Abstract
Background
The diagnosis of disorders of growth hormone (GH) secretion is based on the measurement of GH before and after dynamic stimulation or suppression tests. Over the years, specific cut-off values have been proposed without taking account of the considerable variation in results obtained using different methods for analysing GH. Recent publications have recommended the standardization of GH assays using IS 98/574, which is calibrated in mass and units (U). The IMMULITE range of GH assays have been recalibrated using this standard.
Methods
We analysed 745 samples using the current and the restandardized GH assay kits. This series of samples contained 90 stimulation tests and seven suppression tests.
Results
The data comparing the current and restandardized assays show a similar relationship for both basal and stimulated GH values for all IMMULITE platforms. There is a negative bias by the restandardized assay above approximately 20 μg/L. A conversion can be derived from this study which would mean that patient GH results expressed in milliunits per litre of IMMULITE in-house derived standard will be approximately three-fold of that expressed in micrograms per litre of IS 98/574.
Conclusion
Use of IS 98/574 offers standardized reporting in microgram per litre IS 98/574. This will permit compliance with the consensus recommendations and the development of a firm evidence base for defining decision-making GH values.
Introduction
The diagnosis of disorders of growth hormone (GH) secretion is based on the measurement of GH before and after dynamic tests. Over the years, specific cut-off values have been proposed without reference to the considerable variation in results because of different methods for analysing GH. The many factors contributing to this variance were discussed in a meeting held at the National Institute for Biological Standards and Control (NIBSC) in 2000. 1 One of the conclusions was that the recombinant DNA-derived standard (IS 98/574) should replace the pituitary-derived standard IS 80/505. This was further endorsed by a proposal in 2004 that IS 98/574 should be adopted by the diagnostics industry as its primary calibrant for GH assays and that clinicians and laboratories should adopt mass units for reporting GH results. 2 In 2006, major clinical endocrinology journals advised submitting authors that they would only accept GH data submitted in micrograms per litre of IS 98/574. 3
The key issue with IS 80/505 is that it is defined in units, but has no assigned mass value. This has led to factors between 2 and 3 being used to convert mass to units. The lack of transparency in this conversion has clearly contributed to the confusion over definitions of GH deficiency. The more recent IS 98/574 is a recombinant preparation with defined physico-chemical characteristics and an assigned value of 3.0 U/mg. This will enable reporting of GH results in micrograms per litre or milliunits per litre of IS 98/574; such that values expressed in micrograms per litre will be one-third of the same values expressed in milliunits per litre.
Universal reporting of GH results in terms of micrograms per litre of IS 98/574 should ultimately improve analytical consensus as well as provide a sound basis for improved international agreement on GH decision points, e.g. for stimulation and suppression tests. The IMMULITE range of GH assays has been recalibrated using this standard. These developments were kept pending and to provide information in the interim, we have carried out a comparison study of the current and restandardized kits, as it is possible that the differences in the standards (original IMMULITE versus IS 80/505 versus IS 98/574) might cause minor differences in the measured response to these substances. 4
Methods
We used redundant serum samples after routine clinical measurement of GH. There were a total of 745 samples from 224 individuals. The subjects were women (n = 107) aged 42.3 (38.2) years (median [IQR]) and men (n = 121) aged 42 (41.4) years. There were 50 subjects under 16 years (girls = 19, boys = 31). The collection of serum samples included samples obtained following administration of GH secretagogues: clonidine (two series), glucagon (77), insulin (nine) and unspecified agents. 2 A further series of samples were taken during oral glucose tolerance tests (seven series). All samples were anonymized prior to analysis.
GH was analysed on the three Siemens IMMULITE platforms (1000, 2000 and 2500) using the current and the restandardized GH assay kits (Siemens Healthcare Diagnostics, Llanberis, UK). The current assay is calibrated with an in-house preparation of GH, with a derived conversion factor of 1 μg of the in-house GH = 2.4 mU IS 98/574. The recalibrated assay (to be released shortly) is calibrated with IS 98/574, which is defined with a fixed relationship of 3 mU to 1 μg. The assays are non-competitive and use a murine monoclonal antibody attached to the solid phase and a rabbit polyclonal antibody labelled with alkaline phosphatase. Assays were performed according to the manufacturer's protocol. Intra-assay precision for the IMMULITE 2000 varied between 4.2 and 4.6%; inter-assay precision varied between 4.2 and 6.6%.
Statistical analysis was performed using the Analyse-it add-in package (version 2.08) for Microsoft Excel (Analyse-it, Leeds, UK,
Results
Comparing results from the current and restandardized assays when expressed in milliunits per litre of IS 98/574, Bland and Altman plots (Figures 1a and b) demonstrate agreement between the methods at concentrations up to 20 mU/L. Above this there is some evidence of small differences between the assays which are not likely to be of clinical significance. (The relationship becomes non-linear at values >60 mU/L [>20 μg/L]; NCCLS linearity test.)
