Abstract
Background
The measurement of catecholamines and metanephrines in urine is an important diagnostic test in biochemical screening for phaeochromocytoma. Tandem mass spectrometry (MSMS) has the potential to be used in a profiling method for simultaneous assay of these analytes.
Methods
Optimal conditions were established for the MSMS detection of catecholamines (noradrenalin, adrenalin and dopamine) and metanephrines (normetanephrine and metanephrine), including commercially available isotopically labelled compounds for use as internal standards. Chromatographic separation of all five polar biogenic amines was achieved under solvent conditions that were compatible with MSMS and multiple reaction monitoring. Several types of solid-phase extraction cartridge were used to investigate clean-up conditions for urine, and acid-hydrolysates of urine, prior to LC-MSMS.
Results
Total catecholamines and metanephrines from acid-hydrolysed urines, or free catecholamines and free metanephrines from native urines, were complexed with diphenyl-boronate and recovered in high yield from polymer cartridges after elution with formic acid. Direct injection of eluates into the LC-MSMS system allowed quantitation of catecholamines and metanephrines with a run time of 6 min per sample. Biogenic amine concentrations for patient urines and quality assurance programme samples, and assay imprecision, were similar to values obtained with high-performance liquid chromatography methods, which used electrochemical detection. In normal urines, the ratio of free to total catecholamines was around three-fold higher than the ratio of free to total metanephrines.
Conclusion
The assay of urinary catecholamines and metanephrines can be achieved simultaneously using one LC-MSMS method, which is rapid and reduces labour and consumable costs for routine application.
Introduction
Urinary biogenic amine excretion is used to diagnose phaeochromocytoma as a rare cause of unexplained hypertension. 1 The most commonly performed test for this purpose in clinical chemistry laboratories is the measurement of urinary free catecholamines, which include adrenalin, noradrenalin and also dopamine. This test is easier to carry out than for the methylated metabolites metanephrine and normetanephrine, since it does not require hydrolysis of conjugates formed during metabolism and excretion (sulphates and glucuronides). However, this advantage is offset by evidence that the clinical sensitivity of total urine metanephrine excretion is higher than that of urine-free catecholamines. 2 In Australia, the Medicare benefit schedule regards both tests to be equivalent and funds one test or the other for individual pathology testing, but not both.
Although early methods for measurement of biogenic amines were based on colorimetry, nearly all laboratories now use high-pressure liquid chromatography coupled to electrochemical detection (ECD) at specific voltages to increase analytical specificity. 1 Prior to chromatography, sample clean-up is commonly performed using one or two solid-phase extraction (SPE) columns to remove potentially interfering substances. With the increasing availability of bench-top tandem mass spectrometers in the clinical laboratory, it is possible that these detectors could substitute for ECD as highly specific and robust analytical instruments for urinary catecholamines 3 and metanephrines. 4 In this study, a single SPE procedure has been developed for free catecholamines and metanephrines. Biogenic amines are eluted with formic acid and separated on the same chromatography column for direct analysis by tandem mass spectrometry in multiple reaction monitoring (MRM) mode with isotopically labelled internal standards.
Materials and methods
Materials
The biogenic amines adrenalin, noradrenalin, dopamine,
SPE Oasis cartridges (1 mL – 30 mg) were manufactured by Waters Corporation (Milford, MA, USA) while Bond Elut cartridges and the Vac-Elut SPS-24 sample preparation manifold were obtained from Varian Inc. (Palo Alto, CA, USA). Solvents such as methanol and formic acid were of high purity grade designated for high-performance liquid chromatography (HPLC) and mass spectrometry.
Instrumentation
The HPLC system consisted of an Agilent 1200 quaternary pump with degasser, high-performance autosampler and thermostatted column compartment (Agilent Technologies Australia, Forest Hill, VIC). Analytical columns were purchased from Waters Corporation or Phenomenex (Lane Cove, NSW). The eluate from the HPLC system was connected to an AB 3200 Q-Trap tandem mass spectrometer (Applied Biosystems, Scoresby, VIC) with an electrospray ion source operated in positive mode and controlled by Analyst version 1.4.2 software. The capillary voltage was 5 kV, source temperature 400°C, with curtain gas, GS1 and GS2 nitrogen flows all set at 30 mL/min. The quadrupoles Q1 and Q3 were operated at unit resolution. Declustering potentials, collision cell entry and exit potentials, and collision energies were tuned individually for each biogenic amine, and programmed into a MRM method with a dwell time of 100 ms per transition. Quantitation was by peak area ratios of ion chromatograms with a 1/x weighting of linear least squares regression of calibration data.
