac biomarker concentrations were determined in a reference population of 501 apparently healthy persons who participated in a health check programme at the University Hospital Maastricht, the Netherlands. In accordance with the recent NACB practice guidelines in acute coronary syndromes, the 99th percentile upper reference limits (URLs) were determined in this population. 9 To exclude participants with potential cardiac syndromes, outliers were excluded when cTn (I or T), N-Terminal Pro-Brain-type Natriuretic Peptide (NT-proBNP) or creatine kinase MB isoenzyme (CK-MB) concentrations exceeded the mean concentration by more than three times the standard deviation. The cut-off concentrations for CK-MB and NT-proBNP were defined gender specifically. The cut-off concentrations for CK-MB were 10 μg/L for the men and 7.9 μg/L for the women. The NT-proBNP cut-off concentration was 41 pmol/L (=347 pg/mL) for both men and women. In effect, 22 subjects were excluded because of outlying cardiac biomarker concentrations, 10 subjects on the basis of CK-MB, eight subjects because of NT-proBNP and four subjects because of cTn (I or T). Additionally, two subjects were excluded because they were <18 years old, leaving a total of 477 subjects.
Patient population
A cohort of 44 chronic haemodialysis patients from the Department of Internal Medicine at the University Hospital Maastricht was selected to participate in this six-month longitudinal study. The study protocol was approved by the medical ethical review committee of the University Hospital Maastricht (Medical Ethical Committee azM/UM). All patients provided written informed consent. Blood samples were collected predialysis at the start of the study and subsequently every two months for a period of six months. During the follow-up period, 12 patients were hospitalized. The reasons for hospitalization were very diverse, briefly: pneumonia; stoma operation; removal of kidney transplant; kidney transplant; malaise after chemotherapy; rectal blood loss/diarrhoea; nausea, vomiting; hip replacement; kidney transplant, followed by ileus; hypoglycaemia/collapse; dizziness/fall; and subdural haematoma. Since one of our aims was to study the occurrence of cTn variations in a stable (e.g. with no acute worsening in the patient's condition) population of ESRD patients, hospitalized patients were excluded from our analysis. The blood samples collected were allowed to clot and then centrifuged: the serum samples obtained were stored at −80°C until analysis. Four patients failed to have blood taken at one occasion. Clinical data were collected from the medical records in our hospital. Patients could be divided into two groups on the basis of a history of cardio vascular disease (CVD). CVD was considered present when patients had a history of myocardial infarction, had required coronary intervention (e.g. percutaneous transluminal coronary angioplasty, coronary artery bypass grafting) or suffered from congestive cardiac failure.
Laboratory methods
CTnT was measured on the Elecsys 2010 (Roche Diagnostics, Mannheim, Germany) using the fourth-generation cTnT immunoassay with an LOD of 0.01 μg/L and a 10% CV of 0.03 μg/L (according to package insert). CTnT was additionally measured using the precommercial high-sensitive cTnT (hs-cTnT) assay on the Elecsys 2010. According to the manufacturer's data (J Jarausch, personal communication), the 10% CV was estimated at 0.012 μg/L. CTnI was measured on the Architect i2000SR (Abbott Diagnostics, Wiesbaden, Germany). According to the manufacturer's data, the LOD of the assay is 0.009 μg/L and the 10% CV concentration is 0.032 μg/L. Precision profiles for the hs-cTnT and the cTnI assay were determined in our laboratory by repeated (n = 20) measurements in seven pooled serum samples (cTnI concentrations ranging from 0.017 to 0.088 μg/L and cTnT concentrations ranging from 0.002 to 0.019 μg/L) as described previously. 21 The resulting 10% CV concentrations for the hs-cTnT and cTnI assays were found at 0.009 and 0.032 μg/L, respectively. The LODs for the hs-cTnT and cTnI assays were calculated in our laboratory according to the Clinical and Laboratory Standards Institute EP17 guidelines. According to these guidelines the LOD = (meanblank + 1.645 × SDblank) + (1.645 × SDlow concentration sample). The LOB, determined in cTn-negative serum (supplied to us by Abbott Diagnostics) by replicate (n = 10) measurements, revealed an LOB of 0.006 μg/L for the cTnI assay (mean cTnI = 0.001 μg/L; standard deviation [SD] = 0.003 μg/L) and an LOB of <0.001 μg/L for the hs-cTnT assay (mean and SD <0.001 μg/L). Replicate measurements (n = 20) in a serum sample containing a mean cTnI concentration of 0.017 μg/L showed an SD of 0.003 μg/L for the Architect cTnI assay. Replicate measurements (n = 20) with the hs-cTnT assay in a serum sample containing a mean cTnT concentration of 0.002 μg/L showed an SD of 0.00072 μg/L. The resulting LOD is thus established at 0.011 μg/L for the cTnI assay and at 0.001 μg/L for the hs-cTnT assay. In subsequent calculations, cTnI concentrations measured below the analytical LOD were set equal to the LOD of the assay.