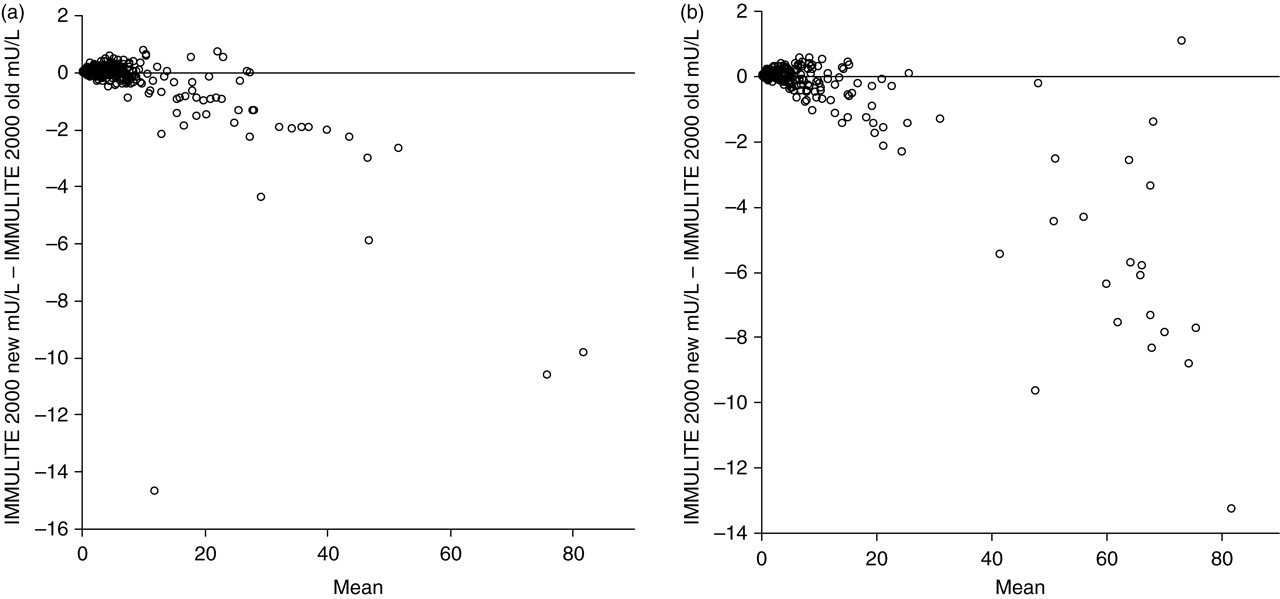
Altman-Bland plots comparing the two growth hormone (GH) assays on the IMMULITE 2000. Results for both assays are expressed in milliunits per litre. Part (a) compares basal and unstimulated samples. Part (b) compares samples following GH stimulation by clonidine, glucagon or insulin. See Methods section for explanation of assays
The data comparing the current assay (results expressed in milliunits per litre) and the restandardized assays (results in micrograms per litre) show a similar, although not identical, relationship for all platforms on both basal and stimulated GH samples (Table 1).
Comparison of the restandardized (y) and current (x) assays were made for growth hormone (GH) measurements on basal and stimulated samples on each of the three IMMULITE platforms
In this table, the data for the new assay (y) is expressed in micrograms per litre; this allows comparison of new assay (y) in micrograms per litre with the old assay (x) expressed in milliunits per litre with an approximate 1:3 conversion (Passing & Bablok regression equations are shown)
Slopes of the regression lines from this study are all close to 0.33, indicating that patient GH results measured in milliunits per litre of IMMULITE in-house derived standard will be approximately three-fold those expressed in micrograms per litre of IS 98/574.
Discussion
Since 2000 the IMMULITE assays have been calibrated using an in-house-derived standard with a value assigned by comparison with pituitary-derived IS 80/505. Since then no other reagents (including antibodies) have been changed and, other than recalibration, the restandardized assay is identical to the previous assay. In 2004, a software upgrade was supplied enabling users to employ a conversion factor of 1 μg to 2.4 mU and allowing users to report results in milliunits per litre IS 98/574. The present study shows the effect of reformulating the assay by replacing the previous in-house-derived standard with IS 98/574, for which there is a defined relationship of 1 μg to 3 mU.
This report describes the effect of this change in the standardization of GH measurements. The assays demonstrate a relationship across the concentrations studied which should give sufficient confidence for clinicians to use the values produced by the restandardized kit provided it is calibrated to IS 98/574. We have included a number of samples taken after administration of GH secretagogues. This is important as these agents cause the release of a number of GH isoforms all of which have different clearance times. 5 These isoforms may have different antibody-binding characteristics resulting in different measured concentrations of total GH when the relative proportions of isoforms change. 6 This may contribute to the widespread confusion regarding appropriate cut-offs for GH deficiency. 7,8 We have shown that the relationship between assays remains constant for values <20 μg/L (60 mU/L) even after stimulation. The negative bias of the recalibrated assay at concentrations >20 μg/L (60 mU/L) should not compromise the clinical interpretation of results.
Standardization with IS 98/574 will permit reporting in micrograms per litre IS 98/574 in line with the recommendations cited above, 1,2 and therefore, future studies of GH will be comparable. IS 98/574 is defined in milliunits and micrograms unlike IS 80/505, which was only defined in milliunits and unspecified conversion factors to convert to micrograms were employed. Furthermore, the reduction in the number of commercially available GH assays suppliers offers an opportunity to progress the adoption of IS 98/574 rapidly. Finally, it is time to develop the evidence base for defining decision-making GH values, which can be put forward for international consensus.
Footnotes
ACKNOWLEDGEMENTS
We are extremely grateful to Dr C Sturgeon and Mr A Ellis of UK NEQAS and Mr G Wieringa for their critical advice.