For method comparison studies, an Agilent 1100 HPLC system with Dionex ED 50 electrochemical detector was used. Prior to HPLC, urine samples were prepared using kit methods from BioRad Laboratories (Gladesville, NSW), which involved sample clean-up on one (catecholamines) or two (metanephrines) ion-exchange columns, according to the manufacturer's instructions. The purified free catecholamines were analysed on a C18 column with ion-pairing agent in the mobile phase, 5 while total metanephrines were separated on a 150 × 2.1 mm Synergi Hydro column (Phenomenex Australia, Lane Cove, NSW) with 20 mmol/L phosphate buffer, pH 3.0 as mobile phase, and quantitated by peak area ratios using Chemstation software (Agilent Technologies, Forest Hill, VIC).
SPE and recovery studies
Urine and 50 mmol/L formic acid were spiked with pure biogenic amines from 1 mmol/L stock solutions prepared in 10 mmol/L HCl to give added concentrations of 0, 0.1, 0.5, 2 and 5 μmol/L. To 0.5 mL of each urine, or 50 mmol/L formic acid as control, was added 1 nmol of each labelled internal standard. Urine samples were subjected to SPE using a Vac-Elut SPS 24 manifold with vacuum pump to control flow rates. After washing, biogenic amines were eluted in 1 mL of 200 mmol/L formic acid for quantitation by direct injection of 20 μL into the LC-MSMS system. Absolute recoveries from SPE were calculated from internal standard peak areas from urines subjected to SPE compared with formic acid solutions that were analysed directly without SPE. These recoveries accounted for losses on the SPE column and any ion suppression effects from the urine matrix. Spike or relative recoveries were calculated by measuring the concentrations of biogenic amines in each urine from their peak areas expressed as a ratio to the peak area of the internal standard. Endogenous urine concentrations were subtracted and the net concentrations were divided by the added concentrations to give percentage recoveries.
Calibrators and quality controls
Commercially available urine calibrator (Cat No. 1956021) for biogenic amines (BioRad Laboratories Pty Ltd) was re-constituted in 0.1 mol/L HCl and stored frozen. A 4-point calibration curve was prepared with 0, 0.1, 0.3 and 0.5 mL of calibrator and 0.1 mol/L HCl to keep the volume constant at 0.5 mL. Calibration curves were linear over the range of concentrations in the calibration series for all five biogenic amines.
BioRad Lyphochek internal quality control materials were purchased for use at low and high concentrations.
Patient urines
Method comparison studies were carried out using acidified 24-h urine collections collected from 29 patients undergoing routine biochemical screening for phaeochromocytoma and from 13 patient urines from confirmed phaeochromocytoma cases and distributed as part of the biogenic amines external quality assurance programme of the Royal College of Pathologists of Australasia Pty Ltd (Sydney, Australia).
Prior to LC-MSMS, acid hydrolysis of urine was carried out in glass tubes in a Dri-Block heater using 0.3 mol/L HCl at 90°C for 25 min, as described by Crockett et al. 6 Labelled internal standards were added to the cooled hydrolysates to prevent decomposition of the deuterated compounds during the hydrolysis. 6
Statistical analysis
All statistical analyses were performed using Analyse-It software version 2.09 (Analyse-It Software Ltd, Leeds, UK).
Results
Mass spectrometer tuning
Voltages and gas flows for optimal detection of the biogenic amines were determined after infusion of pure solutions (10 μmol/L in 50 mmol/L formic acid) of noradrenalin, adrenalin, dopamine, normetanephrine and metanephrine into the electrospray source of the tandem mass spectrometer. Positively charged precursor ions were detected at the first quadrupole (Q1) with m/z values as reported by others 3,4 and shown in Table 1. The five isotopically labelled internal standards were also tuned in the same way, with the expected mass increases due to the number of isotopic atoms present. Precursor ions passed through a collision cell of variable energy to produce specific ion fragments, which were detected at the third quadrupole (Q3) and allowed MRM of ion pairs specific to each biogenic amine. The exception was the transition 184→166, which was common to adrenalin and normetanephrine, which are isobaric. However, the molecular ion for normetanephrine is unstable in the ion source and loses water to produce a predominant ion of m/z 166. 4 The fragmentation of this ion to m/z 134 was used as the ion transition pair for MRM of normetanephrine, whereas 184→166 was used for adrenalin, with chromatographic separation used to enable their separate quantitation.
