Abstract
Background
Although serum calcium has been measured using the o-cresolphthalein complexone (oCPC) method in the clinical laboratory, this method still has some problems regarding linearity and reagent stability. We developed a new measurement procedure using chlorophosphonazo-III (CPZ-III: 2,7-bis (4-chloro-2-phosphonophenylazo) -1,8- dihydroxy-3, 6-naphthalenedisulphonic acid, disodium salt) as a chelator with an acid medium for serum calcium measurement. The present method showed better linearity and reagent stability compared with the oCPC method.
Methods
Characteristics were studied in optimized conditions measuring wavelength by absorption spectra analysis, and interference of protein and metals with Mg2+, Fe2+, Cu2+ and Zn2+. The method was applied to an automated analyser (7170; Hitachi High Technologies Corp). The measurement performance was evaluated for accuracy, precision, recovery rate, linearity and reagent stability with a comparison study against atomic absorption spectrophotometry (AAS).
Results
The within-run and between-run variations (coefficient of variation [CV]) were 0.92–1.01% and 0.75–1.43%, respectively. The linearity was 0–7.0 mmol/L. The comparison study obtained y = 1.002x (AAS) – 0.10, Sy/x = 0.18 mmol/L, n = 50. Reagent stability was at least 20 d at 4°C without daily calibration.
Conclusion
The new calcium measurement method in serum was demonstrated to have reliable and acceptable performances as a routine test in clinical laboratory.
Introduction
Calcium is an important element for maintaining bone growth and strength. Furthermore, it is necessary for muscle contraction, hormone secretion and other important functions. In medical treatment, serum calcium is often used for the diagnosis of hypercalcaemia in paraneoplastic syndrome or metabolic disease related to abnormal thyroid gland functions. Various methods are used in the laboratory to measure calcium in body fluids. Among them, atomic absorption spectrophotometry (AAS) is the most reliable method, 1,2 but it requires specialized and expensive instruments, making it unsuitable for routine assay in clinical laboratories. The most widely used approach is the colorimetric method, which employs chelators such as o-cresolphthalein complexone (oCPC), 3–5 methylxylenol blue (MXB) 6 and arsenazo-III (AZ-III). 7,8 Initially, the oCPC method presented problems of reagent stability, linearity and recovery because magnesium interferes with reactions. That widely used method was subsequently improved by Kessler and Wolfman, 4 who made it more specific for calcium through addition of 8-hydroxyquinoline to mask any magnesium present. However, this method is not entirely satisfactory in terms of recovery at low concentrations and the instability attributable to the alkaline medium reagent. Moreover, the MXB method provides unsatisfactory stability due to the alkaline medium. From that perspective, the AZ-III and CPZ-III methods provide satisfactory stability because the reaction is in a neutral or acid medium. Nevertheless, the former includes a trace of arsenic within its structure. Therefore, its effect on the environment is questionable. Furthermore, although providing high accuracy, enzymatic methods using pancreatic α-amylase 9 or phospholipase D 10 are costly, even on a per-test basis.
Here we report an original, sensitive and specific method for serum calcium using CPZ-III reacting in an acid medium. It uses no heavy metals.
Measurement principle
Figure 1 depicts the constitutional formula of CPZ-III, which has properties of combination for actinide, calcium, magnesium,and so on in the ratio of 1:1–1:3 as a chelate compound. According to Budesinsky, 11 the dissociation constants (pKa) of CPZ-III are −1.1, −1.1, 0.6, 0.8, 1.5, 2.5, 5.47, 7.20, 12.15 and 15.3 (20°C). Its reaction solution, after chelating, becomes purple or violet.
