Abstract
Background
This study was designed to establish a reference interval for sweat chloride for infants without evidence of cystic fibrosis (CF), aged between 5 wk and 6 wk, a time when sweat testing is an integral part of newborn screening for CF. In addition, we compared the gold standard method of sweat testing (quantitative pilocarpine iontophoresis [QPIT, coulometry]) with an emerging methodology (Macroduct™ [ISE]).
Methods
This was a prospective study on healthy infants at 5–6 wk of age. Sweat collection was undertaken at home on both outer thigh areas using two methods (QPIT and Macroduct™). The order of testing was randomly assigned. Filter paper samples (QPIT) were analysed using flame photometry and coulometry. Macroduct™ samples were analysed using ion-selective electrodes (ISE, Abbott Architect c8000, UK).
Results
Insufficient sweat was collected on 28 occasions with the QPIT (coulometry) method and on 31 with the Macroduct™ (ISE) capillary system. We achieved a 92% success rate in undertaking two sweat collections consecutively (n = 177). Sweat chloride concentrations were normally distributed with excellent limits of agreement between the two methods of sweat collection and analysis (n = 150). Median (IQR) sweat chloride was 11.2 mmol/L (8–13) with QPIT (coulometry) method with a 99.5th centile (n = 165) of 24 mmol/L.
Conclusion
The Macroduct™ (ISE) capillary sweat collection system is valid in this age group. Sweat chloride concentrations above 30 mmol/L should prompt assessment in a specialist CF centre.
Introduction
Cystic fibrosis (CF) is a common inherited disorder, particularly in the Caucasian population 1 and newborn screening (NBS) for the condition has been established in many regions. 2 The CF gene defect results in abnormal transepithelial salt transport, which causes disabled clearance of airway surface liquid with chronic airway infection leading to lung damage and respiratory failure. 1 People with CF have a high sweat salt concentration and the sweat test remains an important diagnostic test, despite characterization of the CF gene defect. 1 DNA analysis is an important component of CF diagnosis, however there are occasions when a CF causing gene defect is not recognized and a sweat test is necessary to confirm or refute the diagnosis. 2–4 In these, often asymptomatic babies, most NBS programmes advocate routine confirmatory sweat testing even after two mutations have been identified. When DNA analysis is inconclusive, sweat testing becomes critical and therefore undertaking sweat testing in infants at 5–6 wk of age is an important component of NBS protocols and one that presents challenges. 5
Pilocarpine iontophoresis, first described by Gibson and Cooke, 6 greatly enhanced sweat collection in children. A number of groups have described the difficulties in collecting an adequate volume of sweat in young infants. 7 A retrospective analysis of sweat tests performed in a large CF referral centre identified prematurity and Afro-Caribbean race as risk factors for not being able to collect adequate amounts of sweat. 8 In addition, this study suggested that there was a fall in mean sweat chloride with age (a chloride concentration of over 20 mmol/L in the first week of life fell to below 15 mmol/L after 42 d of age). 8 This finding was supported by another retrospective review of infants tested after NBS, which also confirmed a significant fall in mean chloride concentrations over the first 8 wk of life. 7 Data from the Wisconsin NBS project demonstrated a mean sweat chloride concentration of 10.6 mmol/L (SD 5.2) in infants without CF who did not have the commonest CF-causing gene defect (phe508del). 9 The mean age of these 184 infants was 9.3 wk (SD 5.3). These data suggest that sweat electrolytes are lower in the first few months of life than in older children and newborns, prompting caution in determining the normal cut-off for infant sweat chloride concentrations. 10 Massie et al. 11 reviewed their sweat test data following NBS in New South Wales, Australia. Four of six infants with an initial ‘equivocal’ sweat chloride (40–60 mmol/L) were subsequently determined to have CF. The authors concluded that for sweat testing following NBS for CF, a cut-off of 40 mmol/L should be considered suggestive of a positive diagnosis and that infants with sweat chloride above this require careful assessment and follow-up.