Mass to charge ratios (m/z) of precursor and product ions found at Q1 and Q3 after constant infusion of pure solutions of catecholamines and metanephrines into the electrospray source at 5000 V and various collision cell energies
Smaller product ions (in brackets) were less intense, and were not used for quantitation
Chromatography conditions
A number of HPLC columns were investigated for their ability to separate catecholamines and metanephrines. An important consideration was the composition of the mobile phase, which needed to be compatible with mass spectrometric detection. Ion-pair reagents, which are commonly used with reversed-phase HPLC of catecholamines, 5 were avoided to prevent contamination of the mass spectrometer. With 50 mmol/L formic acid of pH 2.5, containing 2% (v/v) methanol as mobile phase, baseline separation of a standard mixture of the five biogenic amines was obtained on C18 phases modified for polar compounds (Atlantis T3 [Waters] or Synergii Hydro [Phenomenex] columns), as shown in Figure 1. Commercially available isotopically labelled compounds for use as internal standards were also chromatographed to demonstrate their purity. They eluted as symmetrical peaks with retention times similar to, but slightly earlier than, their respective unlabelled compounds. The labelled and unlabelled normetanephrine, and to a lesser extent adrenalin, in Figure 1 appear as double peaks because they share the same ion transitions, as already discussed. Hydrophilic interaction liquid chromatography (HILIC) has been used recently for LC-MSMS of polar compounds, and a method for plasma metanephrines has been published, 7 but this method relies on the mass spectrometer rather than HPLC to separate the compounds of interest.
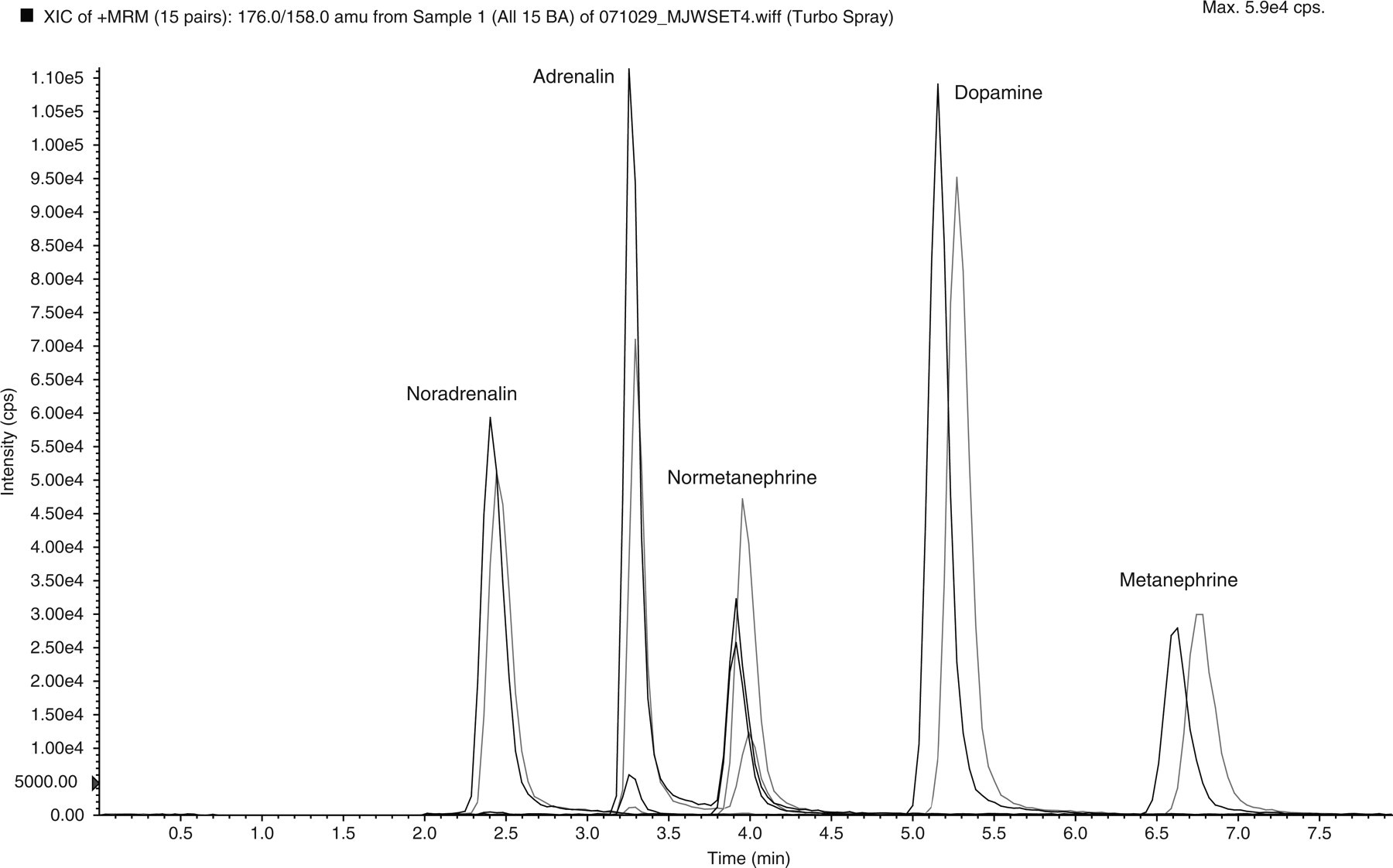
Separation of catecholamines and metanephrines, and their internal standards (50 pmol of each amine), on an Atlantis T3 column (150 × 2.1 mm) using 0.05 mol/L formic acid containing 2% methanol, pH 2.5 at a flow rate of 0.2 mL/min and multiple reaction monitoring of ion pairs for detection. The most intense ion transition for each peak are listed in Table 1. Labelled internal standards (black) eluted slightly earlier than their corresponding catecholamine or metanephrine peaks (grey)
SPE of biogenic amines
SPE was investigated as a potential step for clean-up of urine samples prior to chromatography and tandem mass spectrometry to minimize ion suppression. Using urine spiked with different concentrations of catecholamines and metanephrines, plus a mixture of the five isotopically labelled biogenic amines, a series of recovery experiments were performed using five different types of SPE cartridge. The results are summarized in Table 2.
Mean (±SD) of absolute and relative recoveries of biogenic amines from five different solid-phase extraction (SPE) cartridges
