Abstract
Iron-induced renal phosphate wasting, hypophosphataemia and osteomalacia have previously been reported in a small number of Japanese patients receiving parenteral iron sucrose. We report the case history of a European male who, as a result of regular intravenous iron polymaltose, developed prolonged hypophosphataemia complicated by widespread insufficiency fractures. The pathogenesis of this complication remains unknown however our novel finding of a marked elevation in fibroblast growth factor 23 (FGF23), which normalized after ceasing parenteral iron, suggests an important and previously unreported effect of iron on FGF23 homeostasis.
Introduction
Parenteral iron is commonly used in iron deficiency when patients are unresponsive to or intolerant of oral iron. It is also used, together with erythropoietin, in patients with chronic renal failure and increasingly in the anaemia of cancer and cancer chemotherapy.
In currently available intravenous (i.v.) iron preparations, iron is complexed with a carbohydrate ligand (dextran, sucrose, gluconate or polymaltose) to form a colloidal suspension. High molecular weight (HMW) iron dextran has been associated with anaphylactoid reactions and the non-dextran preparations are reported to have a lower serious acute event rate. 1 Recently, there has been a resurgence of interest in the use of iron infusions with a commentary in a major medical journal suggesting ‘inappropriate underuse’ in patients with chronic disease. 2 Furthermore, it is reported that when HMW dextran iron is excluded, parenteral iron is not associated with an increased risk of adverse events. 3 Although not widely known, i.v. iron sucrose has been associated with hypophosphataemia, renal phosphate wasting and osteomalacia. 4,5
We describe a case of osteomalacia in a patient treated with i.v. iron polymaltose over a 5-yr period. Possible mechanisms and a brief review of the literature are presented.
Case report
In February 2007, a 38-yr-old Caucasian man was reviewed in the Endocrinology clinic with a 3-yr history of severe hypophosphataemia despite phosphate and cholecalciferol supplements. He had incapacitating pain in his feet, lower back and anterior chest wall, which was exacerbated by simple activities such as turning over in bed and walking. He reported a normal diet and did not drink alcohol. Medical history was significant for Crohns disease diagnosed in 1991, and associated with this he had iron deficiency anaemia of 6 years duration. Oral iron supplementation had resulted in an inadequate haemoglobin response, presumably due to chronic mucosal inflammation, and in 2002 parenteral iron was commenced. Haemoglobin improved and regular i.v. iron polymaltose was continued at a dose of 1–2 g three monthly. His other regular medications were azathioprine, calcium carbonate, Phosphate Sandoz™ and monthly cholecalciferol. There was no family history of hypophosphataemia, osteomalacia or rickets. Clinical examination revealed multiple sites of bony tenderness but no skeletal deformities. The remainder of the examination was unremarkable.
Laboratory tests identified a low serum phosphate of 0.4 mmol/L (0.8–1.4 mmol/L), normal concentrations of calcium 2.2 mmol/L (2.2–2.6 mmol/L) and albumin 41 g/L (35–50 g/L), and elevated alkaline phosphatase at 137 U/L (30–120 U/L). 25-Hydroxyvitamin D (25 [OH]D) and parathyroid hormone (PTH) were both within their reference range at 67 nmol/L (50–150 nmol/L) and 4.6 pmol/L (1.6–7.0 pmol/L). Vitamin D was analysed following hexane extraction and reconstitution in 70% aqueous methanol by HPLC-MS/MS and PTH was measured by ELISA (Roche Elecsys 2010). 1,25-Dihydroxyvitamin D (1,25[OH]2D) determined by RIA (Immunodiagnostics Systems Limited, UK) was low at 14 pmol/L (40–150 pmol/L). Calculated urinary tubular reabsorption of phosphate (TRP) was 50% confirming renal phosphate wasting (normal >82%), with no other biochemical evidence of a renal proximal tubular disorder. Serum phosphate concentrations were normal prior to 2004 suggesting an acquired disorder of phosphate handling. A radionuclide bone scan demonstrated multiple discrete areas of increased bony reaction suggesting fractures in multiple ribs, the sacrum and feet (Figure 1). An abnormality in the left femoral head was confirmed to be avascular necrosis on subsequent magnetic resonance imaging (MRI).