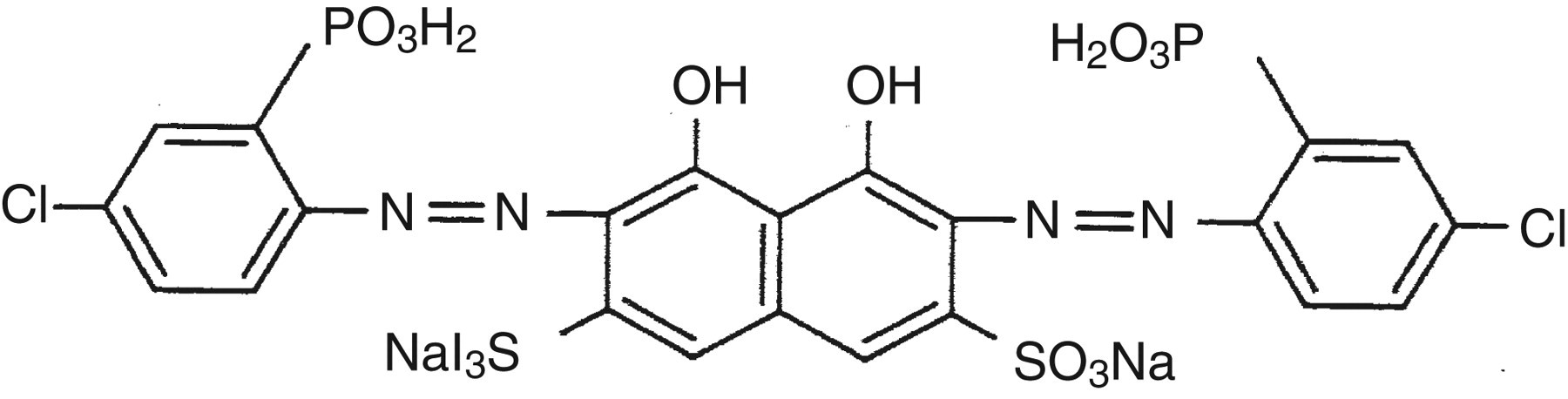
Constitution formula of chlorophosphonazo-III
Materials and methods
Instrumentation
In this examination and comparative studies, we used an automatic analyser (7170; Hitachi High-Technologies Corp, Tokyo, Japan) for the present method, the oCPC method and the enzymatic method.
Reagents
In the assay, we used a two-reagent system. Reagent 1 (R-1) consisted of 50 mmol/L succinic acid (pH 5.0 at 25°C) and 0.5% anionic surfactant (e.g. sodium alpha-olefinsulphonate; CAS RN 29963-33-5). This surfactant concentration was determined to the most adequate level in the measurement, for avoiding interference of albumin (data not shown). The chelate reagent (R-2) contained 1.8 mmol/L CPZ-III (Dojindo Laboratories, Kumamoto, Japan) in R-1.
We obtained a standard solution for the calibrator (concentration of 2.5 mmol/L, Multi-calib 6 mmol/L; Kanto Chemical Co, Inc, Tokyo, Japan) and JCCRM 321 as a certified serum reference material consisting of two levels by Reference Material Institute for Clinical Chemistry Standards (ReCCS, Kanagawa, Japan). Certification values for these serum reference materials were verified using the results of AAS (Secondary Reference Measurement Procedure) calibrated using the National Institute of Standards and Technology SRN 909b. 12,13 We used a control sera (Aalto Control Level I,Z, II,N; Aalto Scientific, Ltd, CA, USA). Measurement reagents were used for the oCPC method (Kanto Chemical Co Inc, Tokyo, Japan); those for the enzymatic method (Toyobo Co, Ltd, Osaka, Japan) were based on activation of porcine pancreatic α-amylase (EC 3.2.1.1) with 2-chloro-4-nitrophenyl-alpha-maltrioside as substrate. An interference test (Interference check A plus; Sysmex Co, Hyogo, Japan) was used for lipids, haemoglobin, and conjugated and unconjugated bilirubin. Other chemical reagents of analytical grade were used (Wako Pure Chemical Industries Ltd, Tokyo, Japan).
Specimens
Serum specimens were collected from inpatients and outpatients of Kyushu University Hospital after receiving their informed consent and approval from our institutional ethics committee.
Statistics
We used Altman–Bland difference plots for comparison studies using regression and correlation coefficients. Data were otherwise treated according to Annals' statistical guidelines.
Procedure
Analytical conditions
For our experiments, we used the following analyser settings: 4 μL specimens were mixed with 210 μL R-1; after incubation for 5 min at 37°C, 70 μL R-2 was added; the mixture was measured using two-point end assay performed at 37°C, with wavelengths of 800/700 nm (sub/main wavelength). Other methods for comparison studies were performed according to the recommended analyser setting information provided by the respective device manufacturers.
Results
Absorption spectra
The CPZ-III solution (0 mmol/L: calcium concentration) showed strong absorption at wavelengths equal to or less than 650 nm; the absorption spectrum changed in direct relation to the calcium concentration. Figure 2 depicts spectra of CPZ-III complexes with calcium (in deionized water or in serum) at pH 5.0 buffer solution. In consideration of the sensibility and influence of biological specimens (chylemia, bilirubin, etc.), we determined the measurement wavelength at 700 nm.