The above studies have provided a useful insight into sweat testing in infants during their first months of life. However, they have not determined a reference interval, but have inferred this from data obtained from infants referred for a positive NBS test (persistent hypertrypsinaemia or a single recognized CF-causing mutation) or because of clinical suspicion. A systematic review concluded that reference intervals for sweat electrolytes are needed for all age groups. 12
The present study was designed to establish a reference interval for sweat chloride for infants, aged between 5 and 6 wk, without evidence of CF. At this age sweat testing may be necessary as part of an NBS protocol. Because of changing laboratory practice during the study period, two methods of sweat collection and analysis were used: (1) the traditional quantitative pilocarpine iontophoresis (QPIT [coulometry]) with filter paper collection (the Gibson Cooke method) and (2) the more recent capillary method (the Macroduct™ [ISE] system). This method change occurred as it is becoming increasingly difficult to obtain equipment for what is considered the gold standard method (QPIT [coulometry]), although this is the recommended method in the North American consensus guidelines on sweat testing. 13 Biochemists are increasingly moving to commercially available kits that comply with international (e.g. kite marking) standards, such as the Macroduct™ (ISE) system.
Methods
Patient recruitment
The study received approval from the Local Research Ethics Committee. Families were approached during the early postnatal period and an appointment was made for sweat collection to be undertaken at home at 5–6 wk of age (35–42 d of age). Preliminary studies validated the sweat transport and storage methods. Undertaking the test in the family home was more convenient and assisted with recruitment to the study. The parents also consented for the NBS blood spot sample to be sent for additional DNA analysis (phe508del CFTR mutations). Exclusion criteria were (1) a congenital syndrome, (2) preterm birth (less than 37 wk postconceptional age) or (3) any illness (for example, respiratory distress or poor weight gain) requiring review by a paediatrician or primary care physician. At the home visit, a clinical history (including a family history of CF) and examination were undertaken and weight and height measured.
Sweat tests
Both sweat collections were undertaken on the outer thigh area (standard local practice supported by national guidelines). A prior random allocation was used to determine which method was undertaken first. The sweat test was performed according to the national guidelines. 14 The second period of iontophoresis was commenced during the sweat collection on the first thigh. After a 30-min sweat collection, samples were appropriately processed to ensure that there was no change in electrolyte concentration during transport. Sweat samples from the Macroduct™ (ISE) tubing and the QPIT (coulometry) gauze pad were placed in their preweighed air-tight container. Minimum sweat weights were predetermined to be 20 mg for Macroduct™ (ISE) and 60 mg for QPIT (coulometry) in order to ensure a sweat rate of over 1 g/m2/min for both methods. Filter paper samples were eluted and sodium concentrations measured using flame photometry and chloride concentrations using coulometry.
For the sweat liquid samples collected in capillary tubes, sodium and chloride concentrations were measured using ion-selective electrodes (ISE) on an Abbott Architect c8000 (Abbott Diagnostics, Maidenhead, UK). These were calibrated using Abbott ICT urine calibrators with concentrations of 50 mmol/L and 180 mmol/L for both sodium and chloride. Fifteen microlitres of sample were required and the analyses were undertaken in urine mode. The working range was 20–400 mmol/L for sodium and 20–300 mmol/L for chloride. As the concentrations of sodium and chloride in sweat are typically much less than the middle of the ISE working ranges, a diluent (Beckman/Coulter – Synchron ELISE calibrator1 solution) with sodium of 100 mmol/L and chloride of 65 mmol/L was used to increase the electrolyte concentrations of the sweat samples into the working range of the ISEs. 20 μL of sweat was mixed with 60 μL of the Syncron calibrator 1 solution so that sweat represented 75% of the total volume. This mixed solution and the neat ‘Syncron’ diluent were analysed and both the results were used to calculate the sweat electrolyte concentrations. The result from the ‘diluent’ is multiplied by 0.75 and subtracted from the ‘diluted’ sweat result. The difference was then multiplied by 4, as the sweat sample was diluted 1 in 4 with the addition of the diluent. Internal quality control samples were prepared in the same manner as the sweat samples using the Syncron diluent and Quantimetrix sweat control 1, 2 and 3 with sodium and chloride concentrations typically in the of range 27–101 mmol/L and 22–98 mmol/L, respectively. The interassay analytical imprecision (coefficient of variation [CV]) of the chloride and sodium measurements were (mean, CV), 25.9 mmol/L, 8.3% and 24.4 mmol/L, 18.5%, respectively. Equivalent values obtained using Coulometry and Flame photometry were (mean, CV) 38.3 mmol/L, 11.0% for chloride and 25.4 mmol/L, 6.1% for sodium. Accuracy of the assays was confirmed by participation in the UKNEQAS scheme for sweat testing. The ‘B’ score (bias) and ‘C’ score (consistency of bias) for chloride measurements are −0.2 (allowable range −10 to +10) and 5.3 (allowable range 0–10), respectively. The ‘B’ score and ‘C’ score for sodium measurements are −3.7 (−10 to 10) and 5.3 (0–10), respectively.
