Abstract
Background
β2-Microglobulin (β2m) has been identified as the precursor protein of dialysis-related amyloidosis (DRA), which is a serious complication for haemodialysis (HD) patients. However, mechanisms underlying β2m amyloid fibril formation remains to be elucidated. We previously demonstrated, in amyloid deposits from HD patients, a conformational isoform of β2m with an unfolded C-terminus. However, no direct experiments have previously been performed to address whether unfolded β2m in the C-terminus may be prone to form amyloid fibrils.
Methods
To evaluate roles of C-terminal amino acids in β2m-induced amyloid formation, we generated six types of recombinant β2m with amino acid substitutions in the C-terminal region. To investigate their conformational change and amyloidogenicity, we measured circular dichroism spectra, the fluorescence intensity of tryptophan and thioflavin-T (ThT) of the recombinant β2m. To analyse morphological change of β2m, we performed electron microscopy (EM) on the samples with elevated ThT fluorescence intensity. We used ultrasonication to enhance β2m destabilization of the protein.
Results
β2M Trp95Leu and Arg97Ala showed conformational changes and increased their amyloidgenicity compared with β2m wild-type (WT). With ultrasonication, β2m Trp95Leu and Arg97Ala generated more amyloid fibrils than did β2m WT even in physiological solution. EM showed that β2m formed amorphous debris containing typical amyloid fibrils at 24 hours, when ThT fluorescence intensity was three-fold lower than that at six hours.
Conclusions
Conformational changes in the C-terminus of β2m may play an important role in DRA and that ultrasonication is useful for analysis of β2m amyloidogenesis.
Introduction
In 1985, Gejyo et al. 1 identified β2-microglobulin (β2m) as the precursor protein of dialysis-related amyloidosis (DRA). DRA, a serious complication occurring in patients receiving long-term haemodialysis (HD), causes various clinical manifestations, such as carpal tunnel syndrome, polyarthralgia, destructive spondyloarthropathy and bone cysts. More than 2,40,000 Japanese patients are treated with chronic HD, and of those, 6.4% have a history of HD for more than 20 years. Up to 50% of patients had developed DRA after 20 years and the percentage was even higher after 25 years. 1 However, to date, no established treatments of DRA have been developed, and the mechanism of β2m amyloid fibril formation remains to be elucidated.
β2M consists of 99 amino acids and appears to be simple in the plane dimension, but in 1985, 2 Becker and Reeke identified a three-dimensional configuration with a complex conformation involving seven β strands. Several variants of β2m have been found in clinical specimens, including amyloid-containing tissues from HD patients. 3–6 However, no reports of unfolded variants in a clinical setting, except for an abstract by Rampino et al., 7 have been published. Stoppini et al. 8 reported that carpal amyloid-containing tissues from HD patients showed a positive reaction for a monoclonal antibody specific to the C-terminal octapeptide (i.e. residues 92–99 of β2m) as well as to a commercial polyclonal anti-β2m serum under physiological conditions. This C-terminal portion is believed to be normally hidden in the core of the quaternary structure of the light chain of class I major histocompatibility complex 9 and therefore is regarded as being non-reactive with commercial polyclonal anti-β2m serum.
That misfolding of the amyloid precursor protein initiates amyloid formation in several types of amyloidosis, such as transthyretin amyloidosis, has been well studied, and therapeutic approaches have been proposed. 10–12 In 1998, Bellotti et al. 13 first demonstrated, in amyloid deposits obtained from HD patients, the presence of a partially unfolded β2m that could refold reversibly into native β2m.
