Abstract
We describe a case where extensive investigations were utilized to identify the aetiology of hypophosphataemia in a patient presenting with non-specific symptoms. The diagnosis of hyperventilation-induced hypophosphataemia was eventually established. Hyperventilation is a relatively common cause of isolated hypophosphataemia, but is easily overlooked as a cause.
Case report
A 28-year-old Caucasian male was referred to our clinic by his General Practitioner with a history of general malaise, anxiety and palpitations. There was no other medical history of note and physical examination was normal. He had hypophosphataemia (0.57 mmol/L; 0.8–1.45 mmol/L), with a corresponding normal serum-adjusted calcium concentration. Significant hypophosphataemia was confirmed on three further occasions before attendance at clinic in November 2005. Other baseline laboratory investigations including parathyroid hormone were normal, although his serum 25-hydroxy Vitamin D (25(OH)D) (15.5 ng/mL; >20 ng/mL), indicated insufficiency. Our initial assessment in clinic was that of symptomatic hypophosphataemia. We organized a series of investigations to ascertain the cause.
Gastrointestinal investigations included normal IgA anti-tissue transglutaminse antibodies, faecal elastase, duodenal biopsy and small bowel barium studies. Renal investigations included normal urinary amino acids and 24-h excretion of phosphate (14.4 mmol/24 h; 12.9–42 mmol/24 h). Tubular maximal resorption of phosphate/glomerular filtration rate (TmP/GFR) is a well-established method for assessment of phosphaturia. 1 This indicated no excess urinary phosphate loss (0.91 mmol/L; 0.8–1.35 mmol/L).
We considered a possible diagnosis of oncogenic osteomalacia, a rare condition presenting with musculoskeletal pain and weakness associated with mesenchymal skeletal tumours. These tumours are thought to release phosphaturic hormones, in particular fibroblast growth factor-23 (FGF-23) resulting in a low TmP/GFR. A normal TmP/GFR in the context of an FGF-23 of 20 U/L (<100 U/L) and a negative indium-labelled octreotide scan effectively excluded this possibility.
Oral Vitamin D2 supplementation was started empirically in the absence of a definitive diagnosis. The 25(OH)D concentration increased to 35.6 ng/mL following this.
The patient was subsequently admitted in an emergency in November 2006 with carpopedal spasm, palpitations and pre-syncope. He had severe hypophosphataemia (0.23 mmol/L). He was not overtly tachypnoeic but arterial blood-gas analysis on breathing room air demonstrated a respiratory alkalosis; pH 7.53 (7.35–7.45), pCO2 3.6 kPa (4.7–6.0), pO2 16.9 kPa (11.1–14.4), actual bicarbonate 23 mmol/L (22–27). An echocardiogram and a 24-Holter (electrocardiogram) monitor were normal. Oral phosphate replacement was commenced with serum phosphate concentrations of 0.65 mmol/L 24 h after admission and 1.30 mmol/L four days later.
Despite continuing to take oral phosphate, a similar presentation in December 2006 precipitated hospital admission. The serum phosphate was 0.24 mmol/L and 48 h later was 0.88 mmol/L despite no specific intervention.
We considered the possibility of hyperventilation-induced hypophosphataemia. This was confirmed with a controlled hyperventilation study. Serum phosphate and ear-lobe capillary blood gases were measured before and after hyperventilation and demonstrated the association between hyperventilation-induced respiratory alkalosis and hypophosphataemia.
Discussion
Hypophosphataemia (<0.8 mmol/L) is common in hospitalized patients, but severe hypophosphataemia (<0.3 mmol/L) is less frequently seen 2 and may have significant adverse consequences. The cause of hypophosphataemia is usually multifactorial, but the main mechanisms are cellular redistribution, renal loss and malabsorption or gastrointestinal loss. Hyperventilation causes a respiratory alkalosis, which in turn mediates an increase in intracellular pH. This stimulates phosphofructokinase activity in the glycolytic pathway with a subsequent increase in demand for phosphate, which is driven intracellularly to form glycolytic intermediate metabolites. 3,4 A similar phenomenon is observed with an increase in intracellular pH, which occurs in metabolic alkalosis. 4
In our patient the hypophosphataemia seemed persistent and the cause elusive. However, hospital assessment demonstrated rapid equilibration of phosphate concentrations indicating cellular redistribution. Hyperventilation is a common cause of hypophosphataemia in hospitalized patients 5 and may not be obvious. We believe that hyperventilation occurred episodically in our patient in association with anxiety. In this case we believe that hypophosphataemia is aetiologically unrelated to his symptoms. Following careful counselling, further laboratory tests have shown no evidence of hypophosphataemia and his symptoms are resolving. Hyperventilation-induced respiratory alkalosis is an easily overlooked cause of hypophosphataemia and if identified will prevent unnecessary investigation.