Statistical analysis
Data analyses were performed using Statistical Package for Social Sciences (SPSS), Version 15.0, for Windows (SPSS Inc., Chicago, IL, USA). Continuous variables are described as median and interquartile range (IQR) or as mean and SD. Categorical variables are described as absolute numbers and as percentages. Biovariability data were analysed by calculating the intraindividual coefficient of variation (CVi) and the range (min–max) of cTn concentrations measured during the follow-up. The CVi was calculated by subtracting the analytical variation (CVa) from the total variation (CVtot), such that CVtot2 = CVi2 + CVa2. The analytical variation is estimated by extrapolating the CVa from the precision profiles for the hs-cTnT and cTnI assays (precision profiles shown previously 21 ). For the fourth-generation cTnT assay the CVa is extrapolated from the precision profile in the package insert for this assay. The maximum percent change during the six-month period for each of the patients is calculated as the ((maximum concentration measured during the 6-month period minus the minimum concentration)/minimum concentration) × 100%. Differences in the cTn concentrations and variations (ranges) between patients with and without cardiac disease were compared using the Mann–Whitney U test. The level of statistical significance was established at P < 0.05.
Results
The reference population
Our reference population consisted of 264 men and 213 women with an average (SD) age of 51 yr (10.5). The mean cTnT concentrations according to the hs-cTnT assay were 0.004 μg/L (SD = 0.003 μg/L) and the mean cTnI concentration was 0.001 μg/L (SD = 0.003 μg/L). The 99th percentile URLs for the cTn assays under investigation were 0.016 μg/L for the hs-cTnT assay and 0.013 μg/L for the cTnI assay. The 99th percentile URL for the current fourth-generation cTnT assay was set at 0.01 μg/L because all of the measurements fell below the LOD (<0.01 μg/L).
