Abstract
Background
The aims of this study were to examine the relationship between proteinuria and albuminuria and to assess the equivalence between the albumin to creatinine ratio (ACR) and the protein to creatinine ratio (PCR) at the cut-offs recommended by the National Institute for Health and Clinical Excellence (NICE) guidance on chronic kidney disease. The sensitivity and specificity of the reagent strips used in our laboratory for the detection of clinical proteinuria was also assessed.
Methods
Urine samples (n = 117) were screened for protein using the Bayer Multistix 10SG and read manually. Urinary total protein and creatinine was measured on the Roche P Modular by the benzethonium chloride and kinetic Jaffe methods, respectively. Urinary albumin was measured by immunoturbidimetry on the Roche Cobas Mira.
Results
The relationship between urinary protein and albumin loss was non-linear (P < 0.05). As urinary protein loss increased the percentage of albumin to total protein increased. At the NICE guidance recommended cut-offs for clinical proteinuria (ACR ≥30 mg/mmol and PCR ≥50 mg/mmol) there was one discordant result between ACR and PCR (ACR <30 mg/mmol and PCR >50 mg/mmol). The Bayer Multistix 10SG had a sensitivity and specificity of 97% and 62%, respectively, for the detection of clinical proteinuria compared with ACR.
Conclusions
The proportion of urinary total protein attributable to albumin changes with concentration. There was only one discordant result between ACR and PCR: therefore either ratio may be used for the identification of clinical proteinuria. As a screening test for proteinuria, the Bayer Multistix 10SG had an acceptable sensitivity but poor specificity.
Introduction
The persistent presence of proteinuria is evidence of kidney damage and an independent risk factor for both kidney and cardiovascular disease. 1 The UK Chronic Kidney Disease (UKCKD) Guidelines 2 recommended that patients with a positive reagent strip reading (≥+1) should undergo confirmation of proteinuria by a quantitative method, either the urinary protein to creatinine ratio (PCR) or the albumin to creatinine ratio (ACR). The recently published National Institute for Health and Clinical Excellence (NICE) guidance on chronic kidney disease 3 suggests that reagent strips in current practice detect albumin and not total protein and should not be used to identify proteinuria unless they are capable of specifically measuring albumin at low concentrations and expressing the result as an ACR. The guidance also suggests that to identify and detect proteinuria ACR should be used in preference to PCR for low concentrations of proteinuria.
The aims of this study were two-fold: first to determine the relationship between albuminuria and total proteinuria and the equivalence between ACR and PCR at the NICE recommended cut-offs for the detection of clinical proteinuria. Secondly, to assess the sensitivity and specificity of the Bayer Urine Multistix 10SG reagent strips as a screening test for the detection of clinical proteinuria.
Subjects and methods
Spot urine specimens from 117 patients were screened by Bayer Multistix 10SG reagent strips (Bayer Diagnostic, MFG Ltd, Bridgend, UK) for the presence of protein (manually read) and reported as negative, trace-spur+, ++ and +++, in accordance with the manufacturer's nomenclature for protein. Albumin, total protein and creatinine were measured in each sample. Urinary albumin was measured by immunoturbidimetry on the Cobas Mira analyser (Roche Instruments, Rotkreuz, Switzerland). Urinary total protein and creatinine were measured by the benzethonium chloride and the rate-blanked Jaffe methods, respectively, on the Roche/Hitachi Modular P analyser (Hitachi High-Technologies Corporation, Japan). From these measurements, the ACR and PCR were calculated and reported in milligrams/millimoles. As the data were non-parametrically distributed, the relationship between albuminuria and proteinuria was examined using Passing Bablok regression analysis with cumulative sum (CUSUM) linearity tests (Medcalc® Software, Belgium). All samples were anonymized in accordance with the Royal College of Pathologists' guidance. 4
Results
Relationship between total proteinuria and albuminuria
As urinary total protein loss increased, the percentage of albumin to total protein also increased. At negative (n = 27), trace-spur (n = 30), + (n = 31), ++ (n = 22) and +++ (n = 7) reagent strip readings, the percentage of albumin to total protein was 31%, 39%, 51%, 68% and 74%, respectively.
The Passing Bablok regression equation for ACR and PCR was ACR (mg/mmol) = −4.0 + 0.68 PCR (mg/mmol). The CUSUM test for linearity showed a significant deviation from linearity (P < 0.01) for the relationship between ACR and PCR.
Equivalence between ACR and PCR
At negative and trace reagent strip readings, the median PCR was 10.5 mg/mmol (range 2.8–49.9 mg/mmol) and the median ACR was 2.5 mg/mmol (range 0.50–27.0mg/mmol). At reagent strip reading ≥ +1, the median PCR was 52.3 mg/mmol (range 11.3–436 mg/mmol) and median ACR was 34.6 mg/mmol (range 1.1–155 mg/mmol).