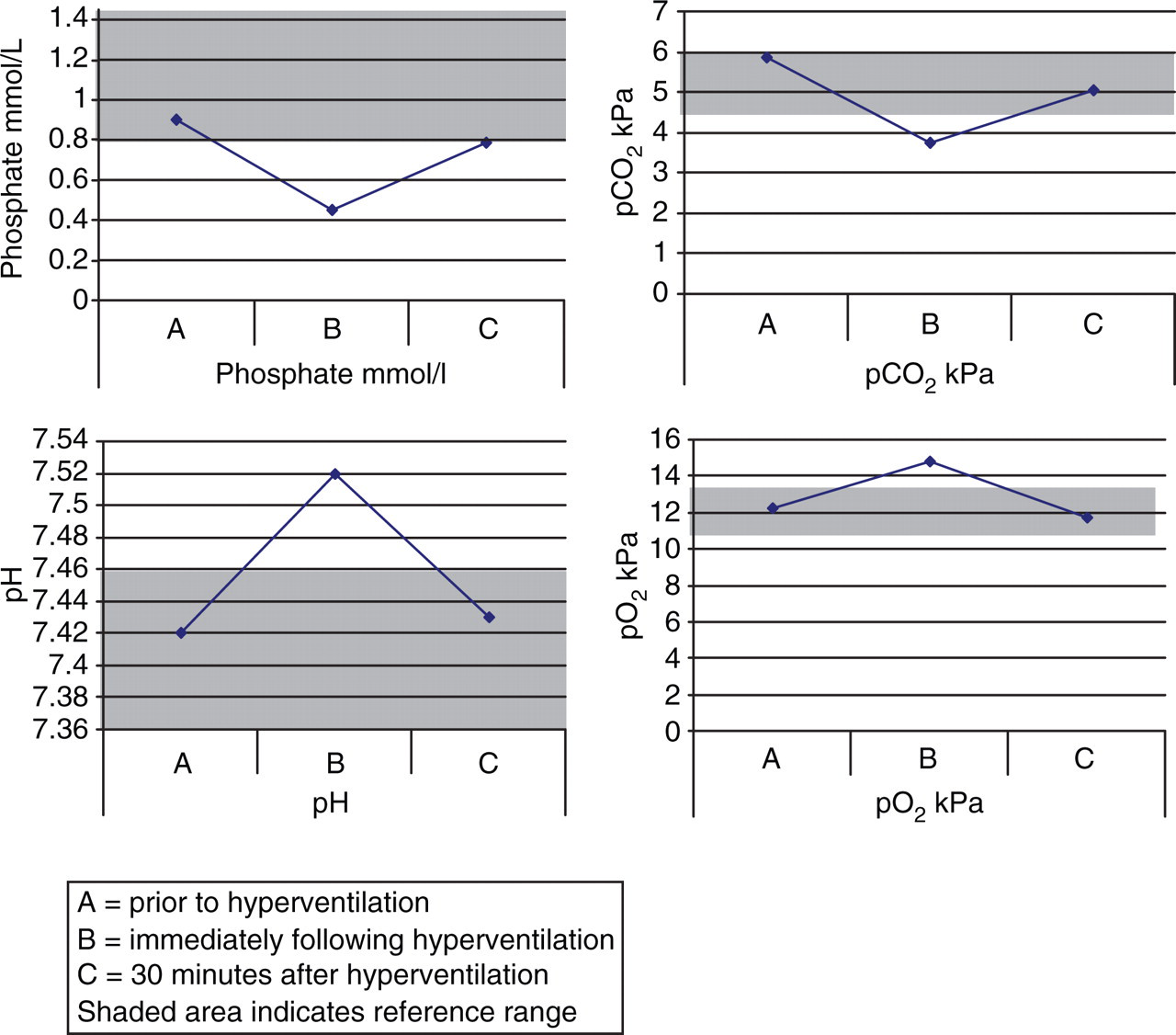
Biochemical parameters during hyperventilation study