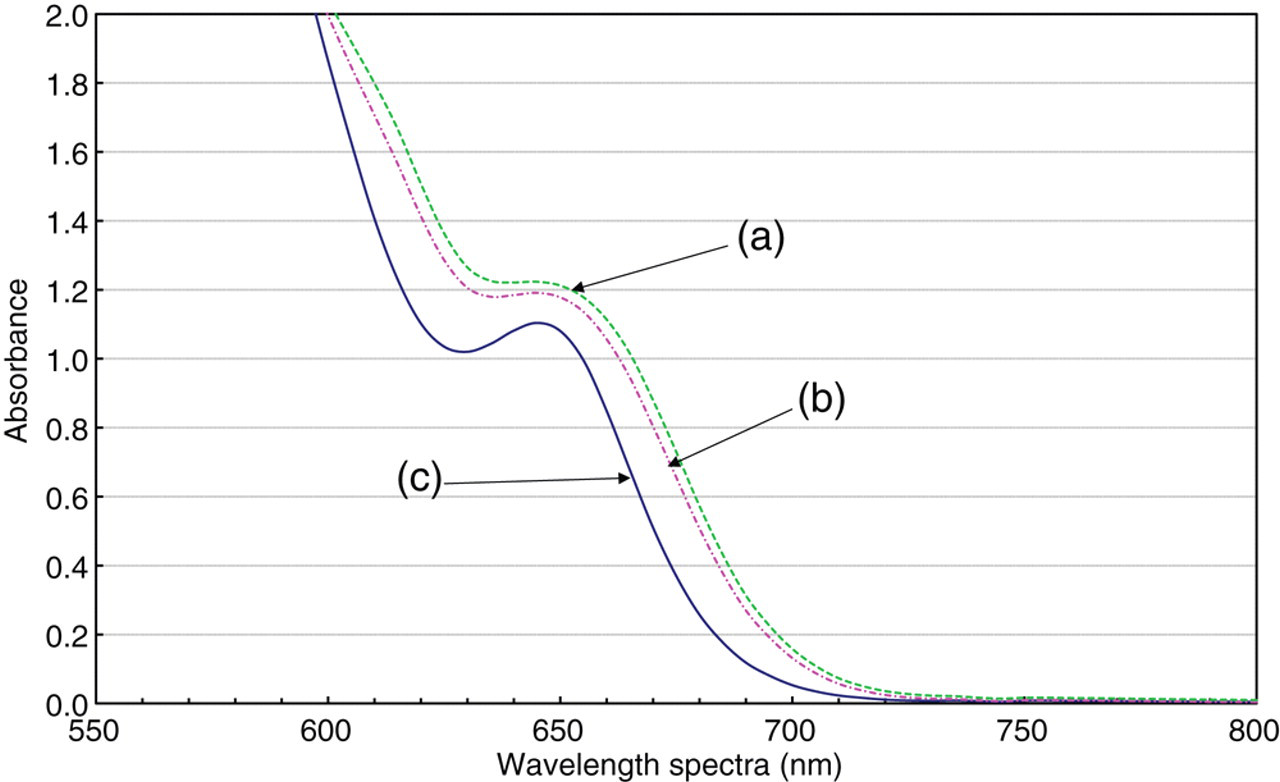
Absorption spectra of chlorophosphonazo-III (CPZ-III) complexes with calcium in a pH 5.0 buffer solution.CPZ-III final concentration = 0.075 mmol/L. Sample volume = 3.0 μL: (a) complexes with human serum (calcium concentration is approximately 2.35 mmol/L), (b) complexes with 2 mmol/L calcium solution and (c) no complexes with calcium (CPZ-III alone)
Optimum buffer pH and CPZ-III concentration
We reviewed the optimum buffer pH and CPZ-III concentration. This CPZ-III is known to react strongly with magnesium by neutral pH. Therefore, we first reviewed the buffer pH (5.0–5.6), which is not muddied by proteins, to find the best response specifically to calcium. As portrayed in Figure 3, buffer of pH 5.0 showed the weakest reaction with 8.23 mmol/L magnesium. Therefore, the preferred buffer pH was decided as 5.0. In addition, CPZ-III concentration in the reagent was adjusted so that the dynamic range of reagents displayed linearity for calcium concentrations between 0.0 and 7.5 mmol/L. We prepared CPZ-III solutions (final concentration 0.02–0.75 mmol/L) of six kinds by dissolving CPZ-III in the buffer at pH 5.0. Based on those results, we decided to use a CPZ-III solution with a final concentration of 0.44 mmol/L.