Figure 1 shows the cTn concentrations measured by the hs-cTnT (Figure 1A) and the cTnI assays (Figure 1B). According to the hs-cTnT assay, 92% of the cTnT concentrations were above the LOD and followed an almost Gaussian distribution. In contrast, only 2% of the concentrations measured by the cTnI assay were above the LOD. Furthermore, the 99th percentile URL calculated for the hs-cTnT assay was above the concentration at which the CV is <10%. Conversely, for the cTnI assay the 99th percentile URL was below the concentration at which the CV was <10%.
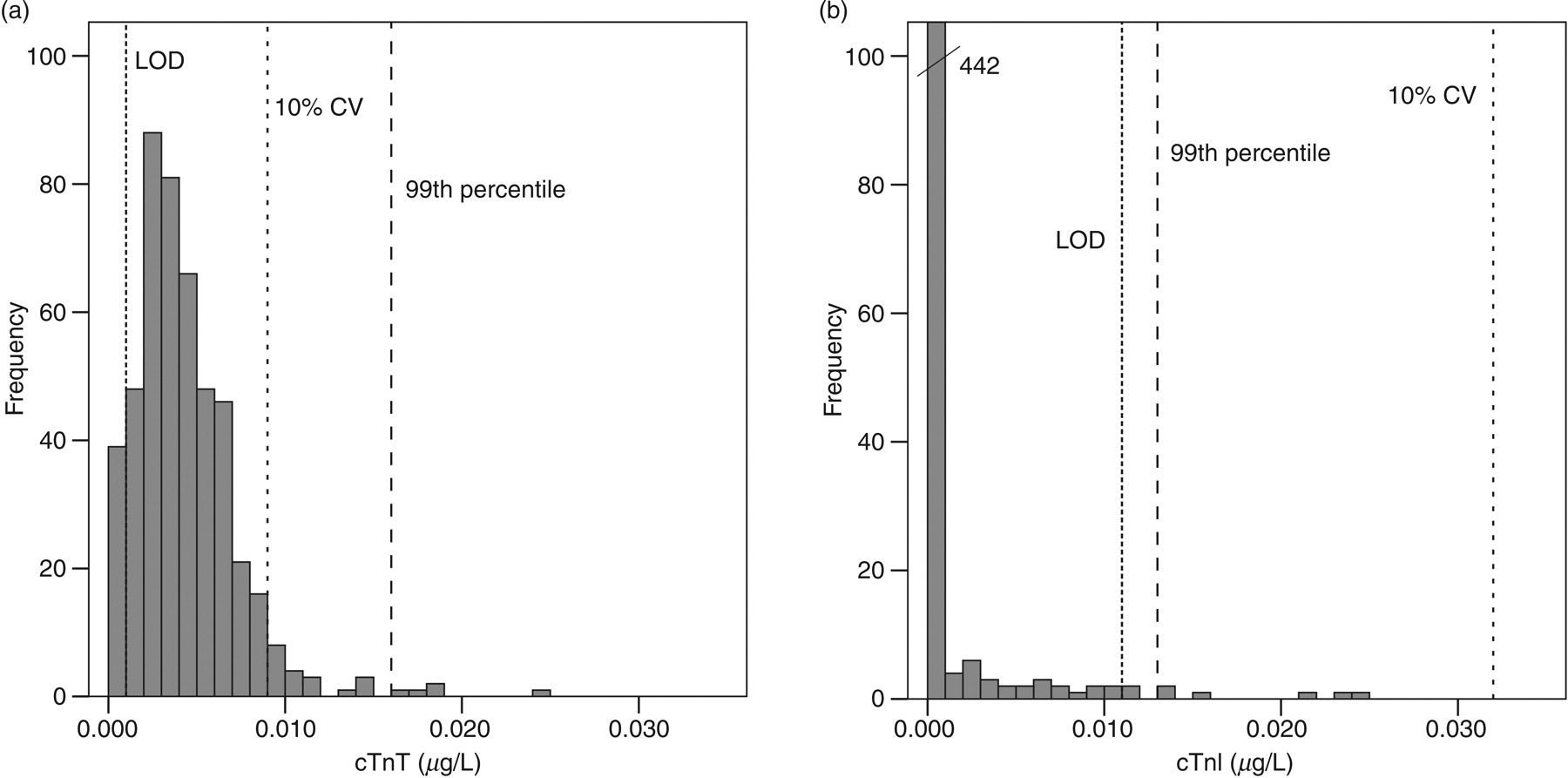
CTn concentrations in the reference population as measured with the (a) hs-cTnT and (b) the cTnI assay
Baseline measurements in the patient population
Table 1 shows the characteristics of the 32 ESRD patients enrolled in our study. Measurements at baseline revealed median cTn concentrations (IQR) as detected by the cTnT, hs-cTnT and cTnI assays of 0.02 μg/L (<0.01–0.05), 0.053 μg/L (0.032–0.076) and 0.014 μg/L (0.011–0.027), respectively. Table 2 shows the number of patients having cTn concentrations above the different analytical cut-off values. At baseline 12 (38%), 30 (94%) and 6 (19%) patients showed cTn concentrations above the designated AMI cut-off values according to the cTnT, the hs-cTnT and the cTnI assays, respectively.
Baseline characteristics of the end-stage renal disease patient population
Number of patients having above cut-off cardiac troponin concentrations at baseline and at least once during the six-month follow-up
*According to recent National Academy of Clinical Biochemistry guidelines the 99th percentile is the recommended cut-off concentration in AMI. However, the assay imprecision (%CV) should be ≤10% at the 99th percentile, 9 otherwise the 10% CV concentration is used
LOD, limit of detection; CV, coefficient of variation; AMI, acute myocardial infarction; hs-cTnT, high-sensitive cardiac troponin T; cTnI, cardiac troponin I
Serial measurements in the patient population
Figure 2 shows the range of concentrations measured during the follow-up in each patient for each of the biomarkers under investigation for both patients with (n = 17) and without (n = 15) a history of CVD. This section discusses the range of cTn concentrations measured in all patients; in the next section, we will investigate the differences between patients with and without CVD.