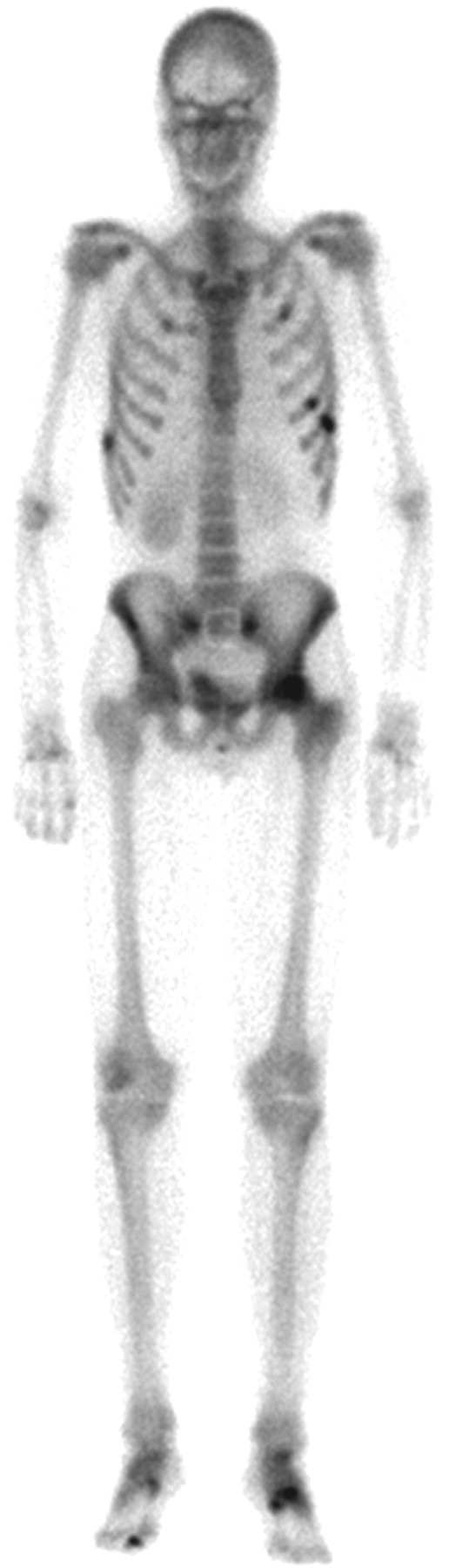
Radionuclide bone scan demonstrates increased bony reaction in ribs, sacrum and feet suggestive of fractures
A diagnosis of tumour-induced osteomalacia was initially suspected and FGF23 requested. Blood was collected into an EDTA tube, immediately centrifuged, separated and frozen at −25°C prior to analysis by a two-site intact ELISA assay (Kainos Laboratories, Tokyo, Japan). CT head and neck, and whole body MRI were normal. While these investigations were occurring, the possible role of parenteral iron was considered and a significant temporal relationship between timing of iron polymaltose infusions and serum phosphate identified (Figure 2). Iron infusions were ceased and his vitamin D preparation was changed from cholecalciferol to calcitriol. Serum phosphate, alkaline phosphatase, 1,25(OH)2D and %TRP normalized over subsequent months and his bone pain and bone scan abnormalities resolved. Subsequently, the FGF23 result became available and was markedly elevated at 480 pg/mL (reference range <71 pg/mL).
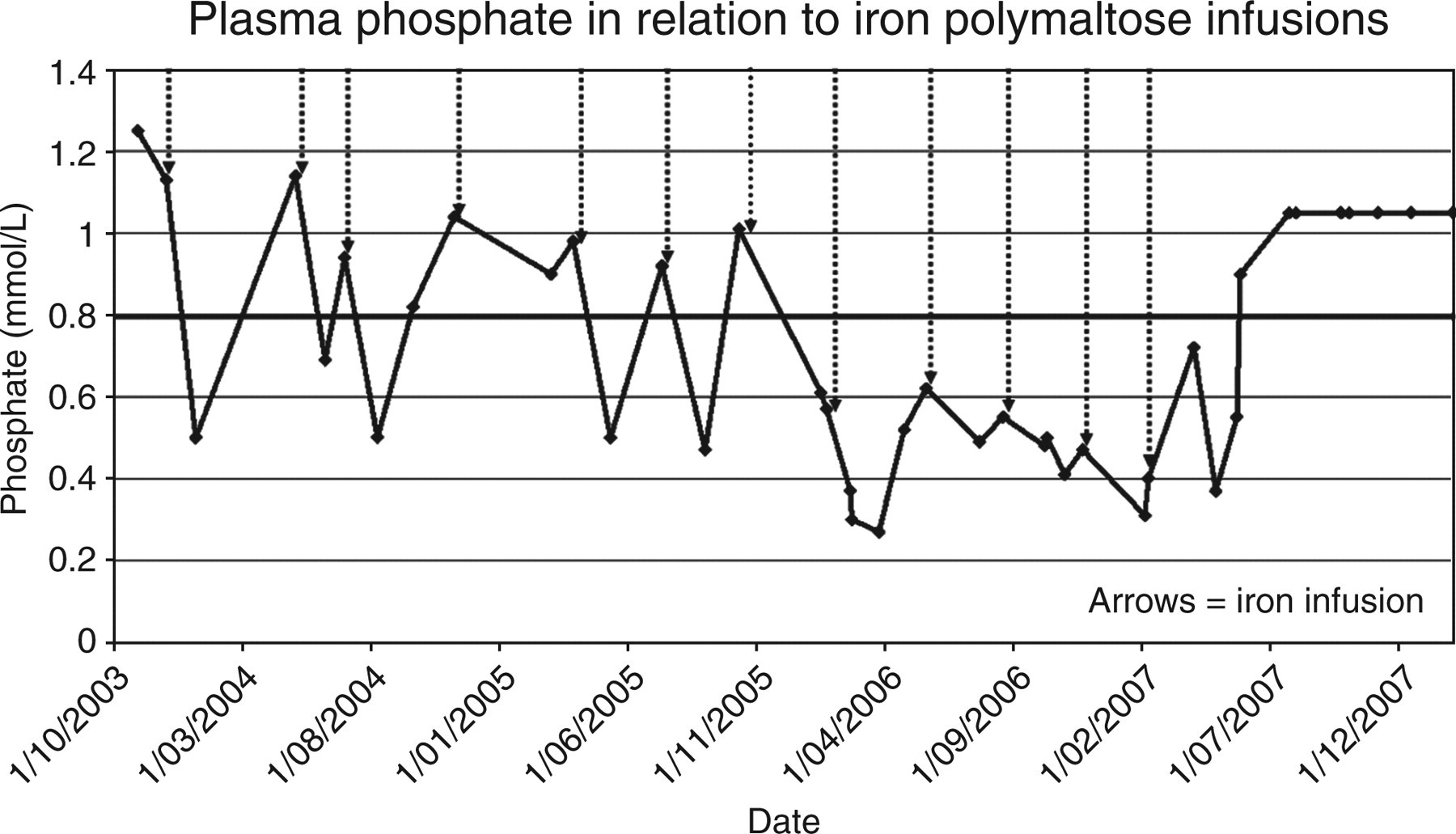
A relationship between the administration of iron polymaltose and a fall in serum phosphate is demonstrated. Phosphate normalized after ceasing iron infusions
Sixteen months after ceasing iron polymaltose and 12 months after ceasing phosphate and vitamin D supplements, FGF23 and phosphate are within the normal range (39 pg/mL and 1 mmol/L, respectively) confirming a diagnosis of iron-induced hypophosphataemia.
Discussion
The kidney is the most important regulator of phosphate homeostasis and under normal physiological conditions %TRP increases, approaching 100%, within hours of hypophosphataemia. 6 In persistent severe hypophosphataemia, bone mineral mobilization usually occurs in an attempt to maintain serum phosphate concentration, with resulting osteomalacia.
Iron-induced hypophosphataemic osteomalacia was first reported in 1982 in the Japanese literature. 