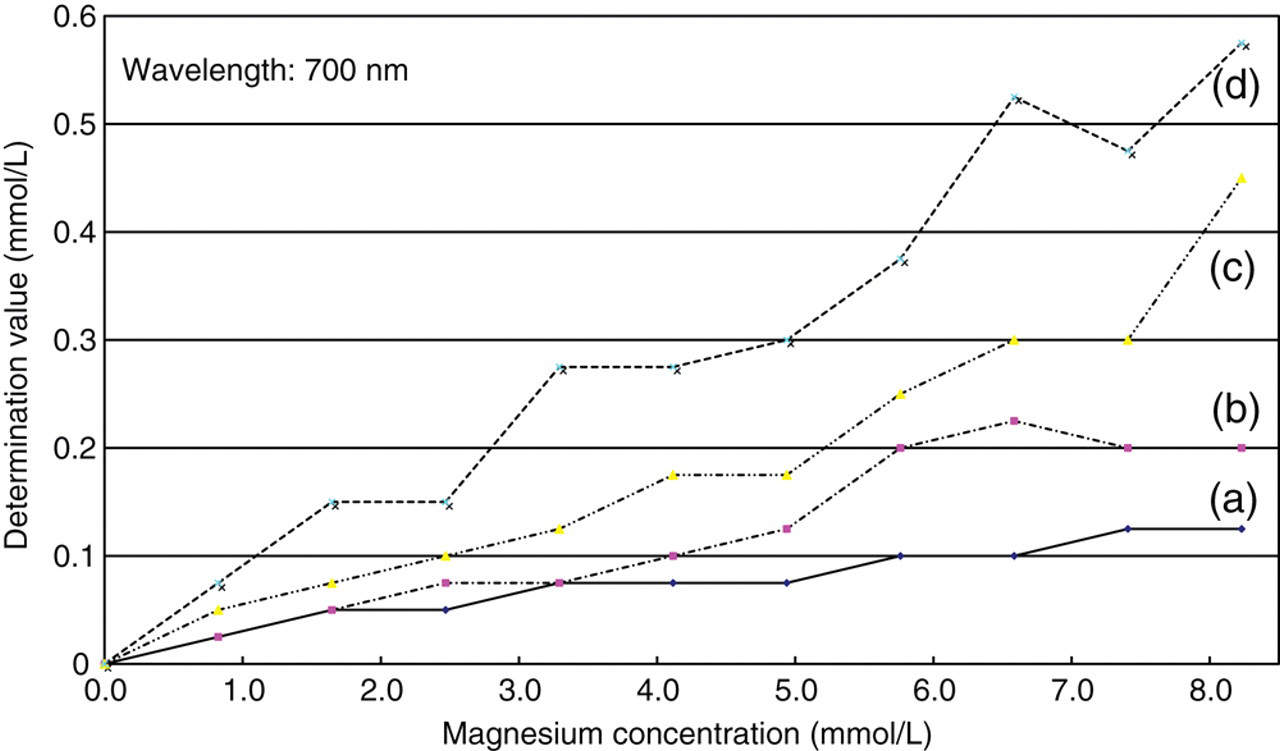
Reactive difference to magnesium by variations of buffer pH.Final chlorophosphonazo-III concentration: 0.075 mmol/L. Magnesium solution: magnesium acetate four hydrate was diluted with distilled water and added to 3.0 μL as a sample to produce (a) pH 5.0, (b) pH 5.2, (c) pH 5.4 and (d) pH 5.6
Reproducibility
Table 1 shows the within-run (n = 20) and between-run (n = 20) variations of data for two-level control sera. The within-run and between-run CVs were 0.92–1.01% and 0.75–1.43%, respectively.
Within- and between-run reproducibility (mmol/L) of chlorophosphonazo-III (CPZ-III) method
SD, standard deviation; CV, coefficient of variance
*Samples of control sera were prepared. Levels 1 and 2, respectively, indicate normal and above-normal level concentrations
†Mean of duplicate measurements
Linearity
The standard curve, obtained with approximately 2.5 and 7.0 mmol/L calcium chloride solutions dissolved using physiological saline, showed a linearity of 0 ∼ 7.0 mmol/L (Figures 4a and b).