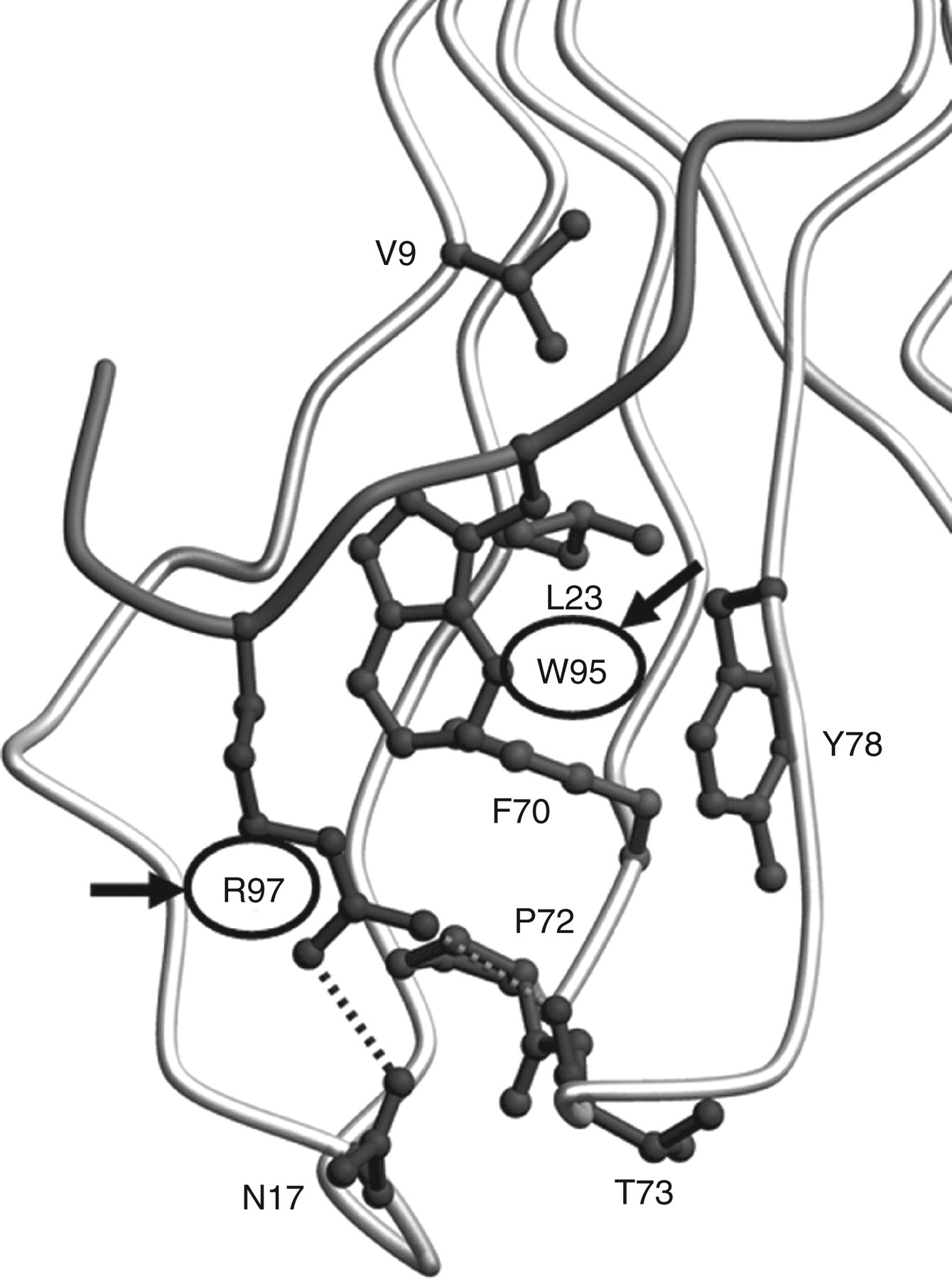
We previously reported preparation of a monoclonal antibody against the C-terminal octapeptide of β2m and identified, in amyloid deposits obtained from DRA patients, a conformational isoform of β2m with an unfolded C-terminus. 14 However, no experiments have been performed to specifically show that β2m that has a misfolded C-terminus may be more likely to produce amyloid fibrils than native β2m. Trp95 and Arg97 are believed to be important residues for stability of β2m (Figure 1). In this study, to evaluate the role of C-terminal amino acids in β2m amyloid formation, we generated six types of recombinant β2m with amino acid substitutions in the C-terminal region (Trp95Leu, Trp95Glu, Trp95Ala, Trp95Val, Trp95Ser and Arg97Ala).