A trend with all SPE phases was that the highest absolute recoveries were obtained for dopamine and metanephrine, while those for noradrenalin and normetanephrine were lower. However, recoveries for all biogenic amines were close to 100% relative to their respective internal standards for all types of SPE cartridge, even when the absolute recoveries were low. The solvent and elution conditions varied between SPE cartridges, which were operated with varying pH buffers under cation-exchange (Oasis WCX and MCX), covalent-binding (Bond Elut PBA) or mixed mode (Oasis HLB and Bond Elut Plexa) principles. Important objectives were to maintain acidic conditions for stability of catecholamines, compatibility of eluting solvents with the HPLC mobile phase (which meant avoiding organic solvents such as methanol or acetonitrile) and the ability for direct injection without the need for solvent evaporation steps. With these points in mind, the final method used Bond Elut Plexa in 1 mL cartridge format for urine clean-up. This phase does not retain the polar biogenic amines effectively unless they are first complexed in solution under alkaline conditions with diphenyl-boronate, as described for urinary catecholamines by Talwar et al. 8 The release of the catecholamines and metanephrines from the Plexa cartridge with formic acid after a wash step allowed direct injection into the LC-MSMS system for separation and quantitation.
Final method for LC-MSMS of urine biogenic amines
The sample preparation and extraction conditions were based on those described by Talwar et al. 8 To 0.5 mL of sample (urine, acid-hydrolysed urine, calibrator or QC) was added internal standard (40 μL of a mixture of five isotopically labelled biogenic amines, each at 25 μmol/L) and 0.8 mL of complexing agent. After vortex-mixing, Bond Elut Plexa columns (1 mL, 30 mg sorbent) were placed on a Vac Elut SPS24 extraction system, activated with 1 mL methanol and conditioned with 1 mL 0.2 mol/L ammonium buffer at pH 8.5. 8 The complexed samples were applied under gravity to the SPE columns, which were then washed with 1 mL of 0.2 mol/L ammonium buffer containing 5% methanol and dried for 30 s at full pressure (20 in. Hg). Biogenic amines were eluted with 1 mL of 0.2 mol/L formic acid before transferring an aliquot to a glass insert in an autosampler vial for tandem mass spectrometry. Samples (20 μL) were injected onto an Atlantis T3 column (150 × 2.1 mm) at 25°C with a flow rate of mobile phase of 300 μL/min.
To illustrate this method, ion chromatograms obtained using MRM for hydrolysed urine from patients without and with phaeochromocytoma are shown in Figure 2a and b, respectively. Relative to the internal standards, there is a large increase in total noradrenalin concentration from 0.15 to 1.48 μmol/L, adrenalin from 0.05 to 1.44 μmol/L, normetanephrine from 0.40 to 3.17 μmol/L and metanephrine from 0.22 to 6.24 μmol/L. The dopamine concentration, however, is similar at 0.52 and 0.58 μmol/L in both urine samples, as indicated by the same size of peak 4 in both chromatograms.