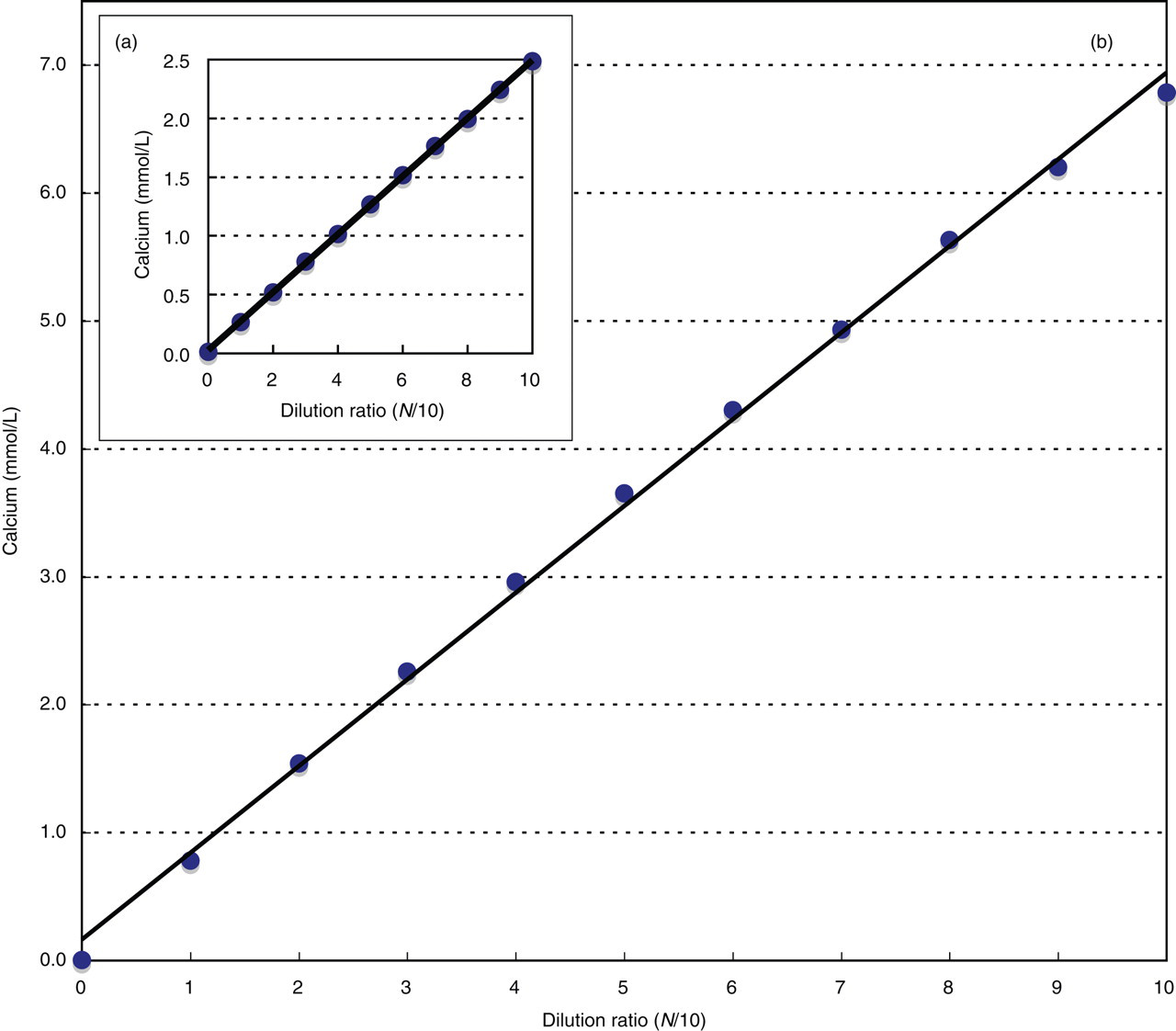
Dilution linearity curve for chlorophosphonazo-III method.Calcium chloride solution was used as the specimen. It was diluted successively with deionized water: (a) low concentration and (b) high concentration
Limit of detection
We estimated the limit of detection using calcium chloride solutions that had been diluted to five levels gradually and successively with physiological saline until zero concentration. The detection limit of our method, defined as the calcium concentration for the zero concentration (limit of absence) of ±3 standard deviation, was 45 μmol/L. The detection limit at the oCPC method was 225 μmol/L. Results show that our method can detect serum calcium of a lower concentration than the oCPC method.
Recovery test
The sample for analytical recovery was prepared as follows. Two kinds of pooled human sera were prepared for recovery tests. One was dialysed overnight in deionized water, the other without it. Nine volumes of the former and one volume of 13 mmol/L calcium chloride solution were mixed. Then four volumes of the latter and one volume of 2.5 mmol/L standard solution were mixed; these mixed solutions were used for recovery tests. Their final Ca concentration was 1.3 or 2.6 mmol/L. The average analytical recovery of triplicate assays was 101.9% and 98.5%, respectively.
Interfering substances
We examined whether interfering substances influence calcium detection. First, we studied the interference of metal ions: Mg2+, Fe2+, Cu2+ and Zn2+. No interference was observed when we added one volume of the metal ions to nine volumes of pooled serum to produce the respective final concentrations of 8.23 mmol/L, 134 μmol/L, 157 μmol/L, and 153 μmol/L: the obtained Ca values were 100.9–104.1% of the expected values. We then examined the interference of conjugated and unconjugated bilirubin, haemoglobin, turbidity and ascorbic acid with the medium for interference tests. Unconjugated bilirubin up to 195 mg/L, conjugated bilirubin up to 181 mg/L, haemoglobin up to 4.9 g/L, turbidity up to 2290 hormadin turbidity units (triglyceride 8.5 g/L, phospholipids 9.1 g/L, free fatty acid 620 μEq/L and bovine serum albumin 2.5 g/L) and ascorbic acid up to 2.8 mmol/L did not interfere.