Amyloid formation can also be enhanced by destabilizing or unfolding proteins by increased temperature, high pressure and chemical agents in vitro. 15–17 Stathopulos et al. 18 reported that ultrasonication of various proteins induced amyloid formation. Ultrasonication is also frequently used to break aggregates into smaller pieces for seeding new aggregate growth in laboratory studies of proteins associated with various conformational disorders, such as prion diseases. 18 Saborio et al. 19 developed a new method using ultrasonication, termed protein misfolding cyclic amplification (PMCA), for early diagnosis of prion disorders. In PMCA, a small amount of prion aggregates can be detected with high sensitivity by amplifying the prion aggregates through cycles of ultrasonication which breaks small amount of prion aggregates into smaller pieces acting as seeds for the formation of new aggregates from soluble prion protein.
In this report, we also employed ultrasonication to enhance destabilizing of those recombinant β2m and investigated amyloid fibril formation in those β2m.
Methods
Protein expression and purification of recombinant β2microglobulin
β2M wild-type (WT) and mutated human β2m were produced in Escherichia coli with six histidine residues and a Factor Xa cleavage site attached to the N-terminus. The pET28-hβ2m was produced as follows. A culture of E. coli was grown in luria broth (LB) medium supplemented with 100 mg/L ampicillin, with agitation, at 37°C overnight. At a bacterial density of OD600 0.6–1.0, isopropyl thio-β-
Structural analysis
The structure of the C-terminus was drawn by using MolScript 21 and Raster3D. 22
Measurements of circular dichroism spectra
Circular dichroism (CD) spectra of β2m WT, β2m Trp95Leu and β2m Arg97Ala were measured at 25°C with a J-720-type spectropolarimeter (JASCO, Tokyo, Japan). The standard buffer for spectroscopic measurements was 20 mmol/L Tris–HCl (pH 7.4). For far-UV measurements (200–250 nm), the CD signal was recorded in a 1 mm path length cell. Data for far-UV CD were recorded with a 1 nm bandwidth, 1 nm resolution, scan speed of 20 nm/min and response time of two seconds. Spectra were recorded at a protein concentration of 10 μmol/L. Results were expressed as mean residue ellipticity (θ).
Tryptophan fluorescence intensity
The fluorescence intensity of trptophan (trp) was measured using a Hitachi F-2000 spectrofluorimeter (Hitachi, Tokyo, Japan) at 25°C. All assays used an excitation wavelength of 295 nm and an emission wavelength of 340 nm. Excitation and emission slits were set at 5 nm. Trp fluorescence intensity was recorded at a protein concentration of 10 μmol/L in 67 mmol/L sodium phosphate (pH 7.4).
Thioflavin-T-based fluorimetric assays
β2M was dissolved in distilled water, syringe-filtered (0.25 μm) to remove aggregates, and diluted in Eppendorf tubes to a final concentration of 30 μmol/L in 50 mmol/L glycine–HCl (pH 2.5) containing 100 mmol/L NaCl. Samples (1.5 mg/mL) were then incubated at 37°C for 10 days. The resulting suspensions were analysed using the ThT fluorescence test. 23,24 Fluorescence spectra were obtained by means of a Hitachi F-2000 spectrofluorimeter (Hitachi) with an assay volume of 1 mL. All assays used an excitation wavelength of 448 nm and an emission wavelength of 489 nm. Excitation and emission slits were set at 5 nm. The reaction mixture contained 5 μmol/L ThT and 50 mmol/L Gly–NaOH buffer, pH 10.0. 23,24 Spectra were obtained at 25°C within minutes after addition of the sample to the reaction mixture.
Ultrasonication
Directional ultrasonic treatment was performed with a water bath-type ultrasonic transmitter (Model SP070-PG-M035SP, Elekon, Tokyo, Japan), which focused 20 kHz ultrasonic waves from two sides and the bottom on the central part of the water bath where the reaction samples were placed. The temperature of the water bath was maintained at 37°C using a thermostatic water bath (Model NP035-08, Elekon). This ultrasonication proceeded for one minute, followed by incubation for nine minutes without ultrasonication. Aliquots were collected to measure the degree of ThT fluorescence.
Ultrasonication-induced fibril formation
For seeded growth in an acidic extension reaction, fibrils of β2m were first induced to form by incubating 2 mg/mL β2m at 37°C with ultrasonic treatment in 50 mmol/
Electron microscopy
Pellets (5 μL) from each extension reaction were diluted with 95 μL of distilled water. Samples were placed on carbon/collodion-coated grids and were allowed to adsorb for one minute. Samples were then stained with a drop of 0.2% uranyl acetate for 30 seconds. Excess stain was drained off, after which grids were air-dried. All images were taken with a Hitachi H-7500 electron microscope (Hitachi) operating at 80 keV.