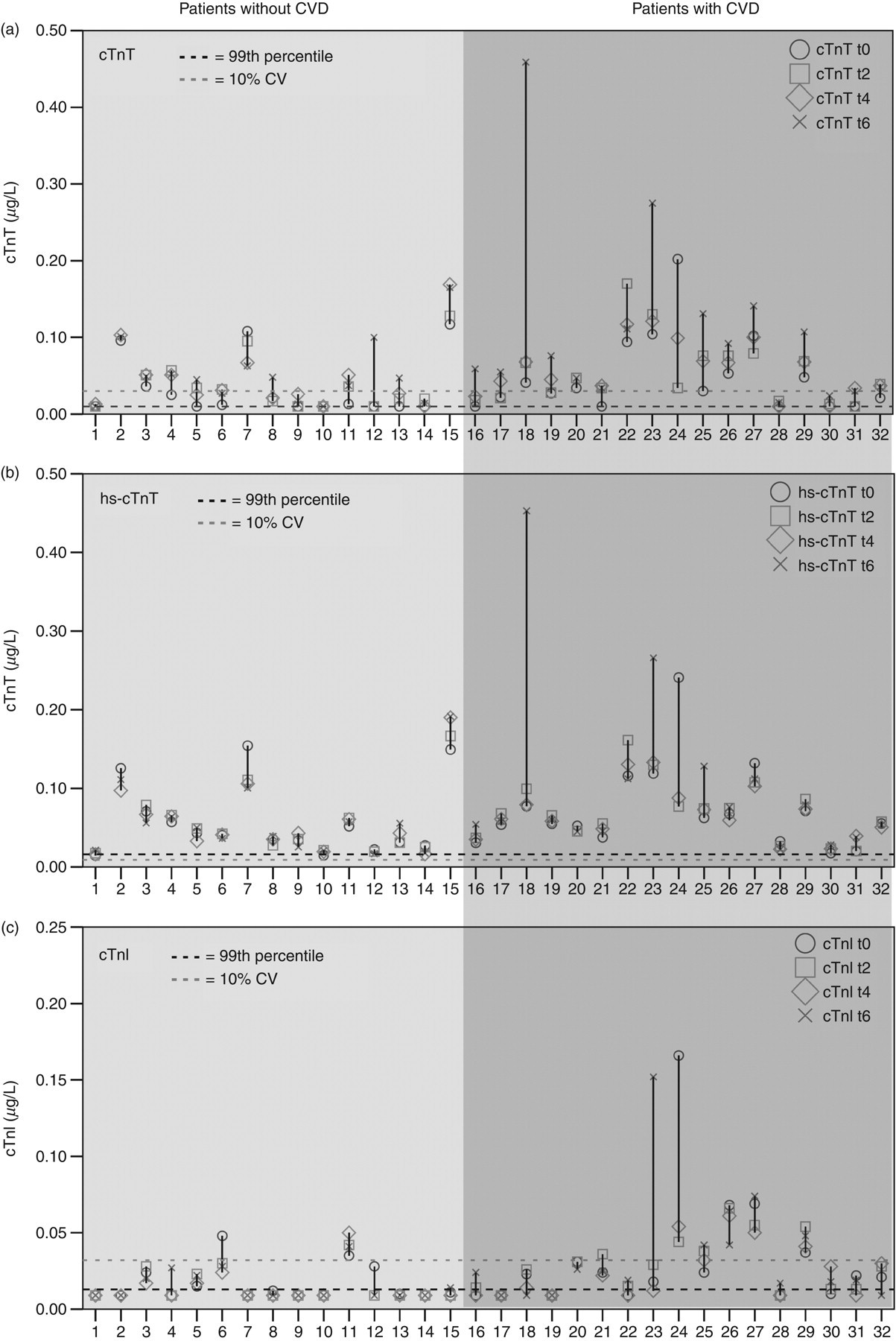
Range of concentrations measured in each patient during the study. (a) cTnT concentrations according to the cTnT assay (b) cTnT concentrations according to the hs-cTnT assay (c) cTnI concentrations
During the six-month follow-up, we found a median (IQR) cTn concentration of 0.037 μg/L (0.017–0.074), 0.055 μg/L (0.033–0.079) and 0.014 μg/L (<0.011–0.028) for the cTnT, hs-cTnT and cTnI assays, respectively. As can be seen in Figure 2, the patients showed huge changes in the cTn concentrations during the follow-up, and the range of concentrations measured within each patient had a median (IQR) magnitude of 0.03 μg/L (0.02–0.06), 0.017 μg/L (0.011–0.029) and 0.011 μg/L (0–0.017) according to the cTnT, hs-cTnT and cTnI assays, respectively. This corresponded to a median within-patient percent change of 150% (72–287), 44% (27–69) and 54% (0–114) for the cTnT, hs-cTnT and cTnI assays, respectively. The total coefficient of variation (CVtot) was 40%, 16% and 20% for the aforementioned assays. The CVa belonging to the median concentrations measured during the follow-up (as estimated from the precision profiles) was 7% according to the cTnT assay (median cTnT = 0.037 μg/L), 5% according to the hs-cTnT assay (median cTnT = 0.055 μg/L) and 15% for the cTnI assay (median cTnI = 0.014 μg/L). The CVi is thus 39%, 15% and 13% for the cTnT, hs-cTnT and cTnI assays, respectively.