We assessed anticoagulation using ethylenediamine tetraacetic acid (EDTA)-2Na and heparin. With 1.6 g/L EDTA-2Na, the measurement value was slightly degraded: the obtained value was 95.0% of the expected value. Heparin up to 400 mg/L showed virtually no interference in calcium measurement: the obtained values were 102.4% of the expected values.
Proteins, especially albumin, must be given due consideration when measuring calcium in serum. We examined interference by albumin using the following method. Human blood plasma albumin (Mitsubishi Chemical Co, Tokyo, Japan) was dissolved to produce various concentrations (0–10 g/dL) using 2.5 mmol/L calcium chloride solution. Their samples were then measured. The estimation of interference by albumin was based on 2.5 mmol/L calcium chloride solution (not including albumin). The oCPC method showed a lower value than the expected value for the sample solutions including 4 g/dL or more albumin. The enzymatic method gently increased for same sample solutions. Even so, our method for calcium measurement up to 8 g/dL showed virtually no interference.
Trueness
Certified serum reference materials (JCCRM 321-3M and JCCRM 321-3H; ReCCS, Kanagawa, Japan) were used for evaluation of trueness. These sera were measured in five replicates. The certified values were 2.39 ± 0.06 and 2.91 ± 0.08 mmol/L, respectively. The biases in these sera were obtained with −0.8 ∼ –.1%.
Reagent stability
We examined reagent stability in the following manner. On the first day, each method was calibrated with each standard solution. We measured control serum using a triplicate assay. Each measurement reagent had been stored in the instrument refrigerator from the first day for 20 d. Furthermore, we measured a control serum, exactly as on the first day, but without calibration. As presented in Figure 5, the determination values declined to about 40% in alkaline measurement media using the oCPC method. The reagents used for this study showed good stability for 20 d, similar to results obtained using the enzymatic method.
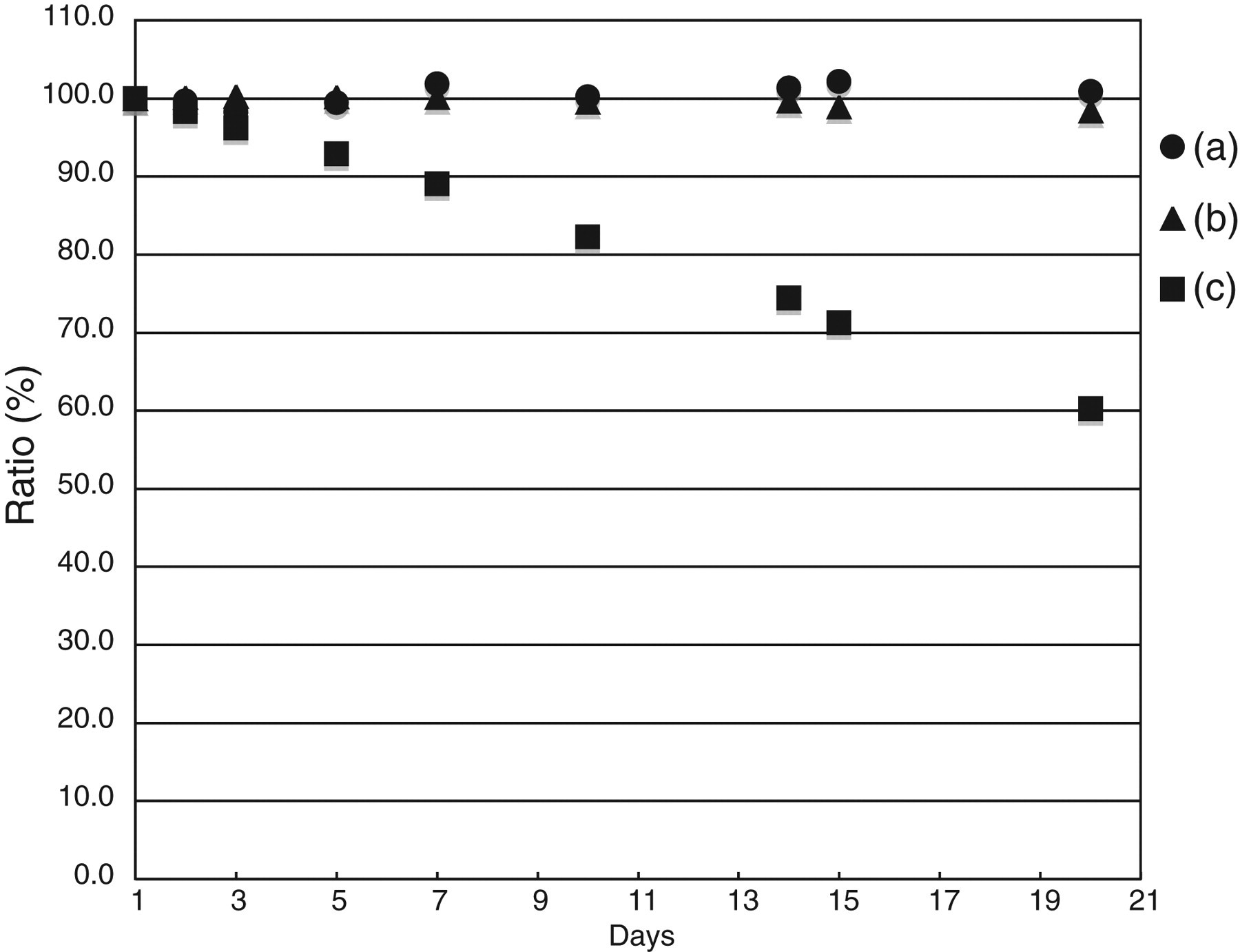
Reagent stabilityThe measurement value for control serum of the first day when calibrated using each method is treated as the reference value. Evaluation was according to the following formula: Ratio (%) = (measurement value of each day/reference value) × 100(a) Present method, (b) enzymatic method and (c) o-cresolphthalein complexone method