Statistical analysis
Student's t-test (two-tailed, for independent samples) was used to determine significant differences (P < 0.05).
Results
Production of recombinant β2m with amino acid substitutions in the C-terminal region
To evaluate the effects of conformational changes in the β2m C-terminal amino acids on amyloid formation, we produced six different kinds of recombinant β2m with an amino acid substitution in strand G of the C-terminal region—Trp95Leu, Trp95Glu, Trp95Ala, Trp95Val, Trp95Ser and Arg97Ala (Figure 1).
We obtained and collected recombinant β2m WT, β2m Trp95Leu and β2m Arg97Ala as pure protein. MALDI-TOF MS of the purified proteins revealed the molecular mass of β2m WT, β2m Trp95Leu and β2m Arg97Ala to be 11,731, 11,655 and 11,645 Da, respectively. However, we could not collect and purify other recombinant proteins—β2m Trp95Glu, β2m Trp95Ala, β2m Trp95Val and β2m Trp95Ser—because of their aggregative nature.
Changes in conformation of β2m Trp95Leu and β2m Arg97Ala
We utilized CD analysis to investigate conformational changes of β2m Trp95Leu and Arg97Ala. The CD spectra of β2m Trp95Leu and Arg97Ala near 218–220 nm were broader and decreased than that of β2m WT, which was consistent with the β-sheet structure of the native protein (Figure 2a). To investigate conformational changes around the Trp residues, we also analysed Trp fluorescence for β2m WT, Trp95Leu