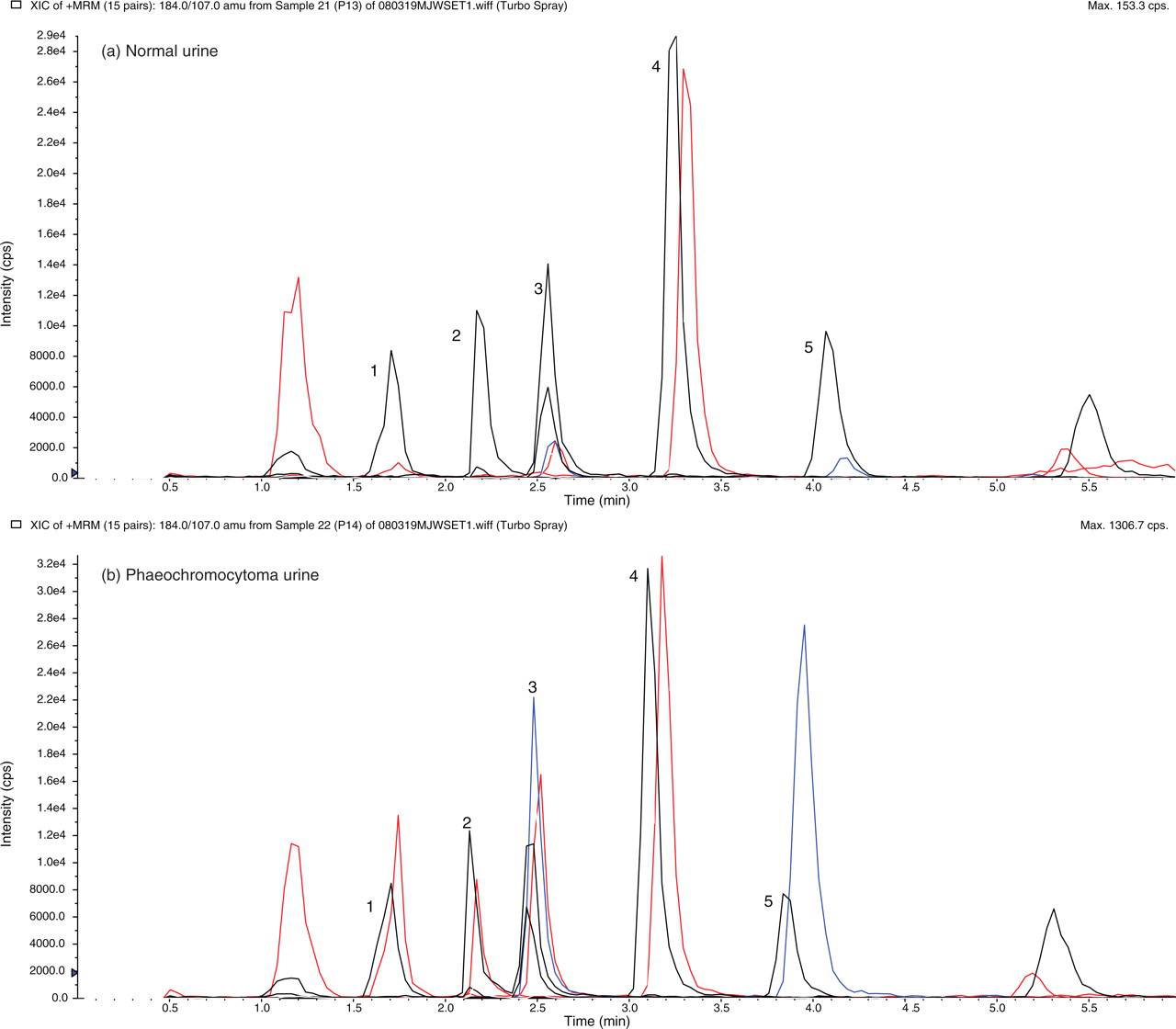
Multiple reaction monitoring (MRM) of biogenic amines recovered from acid-hydrolysed normal urine (a) and urine from a patient with phaeochromocytoma (b). The peaks are (1) noradrenalin, (2) adrenalin, (3) normetanephrine, (4) dopamine and (5) metanephrine. Internal standard peaks eluted slightly earlier than the peaks for endogenous urine catecholamines and metanephrines. The ion pairs used for quantitation of each of the MRM transitions are listed in Table 1
Similar ion chromatograms to those shown in Figure 2 are obtained if non-hydrolysed urine is extracted by the same SPE procedure to analyse free urinary biogenic amines, except that the concentrations are lower. In a subgroup of 13 urines from patients without phaeochromocytoma, the mean (±SD) ratios of free to total noradrenalin and adrenalin were found to be 0.37 ± 0.12 and 0.49 ± 0.20, respectively. For the same urines, these ratios were significantly higher (P < 0.001) than the mean ratios of free to total normetanephrine and metanephrine of 0.13 ± 0.05 and 0.19 ± 0.06, respectively. Thus, the ratio of free to total catecholamines in normal urines was around three-fold higher than the ratio of their free to total metanephrine metabolites.