Data analysis
We aimed to have complete data on 200 babies to enable a reference interval to be established. Data were analysed using descriptive statistics and sweat chloride values obtained by the two methods were compared using Bland-Altman difference plots. 15 From these, limits of agreement were calculated with 95% confidence intervals.
Results
A total of 315 families were recruited in the early postnatal period. Subsequently 39 were not contactable, 49 withdraw, five infants were unwell on the day of testing and 17 families were not able to visit during the allotted time period. In total therefore, 205 infants were tested at home. Eight samples were not analysed due to machine failure. Four infants were subsequently recognized to be heterozygous for the commonest CF mutation phe508del. No CF-causing mutations were recognized in the remaining 193 infants, including one infant with a positive family history of CF. Patients details are given in Table 1.
Demographic features and nutritional parameters of the 193 infants in the study
Insufficient sweat was collected on 28 occasions with the QPIT (coulometry) method and on 31 occasions with the Macroduct™ (ISE) (16 with both methods) giving an overall success rate of 92%. This was not related to the order of sweat collection. On 150 occasions sufficient sweat was collected by both the methods for the method comparison study (Figure 1). In the 24 non-Caucasian infants sufficient sweat was collected by both methods on 10 occasions which is a significantly lower proportion than for Caucasian infants (χ 2, P < 0.0001).