and Arg97Ala. The Trp fluorescence spectrum of β2m WT had a maximum at 334 nm, but the maximum for the β2m Trp95Leu and Arg97Ala spectrums had shifted to 349 and 342 nm, respectively, which indicated exposure of the Trp residues. Compared with β2m WT, β2m Trp95Leu and Arg97Ala had lower intensity in Trp fluorescence (Figure 2b).
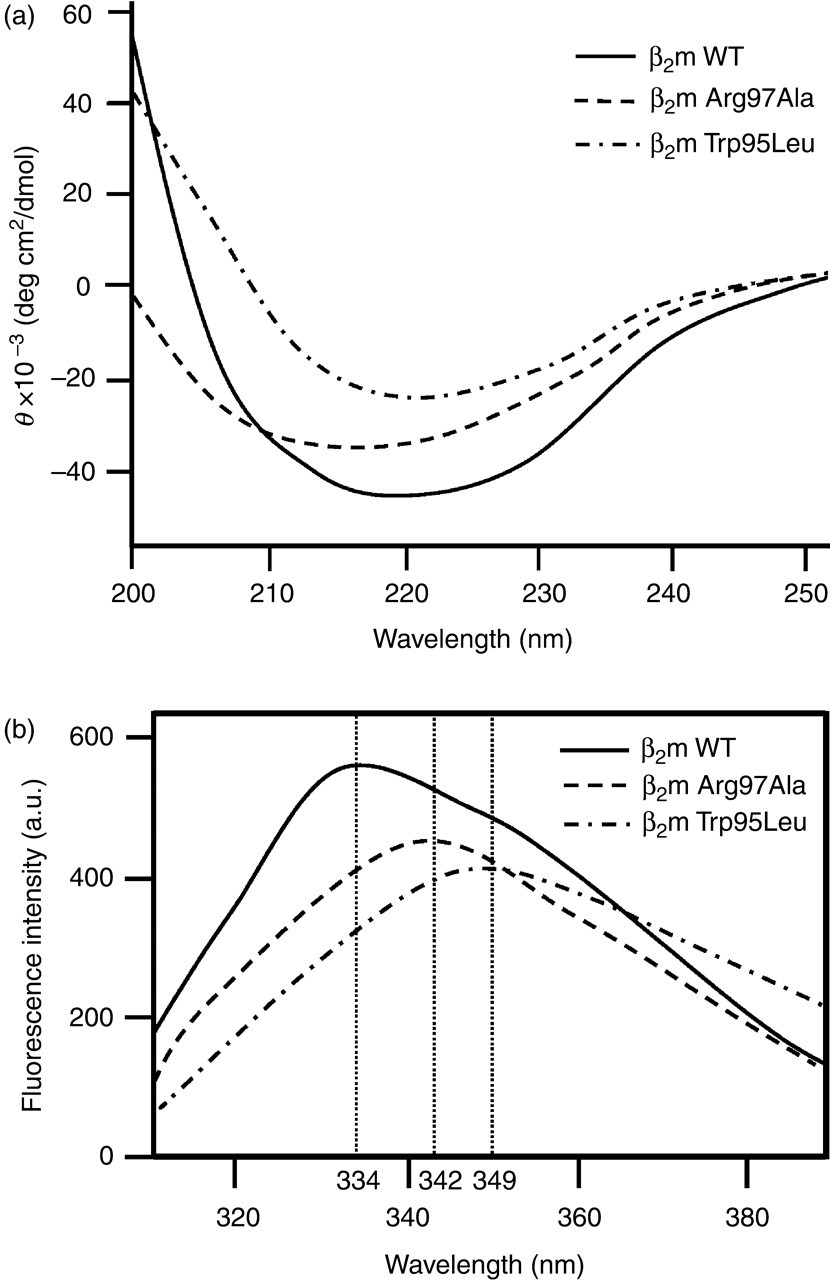
Circular dichroism (CD) spectra and tryptophan (Trp) fluorescence intensity for β2m wild-type (WT), Trp95Leu and Arg97Ala. (a) Far-ultraviolet CD spectra of β2m WT, Trp95Leu, and Arg97Ala. Samples of 10 μmol/L β2m WT, Trp95Leu and Arg97Ala (in 67 mmol/L sodium phosphate, pH 7.4) were applied to a JASCO J-720 spectropolarimeter. (b) Trp fluorescence intensity for β2m WT, Trp95Leu and Arg97Ala. Fluorescence intensity of 2 mL samples of 10 μmol/L β2m WT, Trp95Leu and Arg97Ala was measured using a Hitachi F-2000 spectrofluorimeter. Solid line, β2m WT; dashed lines, β2m Trp95Leu and Arg97Ala
Amyloid fibril formation of β2m WT, Trp95Leu and Arg97Ala at acidic pH
To examine the differences in amyloidogenesis of β2m WT, Trp95Leu and Arg97Ala, samples were incubated at pH 2.5. The ThT fluorescence of β2m Trp95Leu and Arg97Ala were significantly greater than that of β2m WT (Figure 3).
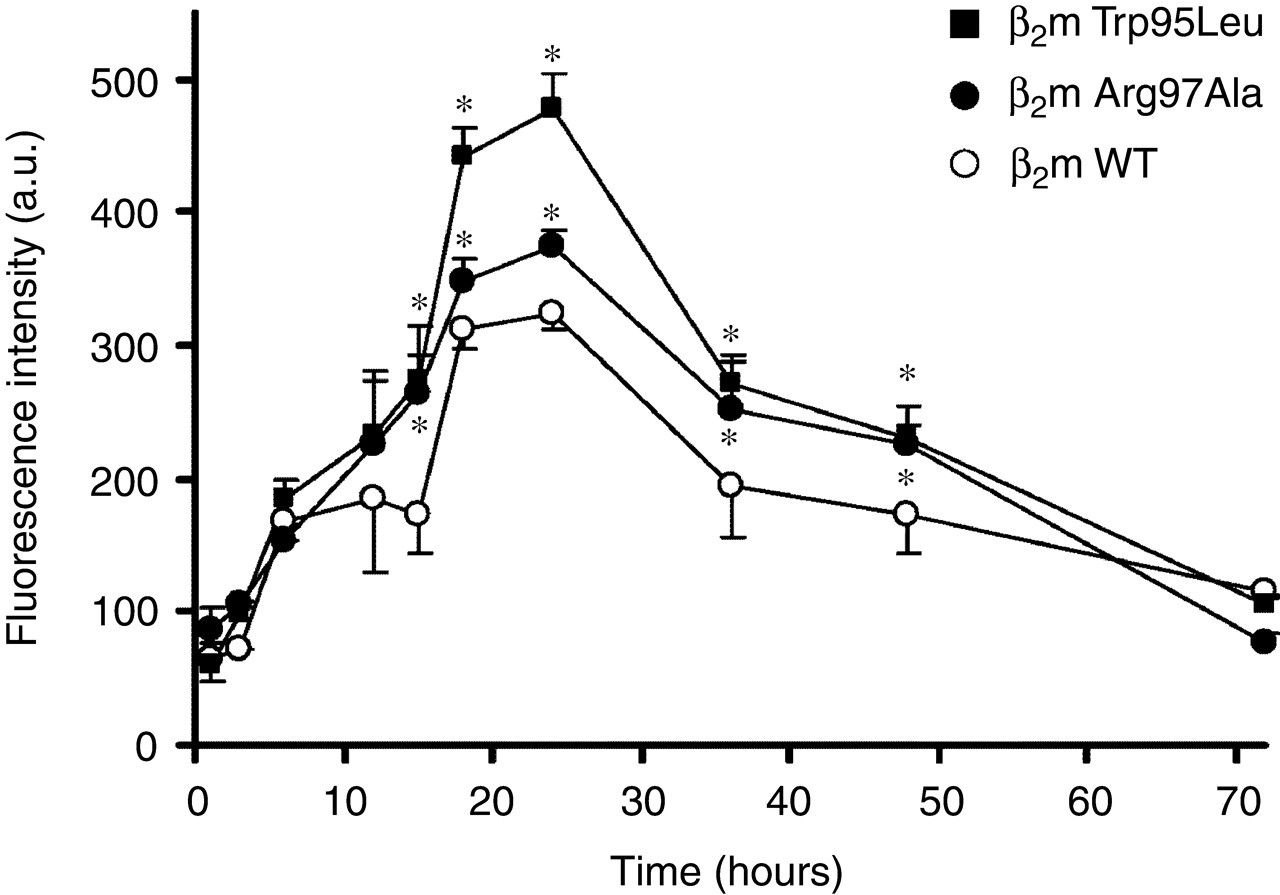