Correlation with AAS
We examined correlation between the present method (y) and AAS (x) using serum specimens (n = 50) collected from inpatients and outpatients of Kyushu University Hospital. We adopted the Deming method for regression analysis; we performed it using Altman–Bland difference plots. The results are presented in Figures 6a and b. Results showed good correlation with the present method.
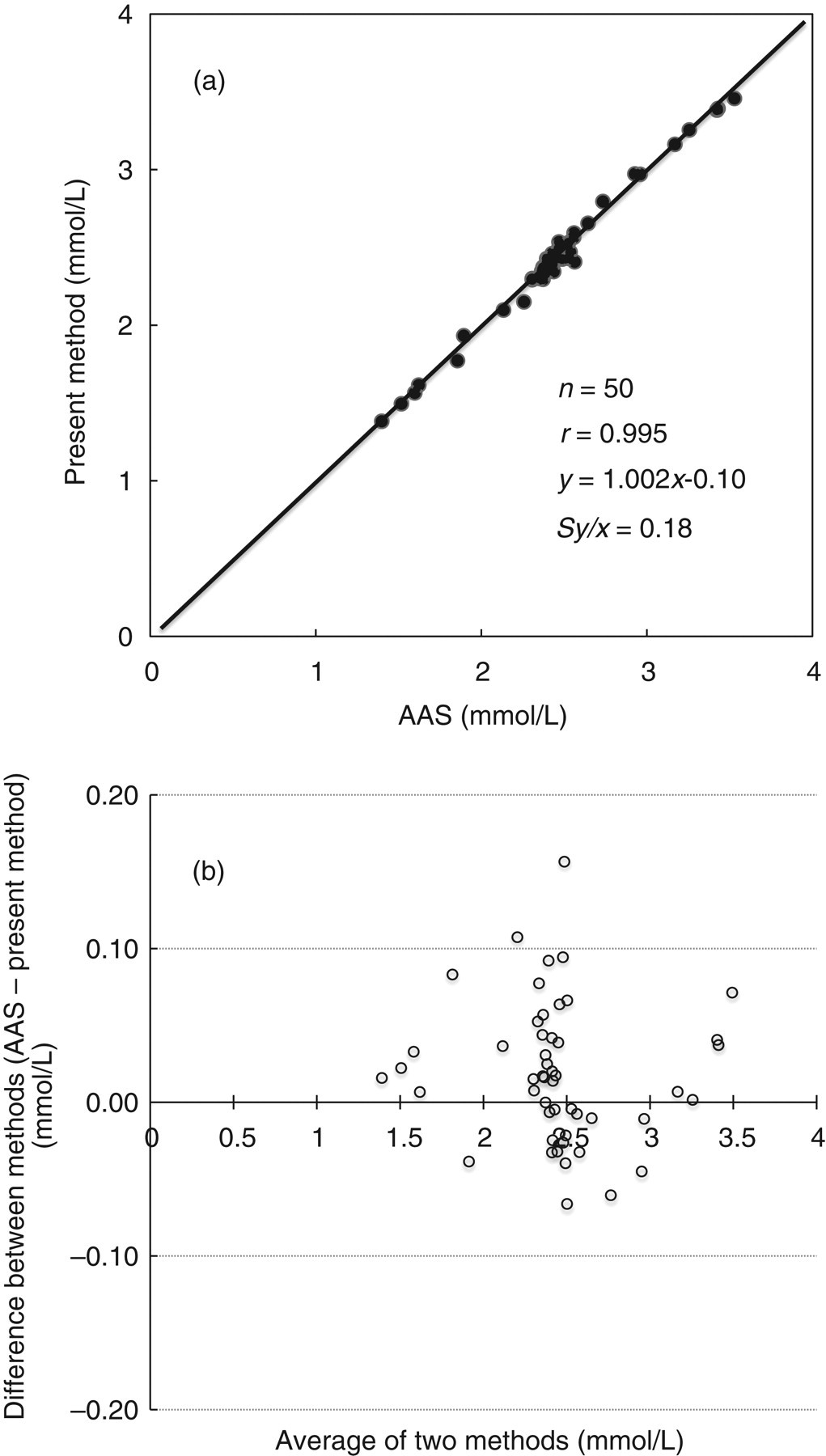
(a) Correlation between the present method (y) and atomic absorption spectrophotometry (x) in serum samples. (b) Altman–Bland difference plots for serum samples
Discussion
Kessler and Wolfman 4 improved the conventional, oCPC method in 1964, and it since became widely used for assaying serum calcium. Nevertheless, the oCPC and MXB methods provide unsatisfactory stability in alkaline media. Among various measurement reagents with chelators, the method using AZ-III chelator produces good accuracy and stability because of its neutral medium; moreover, its tests are inexpensive. However, this method includes arsenic in its processes and therefore remains questionable in relation to its effect on the environment. In recent years, enzymatic methods using pancreatic α-amylase 9 or phospholipase D (EC 3.1.4.4) 10 have been developed and are commercially available. As for accuracy and stability, the enzymatic method is superior to other methods using chelators. Kayamori and Katayama 9 reported that the enzymatic method is highly sensitive and accurate, yielding results that show a good correlation with those obtained using AAS. Notwithstanding, enzymatic testing is costly. Therefore, through this study we sought to develop a new measurement reagent with the good accuracy and stability of the enzymatic method, but obviating the enzyme, instead using CPZ-III as the chelator to provide a low cost per test.
Ferguson et al. 14 first reported a measurement method using CPZ-III in 1964. This CPZ-III has the properties of combination for actinide, calcium, magnesium, etc., in the ratio of 1:1–1:3. Actually, in our buffer solution of pH 5.0 used this time, CPZ-III was combined with calcium in the ratio of 1:1 (dates not shown). The reaction solution, after combination, changes to purple or violet, as does a solution of AZ-III. This chelator has no strong agent or poison in its composition, in contrast to AZ-III. Ferguson indicated that this chelator can measure calcium and magnesium according to differences in pH of the medium.