Considering the magnitude of the variation, it is not surprising that the serial measurements identified additional patients who had cTn concentrations above the previously mentioned cut-off concentrations (at least once) during the six-month period. During the follow-up we found that 26 (81%), 32 (100%) and 9 (28%) patients had cTn concentrations above the AMI cut-off level according to the cTnT, hs-cTnT and cTnI assays, respectively. Table 2 shows the number of patients exceeding the predefined cut-off concentrations during the six-month period.
Patients with and without a history of cardiovascular disease
As could already be seen in Figure 2, large differences exist between the cTn concentrations and variations in patients with and without a history of CVD. Table 3 quantifies the differences in the cTn concentrations measured during the follow-up. The three assays showed significantly higher concentrations of cTn in the patients who suffered from CVD. In addition, as might be expected, the relative number of patients having elevated concentrations of cTn was higher in the group with CVD. This difference was particularly large for the cTnI assay, where only two out of 15 patients without CVD had cTnI concentrations above the AMI cut-off concentration, compared with seven out of 17 in the patients with CVD. Moreover, the variations in the cTn concentrations, as assessed by the ranges of concentrations measured during the follow-up, were larger in the CVD patients. Note however, that, as can be seen in Table 4, the variations in cTn concentrations are, in part, the result of analytical rather than biological variations.
Differences in cTn concentrations between patients with and without a history of CVD
cTn, cardiac troponin; CVD, cardio vascular disease; IQR, interquartile range; cTnT, cardiac troponin T; hs-cTnT, high-sensitive cardiac troponin T; cTnI, cardiac troponin I
Intraindividual coefficients of variation in patients with and without a history of CVD
CVD, cardio vascular disease; CVtot, total variation; CVa, analytical variation; CVi, intraindividual coefficient of variation; cTnT, cardiac troponin T; hs-cTnT, high-sensitive cardiac troponin T; cTnI, cardiac troponin I
*The coefficients of variations for the patient without CVD could not be determined accurately for the cTnI assay
When the results were assessed for the individual patients, we found a large degree of heterogeneity in the agreement between cTnT and cTnI elevations. In some cases, as with patients 23 and 24, we found drastically elevated concentrations of both cTnT and cTnI, both of which seemed to increase/decrease at the same time. On the other hand, there were patients, such as 7, 15 and 18, who showed very strong elevations (i.e. >0.1 μg/L) in cTnT (according to both cTnT assays), and yet virtually no elevations in cTnI concentrations. In general, it seems that patients with elevated concentrations of cTnI also have elevated concentrations of cTnT, but patients with elevated concentrations of cTnT do not always have elevated concentrations of cTnI.