Amyloid fibril formation of β2m wild-type (WT), Trp95Leu and Arg97Ala at an acidic pH (pH 2.5), monitored by thioflavin-T fluorescence. β2m WT (○), Trp95Leu (▪) and Arg97Ala (•). Data are expressed as mean ± standard deviation; n = 3. *P < 0.05 compared with β2m WT
Ultrasonication-induced amyloid fibril formation
To enhance destabilization of β2m, we used ultrasonication as described in Materials and Methods. Incubation of samples of β2m WT, Trp95Leu and Arg97Ala at pH 2.5 with ultrasonication dramatically increased the ThT fluorescence intensity of β2m Trp95Leu and Arg97Ala compared with that of β2m WT (Figures 4a and b). With ultrasonication, the ThT fluorescence intensity peaked earlier than those without ultrasonication (Figures 3, 4a and b). Interestingly, the ThT fluorescence intensity of the β2m samples both with and without ultrasonication, gradually decreased after 6 and 12 hours, respectively. In addition, at physiological pH, we also incubated β2m Trp95Leu, Arg97Ala and WT with or without ultrasonication. With ultrasonication at neutral pH, the ThT fluorescence intensity of β2m Trp95Leu and Arg97Ala increased more markedly than did that of β2m WT, with the maximal value achieved at five days (Figure 5), although those β2m did not form amyloid fibrils without ultrasonication (data not shown). The ThT fluorescence intensity of β2m at neutral pH with ultrasonication peaked later than those at acidic pH with ultrasonication (Figures 4a, b and 5).