As they reported, this chelator is useful to measure calcium and magnesium in a neutral medium. Nevertheless, it reacts uniquely to calcium in an acid medium (pH 2.2); the molar absorptivity of the CPZ-III complex with calcium at pH 2.2 is 14,600/L/mol/cm. In our reagent (pH 5.0, 700 nm), the molar absorptivity was 16,300/L/mol/cm. The molar absorptivity of the CPZ-III complex with magnesium at neutral solution is about 15,000/L/mol/cm. However, at a pH of less than 6.0, magnesium reacts very little with CPZ-III. Therefore, we examined the buffer pH, which was useful to measure calcium specifically in a more acid medium if the buffer is not muddied by proteins. The buffer pH was determined as 5.0. As presented in Figure 2, absorption of CPZ-III complexes with calcium was very strong at wavelengths less than 650 nm. Therefore, the reagent composition was determined while considering a sufficient measurement range. As described earlier, the reproducibility, precision and recovery test of our method were good. Furthermore, because the linearity extends to 7.0 mmol/L, we presume that urine specimens can be assayed without dilution. In experiments to assess interference by substances, we demonstrated that our presented method was not affected easily by metals (Mg2+, Fe2+, Cu2+ and Zn2+) and in vivo interfering substances reviewed this time. For anticoagulant EDTA, our presented method was slightly decreased. However, EDTA concentration used for blood sampling was usual, 1.0 g/L. Actually, in the case of 1.2 g/L EDTA, our presented method was merely reduced by 1.8% for expected value. Therefore, we estimated that most influences were absent for anticoagulant EDTA. However,in the oCPC method, it cannot be measured at all. It was presumed that because the reagent used reacts in an acid, the chelate action itself weakens in a medium for calcium of EDTA. Moreover, in the reagent stability test, our reagent using CPZ-III was stable for 20 d at 4°C without daily calibration. On the other hand, the oCPC method decreased the control serum value dramatically. We inferred that the buffer pH was altered because the alkaline medium reagent with the oCPC method absorbed atmospheric bicarbonate.
In conclusion, this new stable and accurate method obviates the use of heavy metals, dangerous materials and mutagenic substances. It is suitable for routine clinical use in the laboratory.