7 The following year a prospective Japanese study demonstrated a significant reduction in serum phosphate during treatment with parenteral iron sucrose accompanied by a parallel decrease in %TRP. 8 In addition to renal phosphate loss, studies suggest an inhibitory effect of parenteral iron on renal 25(OH)D 1α-hydroxylase activity with a fall in 1,25(OH)2D levels despite normal 25(OH)D. 4,5 These changes in phosphate handling are reversible, with the rate of recovery related to the duration of parenteral iron therapy. As iron sucrose (molecular weight 43,000 daltons) is filtered by the glomerulus and is detectable in urine, previous authors have hypothesized a direct toxic effect of iron on renal tubular cells. 4,5 Iron polymaltose is not excreted in urine however due to its higher molecular mass (462,000 daltons) arguing against this hypothesis. The dual inhibition of tubular phosphate reabsorption and 1α-hydroxylation of vitamin D suggests that a phosphatonin may play an aetiological role although this has not previously been reported.
The phosphatonin FGF23 is predominantly expressed in bone osteocytes. 9 Increased concentrations of the circulating peptide are central to the pathogenesis of several hypophosphataemic diseases including autosomal-dominant hypophosphataemic rickets, X-linked hypophosphataemic rickets, autosomal recessive hypophosphataemic rickets, tumour-induced osteomalacia and selected cases of McCune-Albright syndrome. 10,11 To our knowledge, this is the first time that FGF23 elevation has been reported in iron-induced osteomalacia. The mechanism by which iron raises FGF23 remains speculative; however may involve upregulation of synthesis or secretion at the level of the osteocyte, or inhibition of proteolytic cleavage and inactivation.
Reviews on parenteral iron claim that this therapy is underutilized and not associated with increased risk. 2,3 However prolonged hypophosphataemia and osteomalacia has now been reported with both iron polymaltose and iron sucrose and may result in significant morbidity due to bone pain and fractures. The prevalence of this complication remains unknown and of note, is not listed on product information sheets. We urge clinicians to consider the potential for this complication when parenteral iron is being prescribed. Discontinuing iron is the mainstay of treatment and 1,25(OH)2D and phosphate supplements are recommended for those patients with severe symptomatic hypophosphataemia.