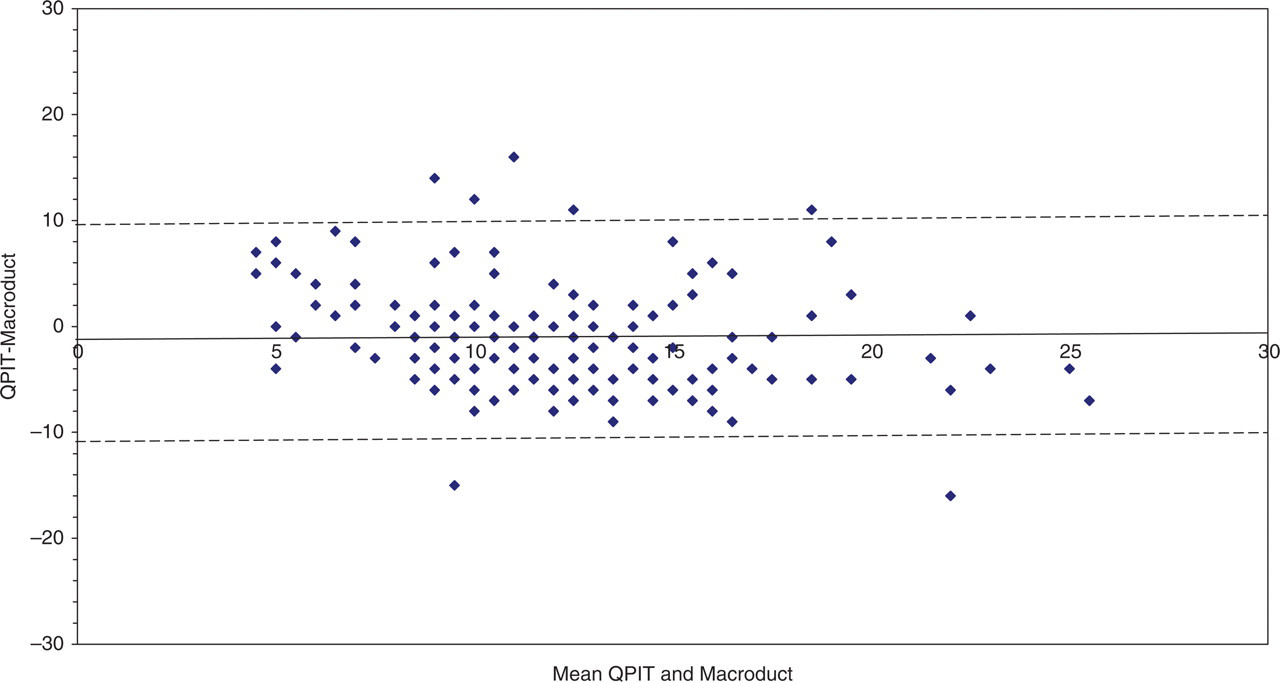
A Bland-Altman difference plot comparing sweat chloride concentrations obtained by QPIT (coulometry) and Macroduct™ (ISE). The x-axis is the mean of the two methods and the y-axis, the concentration from QPIT (coulometry) minus the concentration from Macroduct™ (ISE). The mean difference between the two methods QPIT (coulometry) minus Macroduct (ISE)) was −1.0 mmol/L (95% limits of agreement −10.8 mmol/L to 9.8 mmol/L [dotted line])
The median (interquartile range [IQR]) sweat chloride for the QPIT (coulometry) method was 11.2 mmol/L (8–13 mmol/L) (Table 2). The difference plot demonstrated excellent limits of agreement between the two methods, although the Macroduct™ (ISE) method did result in higher chloride concentrations (median 12.3 mmol/L). The sweat chloride concentrations showed a normal distribution (confirmed by two tests of normality) and a 99.5th centile of 30 mmol/L (Figure 2). Sweat sodium concentrations showed a skewed distribution (Figure 3) with a higher median (IQR) concentration of 15.6 mmol/L (10–20 mmol/L, Macroduct™ method) (Table 2). For the four infants, heterozygous for the phe508del mutation, sweat chloride concentrations were 21, 21, 5 and 9 mmol/L. The reference interval for sweat chloride in this age group was 1–22 mmol/L using the Macroduct™ (ISE) method.
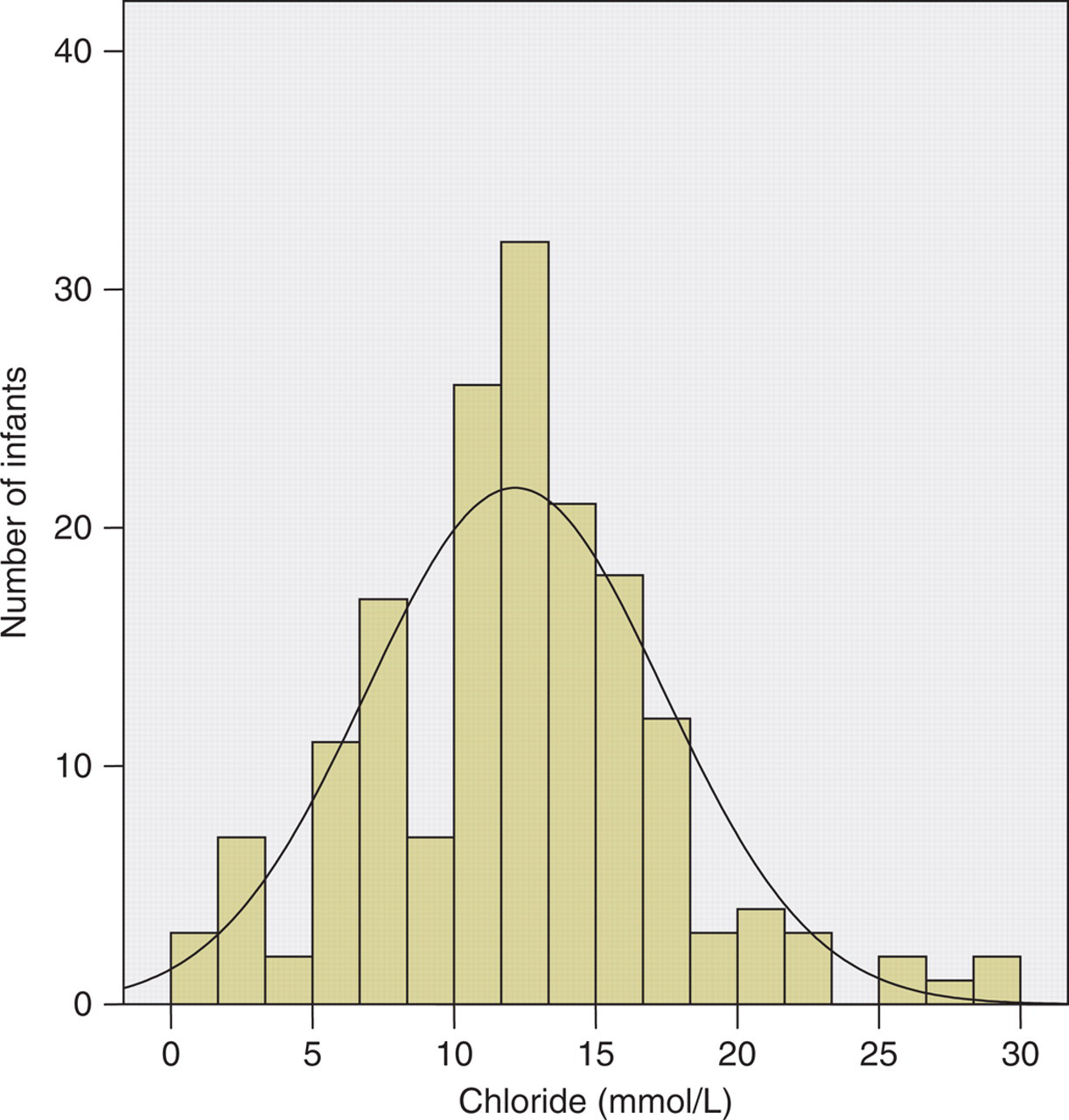
Distribution of sweat chloride concentrations from 165 infants between 5 wk and 6 wk of age (sweat collected by the Macroduct™ [ISE] method)