Imprecision and accuracy
The same lot number of commercial internal quality control materials at two concentrations were analysed in repeated runs over a five-month period. Free catecholamines were determined without a sample hydrolysis step, whereas total metanephrine concentrations were measured after acid hydrolysis. The mean concentrations and their coefficients of variation (CV %) are shown in Table 3. The CV were consistently around 5–7% at low and high concentrations of biogenic amines.
Inter-run imprecision over five months in the measurement of free catecholamines and total metanephrines for commercial QC material (BioRad Lyphochek) at two concentrations
External quality assurance programme samples (n = 18) from RCPA QAP Pty Ltd were analysed by the LC-MSMS method over one six-monthly cycle and agreed well with target values set by the programme coordinator. The recovery studies summarized in Table 2 for urine samples spiked with different concentrations of pure catecholamines and metanephrines also illustrated that the method was accurate.
Limits of detection and linearity
The lower limits of detection, defined as the concentration of analyte that resulted in a signal to noise ratio of 5, were 0.01, 0.01 and 0.005 μmol/L for the three catecholamines – noradrenalin, adrenalin and dopamine, respectively, and 0.005 μmol/L for the two metanephrines. These detection limits were well below concentrations found in normal urines of all five biogenic amines, except for adrenalin which occasionally was reported as <0.01 μmol/L.
The commercial calibrator with urine matrix gave a linear detector response up to the stated concentrations of biogenic amines, which were 1.2, 0.6, 2.0, 4.2 and 2.0 μmol/L for noradrenalin, adrenalin, dopamine, normetanephrine and metanephrine, respectively. These concentrations cover those expected in biochemical screening of most urines. However, the mass detector had a much higher linear dynamic range, as determined from the assay of pure compounds up to 20 μmol/L and from the dilution of selected urines from patients with phaeochromocytoma with concentrations of biogenic amines >50 μmol/L.
Method comparison studies
The results of the comparison of the new LC-MSMS assay with our previous HPLC assay with ECD are shown for free catecholamines in Figure 3 and total metanephrines in Figure 4. Urines (n = 13) collected in acid over 24 h from patients with confirmed phaeochromocytoma, and distributed as part of an external quality assurance programme, were analysed along with 29 urines from patients who were being screened for phaeochromocytoma. Results were calculated as excretion values in μmol/d and are presented as Bland-Altman plots. The LC-MSMS assay gave similar results to HPLC with ECD, with the mean percent biases being −2.6% for noradrenalin, 1.1% for adrenalin and 4.9% for dopamine. For adrenalin, analytical interference prevented a result being calculated for two urines using HPLC-ECD, but this was not a problem with the highly specific LC-MSMS method. Regression equations by Passing-Bablok were LC-MSMS = 1.07 (HPLC-ECD) −0.01 for noradrenalin, LC-MSMS = 1.20 (HPLC-ECD) −0.00 for adrenalin, and LC-MSMS = 1.03 (HPLC-ECD) +0.02 for dopamine.