The NICE guidance would consider clinically significant proteinuria in people without diabetes to be present when the ACR is ≥30 mg/mmol (approximately equivalent to PCR ≥50 mg/mmol). Using these cut-offs, there was only one discordant result between ACR and PCR (n = 103) for the detection of clinical proteinuria. This result had an ACR <30 mg/mmol and a PCR >50 mg/mmol (Figure 1A).
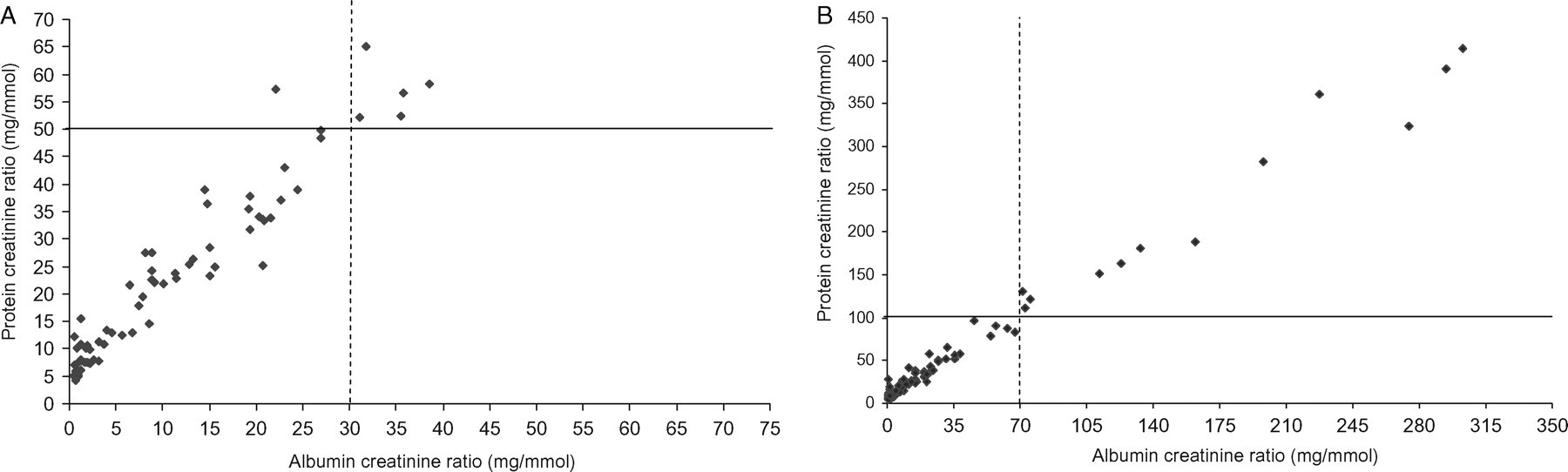
(A) Scatter plot showing the relationship between albumin to creatinine ratio (ACR) and protein to creatinine ratio (PCR) at the stated NICE guidance cut-offs for clinical proteinuria: ACR 30 mg/mmol (- - - -) and PCR 50 mg/mmol (———). (B) Scatter plot showing the relationship between ACR and PCR at the stated NICE guidance cut-offs for heavy proteinuria: ACR 70 mg/mmol (- - - -) and PCR 100 mg/mmol (———).
The NICE guidance also suggests that if the initial ACR ≥70 mg/mmol which is approximately equivalent to PCR ≥100 mg/mmol, a repeat urine sample need not be tested and that these cut-offs are consistent with heavy proteinuria. In this study there were no discordant results between ACR and PCR at these stated cut-offs as shown in Figure 1B.
The sensitivity and specificity of Bayer Multistix Dipstick
Using ACR at a cut-off ≥30 mg/mmol as the ‘gold standard’ for the detection of clinical proteinuria, the Bayer Multistix 10SG reagent strip reading of + had a sensitivity of 97% and a specificity of 62%.
Discussion
This study concurs with other studies which have shown a non-constant relationship between the proportion of urinary total protein attributable to albumin. 5 The NICE guidance recommends the use of urine ACR in preference to PCR for the detection of low concentrations of proteinuria. In this study, there was only one discordant result between ACR and PCR at the recommended cut-offs of 30 mg/mmol and 50 mg/mmol for clinical proteinuria, respectively. Unfortunately, we were unable to confirm the nature of the proteinuria in this patient. However, results from this study would indicate that the use of either ratio may be appropriate for the detection of clinical proteinuria.
As a screening test for clinical proteinuria, the Bayer Multistix 10SG reagent strips had an acceptable sensitivity because of the small number of false-negatives but had a poor specificity resulting in a large number of false-positives for clinical proteinuria.