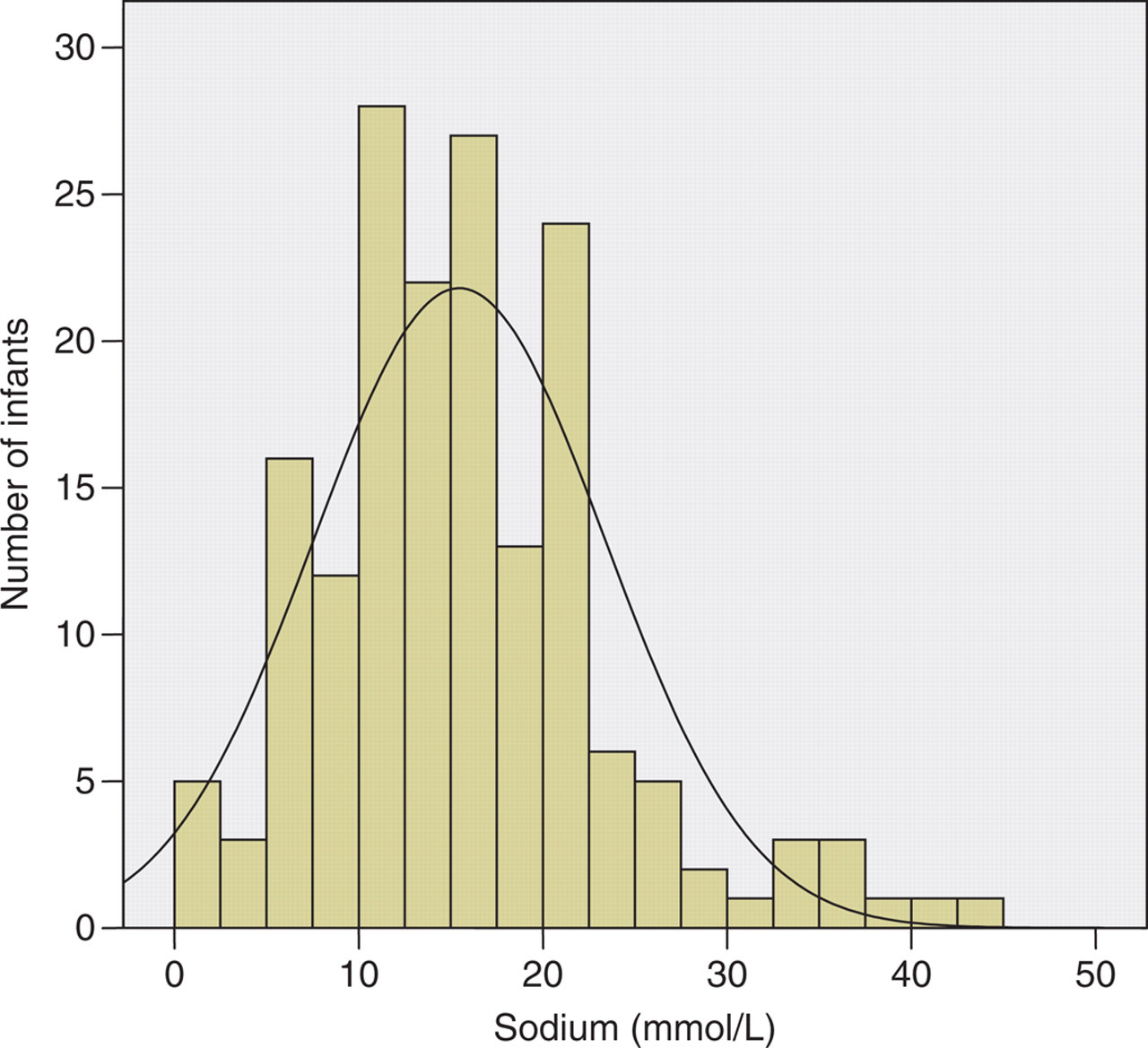
Distribution of sweat sodium concentrations from 165 infants between 5 wk and 6 wk of age (sweat collected by the Macroduct™ [ISE] method)
Sweat test data on 193 healthy infants at 35–42 days of age
QPIT, quantitative pilocarpine iontophoresis; IQR, inter-quartile range
Discussion
The primary aim of this study was to establish a reference interval for sweat chloride for infants, aged between 5 and 6 wk, when sweat testing is required to confirm a diagnosis of CF following NBS. Through the design of this study we are keen to ensure that the results would translate to current or evolving practice. To that end we included a comparison study between what is considered the gold standard sweat test method (QPIT with coulometry) and a method that is increasingly being used because of the availability of equipment and development of analysers (Macroduct™ [ISE]). The reference interval for sweat chloride in infants aged between 5 and 6 wk was 1–22 mmol/L using the Macroduct™ (ISE) method. This age was chosen as it represents the point in the NBS protocol that sweat testing is often required to clarify an equivocal result (for example, an infant with persistently raised immunoreactive trypsinogen but only one CFTR mutation recognized). Data from previous studies suggest that this age may represent a nadir in sweat chloride concentration, with a reduction from the high concentrations seen in the immediate postnatal period and lower than those seen in older children and adults. 16 Our data supports this, with a median (IQR) sweat chloride of 11.2 mmol/L (8–13 mmol/L) obtained using the traditional QPIT (coulometry) method. A concentration above 30 mmol/L is above the 99.5th percentile and we would recommend that infants with a sweat chloride above this concentration are assessed in specialist CF centre. This is consistent with the recommendation of the most recent North American sweat test guidelines. 12 A survey of practice across Europe suggests that a cut-off of 40 mmol/L is used in most CF centres and is also contained in the current UK sweat test guidelines. 14
We compared both the methods of sweat collection and found no difference in the rate of successful sweat collection, irrespective of which method was undertaken first. The minimum acceptable weight of sweat used in this study ensured a sweat production rate of greater than 1 g/m2/min. Sweat collection is usually undertaken in hospital, however to facilitate recruitment into this study we visited the family home. The samples were refrigerated and analysed within 48 h, as per UK guidelines. Overall, we had a 92% success rate of sweat collection. In 16 of the 193 infants studied we were unable to get an adequate collection through either method. These data support the use of the capillary method for sweat collection in this age group. The collection of sweat outside the laboratory raises the possibility of providing an ambulatory service, but this requires a definitive study of cost benefit.