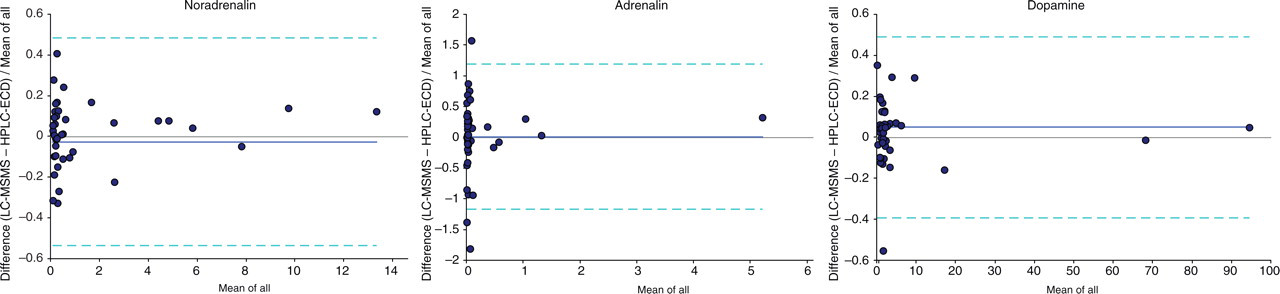
Bland-Altman plots comparing the fractional difference in excretion (μmol/d) of free catecholamines noradrenalin, adrenalin and dopamine in 13 phaeochromocytoma urines and 29 normal urines, as measured by liquid chromatography-tandem mass spectrometry and high-performance liquid chromatography with electrochemical detection. Dark lines show the mean bias, while dotted lines give 95% confidence limits
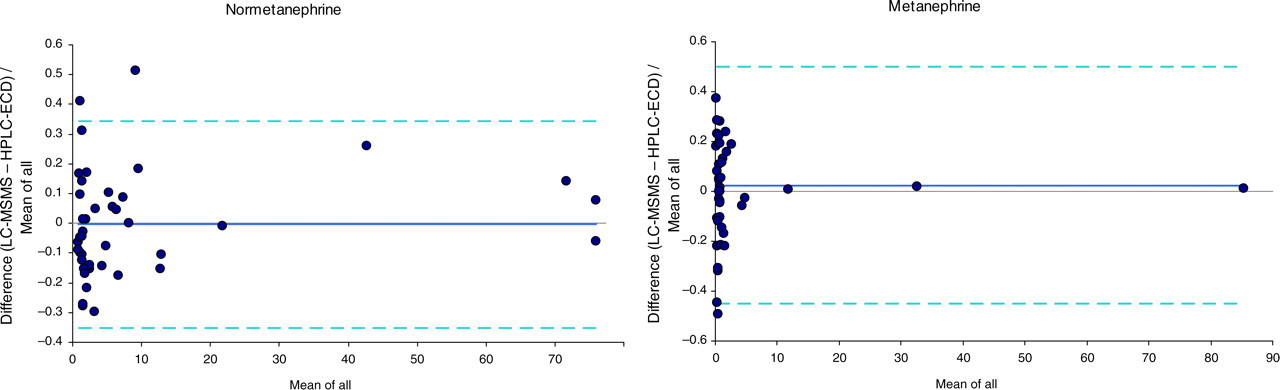
Bland-Altman plots comparing the fractional difference in excretion (µmol/d) of total normetanephrine and metanephrine in 13 phaeochromocytoma urines and 29 normal urines, as measured by LC-MSMS and HPLC with electrochemical detection. Dark lines show the mean bias, while dotted lines give 95% confidence limits
For metanephrine excretions, the mean biases were −0.4% for normetanephrine and 2.5% for metanephrine, while the corresponding regression equations were LC-MSMS = 1.01 (HPLC-ECD) −0.06 for normetanephrine and LC-MSMS = 1.01 (HPLC-ECD) −0.0 for metanephrine. Both methods with different detectors correctly classified all 13 phaeochromocytoma patients as having raised urine excretions of total normetanephrine and/or metanephrine above the reference ranges of <3.0 and <1.3 μmol/d, respectively. One of these phaeochromocytoma patients was found to have normal noradrenalin and adrenalin excretion amounts by both methods, consistent with the lower sensitivity of urinary free catecholamines compared with total metanephrines for the biochemical diagnosis of phaeochromocytoma. 2 Furthermore, using cut-off values of 650 and 350 nmol/d for free urinary normetanephrine and metanephrine outputs, respectively, 9 all patients with phaeochromocytoma were identified as having excessive excretion of normetanephrine with seven also having raised metanephrine excretion values.