Discussion
This study describes, for the first time, the performance of a precommercial hs-cTnT assay in measuring cTn elevations and variations in a population of ESRD patients during a six-month follow-up. In addition, we compared its performance with the current fourth-generation cTnT and the Architect cTnI assays. In the past, several studies have shown the occurrence of elevated cTn concentrations in ESRD patients without apparent cardiac damage and clinical symptoms. 22–26 These elevated concentrations of cTn are of cardiac origin 27 and were shown to be highly predictive for a cardiovascular event. 3,4,24 A meta-analysis combining results from 28 studies (3931 patients) showed that cTnT concentrations above 0.1 μg/L were associated with an increase in all-cause mortality (relative risk: 2.64). 3 Other studies have shown an increased mortality in ESRD patients with cTnT concentrations above 0.026 μg/L24 and 0.01 μg/l. 4 Due to the lack of standardization in the available assays, the prognostic value of cTnI is less conclusive and cut-off concentrations cannot be compared between assays. Nonetheless, elevated concentrations of cTnI have also been shown to be associated with an increased mortality in ESRD patients. 4,22
In agreement with previous studies, 22–26 we found that 12 (38%) of our patients had elevated concentrations of cTnT according to the current cTnT assay and six (19%) had elevated concentrations of cTnI according to the Architect cTnI assay. Using the recently developed precommercial hs-cTnT assay we find that the occurrence of elevated concentrations of cTnT is far greater than previously described, and at baseline 30 (94%) of the ESRD patients showed elevated concentrations of cTnT.
By following the cTn concentrations at regular points in time, we have identified an additional number of patients with elevated cTn concentrations. During the follow-up, 26 (81%), 32 (100%) and 9 (28%) of the patients showed cTn concentrations above the AMI cut-off concentrations according to the cTnT, hs-cTnT and cTnI assays, respectively. As elevated concentrations of cTn are highly prognostic of adverse events in ESRD patients, the use of serial measurements has thus identified additional patients at risk for such events. The fact that we find cTn concentrations to be higher in patients with a history of cardiovascular disease seems to be in line with this.
We should note, however, that due to differences in the precision with which the 99th percentile can be determined, the AMI cut-offs for the assays are set at different concentrations. For the cTnT and cTnI assays, the 10% CV cut-off concentrations are used, whereas for the hs-cTnT assay, the 99th percentile URL is used. When we assess the occurrence of cTn elevations above the 99th percentile URL, we find that the difference between the assays is much smaller and 31 (97%) of the patients have cTnT elevations and 22 (69%) have cTnI elevations (versus 32 [100%] for the hs-cTnT assay). Considering the fact that the fourth-generation cTnT and the Architect cTnI assays were not able to detect the 99th percentile with sufficient precision (<10 CV%) these findings, however, should be interpreted with caution. Nonetheless, they suggest that more sensitive cTnI measurements could reveal that the occurrence of cTnI elevations in ESRD patients is more frequent than is presently thought. This idea is supported by recent findings with sensitive cTnI assays in ESRD 20 and chronic kidney disease patients, 28 which find more cTnI elevations than previously described.
In general, there seems to be a huge degree of heterogeneity between the patients in the extent of variation in their cTn concentrations. For instance, according to the new hs-cTnT assay, eight (25%) of the patients showed changes in cTnT >0.03 μg/L, whereas another eight (25%) of the patients showed variations <0.01 μg/L during the six-month follow-up. Thus, as can also been seen in Figure 2, the patient population consisted of a group of patients with relatively stable cTn concentrations and a group with a high degree of variation in their cTn concentrations. Further research in a larger population is needed to identify whether the patients with the higher degree of variation are at a greater risk for adverse events.
According to recent NACB guidelines, only patients with chronically elevated concentrations of cTn, with changes in cTn (>20%) 6–9 h after the onset of clinical symptoms, are classified as suffering from an AMI. 11 Therefore, none of our patients would have been classified as having an AMI. Nonetheless, as increases in cTn represent further (subclinical) myocardial damage and an increased likelihood for cardiac events, they should not be ignored. It would therefore be prudent to perform a detailed cardiac evaluation in patients showing large variations in cTn concentrations, even if these variations are not accompanied by clinical symptoms.
The increases in cTn, as seen in many of our patients, would, if not for the longitudinal study, have been detected. Assessing cTn concentrations at regular points in time therefore appears to be a sensible tool to increase clinical vigilance for the presence of myocardial damage and as a means for possible intervention. This is in agreement with previous studies that provided evidence for the increased ability of serial versus single measurements to identify patients at risk for an event. 12–16
In the setting of heart failure, previously undetectable concentrations of cTnT were shown to have an important prognostic value. 29 The use of highly sensitive cTn assays, like the hs-cTnT assay, will enable better differentiation between cTn concentrations in healthy and diseased patients and could improve risk stratification. Unfortunately, our study was limited by a small sample size and the absence of outcomes. Therefore, additional research is needed to show whether this prognostic value exists in the setting of ESRD and how patients can further benefit from serial measurements with highly sensitive cTn assays.
Declarations