Previous studies suggest that new-born infants have high sweat electrolytes during the first 24 h of life. 8,16 It therefore appears that following adaptation to postnatal life, sweat electrolyte concentrations fall significantly over the first weeks of life. This salt conservation may represent a physiological requirement, as infants have a relatively low salt intake and large surface area for sweat losses.
In addition to establishing a reference interval, we compared the QPIT (coulometry) method with the Macroduct™ (ISE) capillary collection system. A comparison of paired chloride concentrations obtained by both methods demonstrated excellent limits of agreement between the two methods. However, sweat chloride concentrations obtained from the Macroduct™ (ISE) method were about 1 mmol/L higher, although this is not clinically significant. As the QPIT (coulometry) system has become obsolete, in our centre we have moved to collecting sweat with the Macroduct™ (ISE) system (which is the only available system that complies with current standards for medical equipment). The 99.5th centile for sweat chloride with the Macroduct™ (ISE) method was 30 mmol/L compared to 24 mmol/L with the QPIT (coulometry), supporting the conclusion that the Macroduct™ (ISE) may be producing slightly higher sweat chloride value.
The infants were well and representative of a cosmopolitan Northern European population. There was no evidence of differences in sweat chloride with respect to race (median 10.4 mmol/L), although numbers were small for this non-Caucasian subgroup analysis. Sweat collection appeared to be more difficult in the non-Caucasian infants as there was a higher proportion of insufficient sweat collections, but numbers were too small to draw a definitive conclusion. Four infants were heterozygous for the phe508del mutation and their sweat electrolyte concentrations were not included in the reference interval data. All infants were tested for phe508del mutation and if found heterozygous had a further extended panel of mutations screened. We cannot be certain that carriers of more unusual mutations were not included in this cohort. Previous studies have demonstrated significantly higher sweat chloride concentrations in CF carrier infants. 9
Collection of sweat by the capillary system facilitates the measurement of conductivity, which gives an overall measure of the ionic load of sweat. 17 Technically this is easier, and newer technology has enabled this to be undertaken at the bedside of small infants (the Nanoduct™). 18 However, a number of consensus groups consisting of experts from Europe and the USA have stated that conductivity does not have sufficient validity as a standalone diagnostic test and sweat chloride is considered the gold standard. 13,19 Our results demonstrate a normal distribution of sweat chloride concentration in these infants without CF and would support the validity of using this outcome in determining an abnormal sweat test.
In summary we have investigated sweat testing in healthy infants at 5–6 wk of age and have generated a reference interval for sweat chloride. We have shown that the Macroduct™ (ISE) sweat collection system is as valid as QPIT (coulometry) in this age group with comparable concentrations and similar rates of adequate sweat collection. Our data support previous studies which suggest that sweat chloride concentrations are lower in this age group and we recommend that any 5–6-wk-old infant with a measured sweat chloride of greater than 30 mmol/L should be assessed in a specialist CF centre.
Footnotes
Acknowledgements
The authors would like to thank Dr Jean Craig for valuable advice on the methods comparison study, Dr Anne Green and Dr Phil Farrell for helpful comments and the families for their enthusiastic support. Dr Jayaraj was supported by a Royal Liverpool Children's Hospital Trust Research Fellowship.