Discussion
The LC-MSMS method described in this work has a number of unique features that provide an advance on existing techniques. First, liquid chromatography conditions are described that separate all five biogenic amines of interest, using a simple acidic mobile phase that is mass-spectrometry compatible without containing ion-pairing reagents. Chromatographic separation of compounds was particularly important for adrenalin and normetanephrine, which are isobaric and share some ion transitions used in the MRM mode of detection. Other tandem mass spectrometric methods have been published, but they use different chromatographic conditions for catecholamines 3 and metanephrines. 4,7,10
Second, the present method permits the quantitation of catecholamines and metanephrines simultaneously with a short run time of 6 min or less. In this regard, the high specificity and scan speed of the tandem mass spectrometer are very beneficial for analyte profiling. Previous attempts have been made to develop uniform chromatography conditions for quantitation of biogenic amines using a C18 HPLC column with ion-pairing mobile phase and electrochemical detector set at a fixed potential 11 or a fluorescence detector. 12 These methods however were subject to analytical interferences and required separate extraction techniques for sample preparation. 11 More recently, Davidson 9 described an automated HPLC system for separation of free catecholamines and metanephrines from urine, but reported a high rate of chromatographic interference by drugs and other unknown substances. The very high specificity of tandem mass spectrometry overcomes these interference problems. 3,4
The third significant advantage of the present method is that one SPE cartridge is used to recover both catecholamines and metanephrines in high yield with simple acid elution for direct injection into the LC-MSMS. In developing a suitable SPE step to clean-up urine or urine hydrolysates prior to LC-MSMS, there are several possible phases available with different mechanisms of binding and operation. Initially, weak-cation exchange cartridges were used to extract metanephrines from urine, with similar conditions to those described by De Jong et al. 7 except that they used an on-line format. However, their method recovers metanephrines in acidic acetonitrile, which necessitates an extra evaporation step prior to the chromatography conditions used in this study. The known affinity of catecholamines for solid-phase boric acid, as found in Bond Elut PBA cartridges, 13 and release under acidic conditions, was attractive as a potential selective clean-up step prior to tandem mass spectrometry. However, the procedure of Talwar et al. 8 which converts urinary catecholamines to complexes with diphenyl-boronate in solution at alkaline pH prior to their retention on a C18 SPE sorbent, was adopted. This had the advantage of pH control and stabilization of catecholamines, so that they were recovered in high yield after specific elution with formic acid for direct injection. Unexpectedly, it was found that the metanephrines were also retained under the same conditions, even though they are methylated and do not contain the cis-diol moiety that provides the covalent linkage binding mechanism for catecholamines. Nevertheless, studies of the interaction of catecholamines and related compounds with dihydroxyboryl-silica have shown that metanephrines do have some affinity for this sorbent. 14 Further evidence of the importance of boron was provided by the finding that the recovery of metanephrines from Bond Elut Plexa cartridges was one-third less in the absence of diphenyl-boronate. Since this compound acts as an affinity-pairing agent at pH 8.5 to link catecholamines to the Bond Elut Plexa support, it would seem that it may perform a similar function for metanephrines and allow a one-step SPE clean-up for both catecholamines and metanephrines with simple acid elution.
For convenience, electrospray was chosen as the ionization mode for the aqueous formic acid mobile phase introduced into the tandem mass spectrometer. It has been argued that the presence of high concentrations of organic solvents enhances ion production, and that HILIC chromatography, as used for plasma metanephrines, 7 gives greater sensitivity. The method described in this work was adequate for urine catecholamines and metanephrines, and avoided additional solvent evaporation steps. Furthermore with our instrument, the same LC-MSMS conditions are suitable for plasma-free metanephrines, which can be extracted and concentrated as described by Lagerstedt et al. 10 prior to analysis, so that ion production is adequate even to measure the low concentrations (<1 nmol/L) of free plasma metanephrines. In addition, although some ion suppression effects were apparent in urine recovery studies, for accurate quantitation they are compensated using isotopically labelled internal standards, which are available from commercial sources.
With the advent of methods to measure free plasma metanephrines as well as urine biogenic amines, the choice of best test for biochemical screening for phaeochromocytoma has been debated. 2,15 The present LC-MSMS method is able to profile catecholamines and metanephrines by MRM in the same chromatogram, but for urine specimens separate runs must still be performed to measure the total (with hydrolysis step) or free (without hydrolysis step) analytes. Because of their high concentrations in urine and to avoid problems with instability due to oxidation at high pH, catecholamines are nearly always reported as free noradrenalin and adrenalin excretion after direct urine analysis. In this work, it was found that around one-third of urine noradrenalin and one-half of adrenalin was present in the free form. On the other hand, urinary metanephrines nearly always are reported as total (free plus conjugated) excretions, after acid hydrolysis to breakdown, predominantly sulphate conjugates. 16 Recently however, free fractionated urine metanephrines have been proposed as a useful alternative to total metanephrines 9 and found to be superior in diagnostic accuracy when compared with plasma or urine catecholamines. 17 In the present study, the urines of the 13 phaeochromocytoma patients were correctly classified using excretion values of either total or free metanephrines. If this finding is confirmed in large clinical studies, the present profiling method will give diagnostic information from both free catecholamines and metanephrines in a single analysis. In conclusion, having one rapid LC-MSMS method, which measures both catecholamines and metanephrines simultaneously, simplifies laboratory operation and provides a more cost-effective routine service for these analytes.
Footnotes
Acknowledgements
I would like to thank the laboratory staff in special chemistry and pharmacology for their assistance in the development of this project.