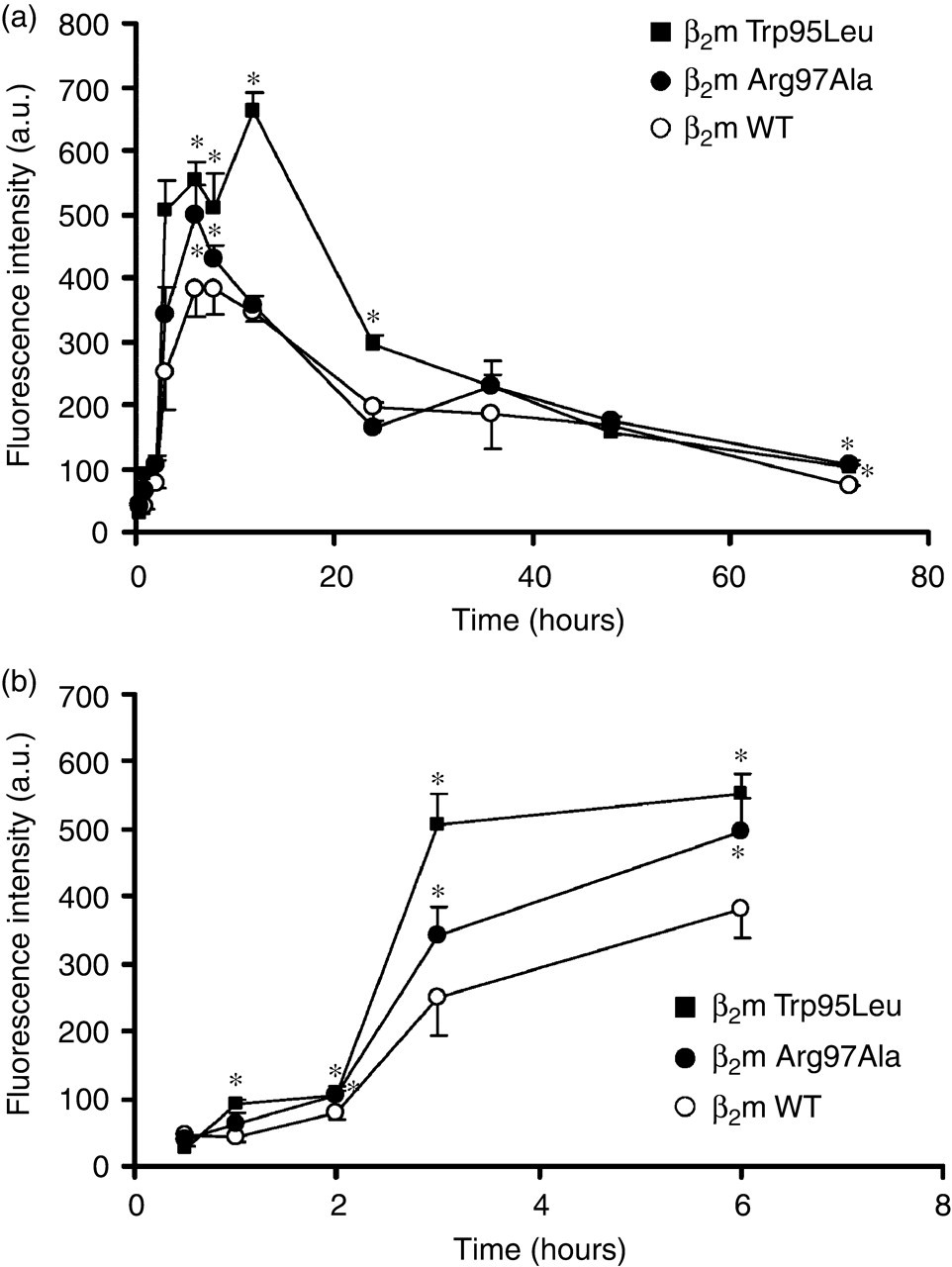
Time course of amyloid fibril formation induced by β2m wild-type (WT), Trp95Leu and Arg97Ala as monitored by thioflavin-T (ThT) fluorescence. (a) ThT fluorescence intensity of ultrasonicated samples of β2m WT, Trp95Leu and Arg97Ala at pH 2.5. (b) Early changes (0–6 hours) of β2m fibril formation at pH 2.5 with ultrasonication. β2m WT (○), Trp95Leu (▪) and Arg97Ala (•). Data are expressed as mean ± standard deviation; n = 3. *P < 0.05 compared with β2m WT
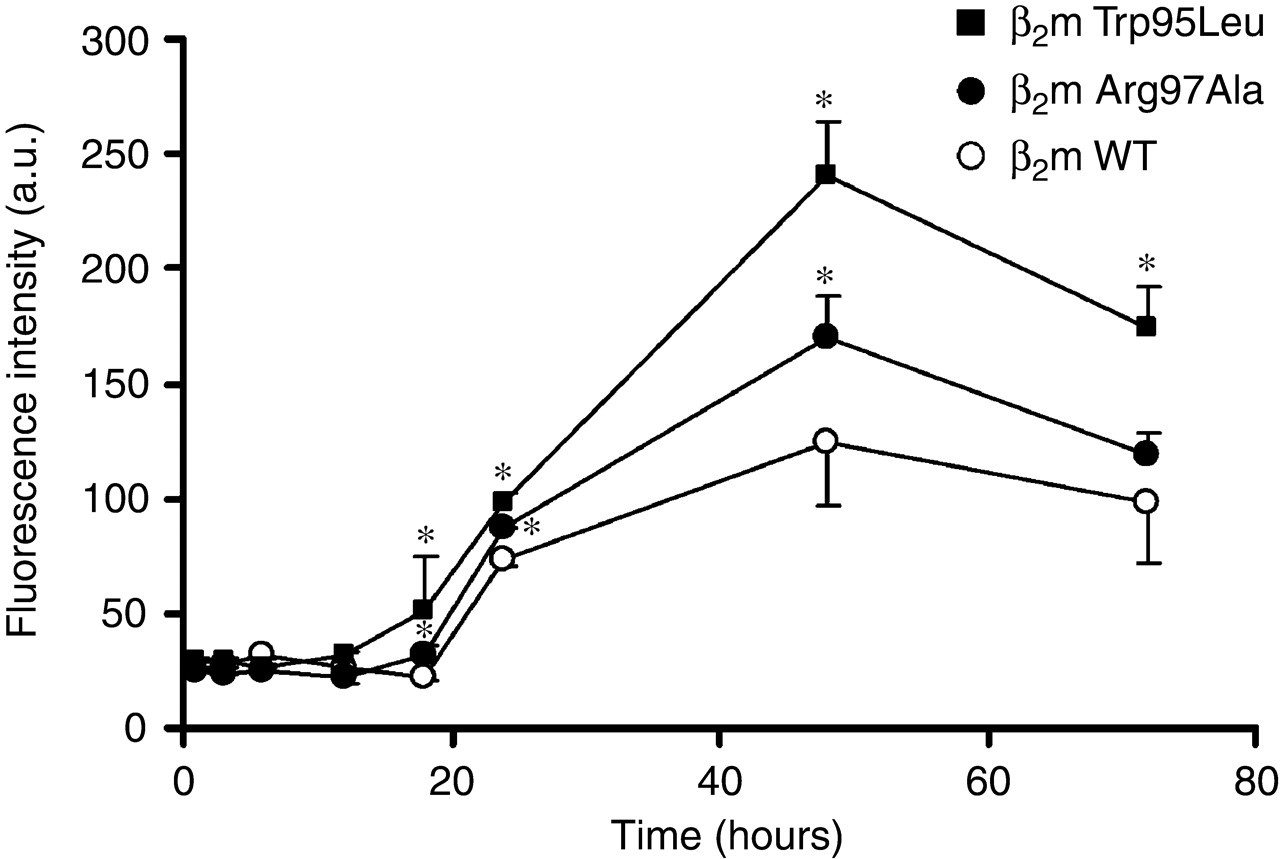
At neutral pH (pH 7.0), ultrasonication inducing amyloid fibril formation of β2m wild-type (WT), Trp95Leu and Arg97Ala, monitored by thioflavin-T fluorescence. β2m WT (○), Trp95Leu (▪) and Arg97Ala (•). Data are expressed as mean ± standard deviation; n = 3. *P < 0.05 compared with β2m WT
Time-dependent morphological changes of β2m Arg97Ala with ultrasonication
To evaluate morphological changes in amyloid fibrils at different times (6 and 24 hours), we obtained electron microscopic (EM) images of β2m Arg97Ala incubated with ultrasonication at acidic pH. At six hours, β2m Arg97Ala formed long, straight fibrils and also partially curved fibrils (with diameters of approximately 10 nm) (Figure 6a). At 24 hours, when ThT fluorescence intensity of β2m Arg97Ala was three-fold lower than that at six hours, EM images showed amorphous debris that contained long, straight fibrils (also with diameters of approximately 10 nm) (Figure 6b). The morphology of fibrils in the debris was that of typical amyloid fibrils.
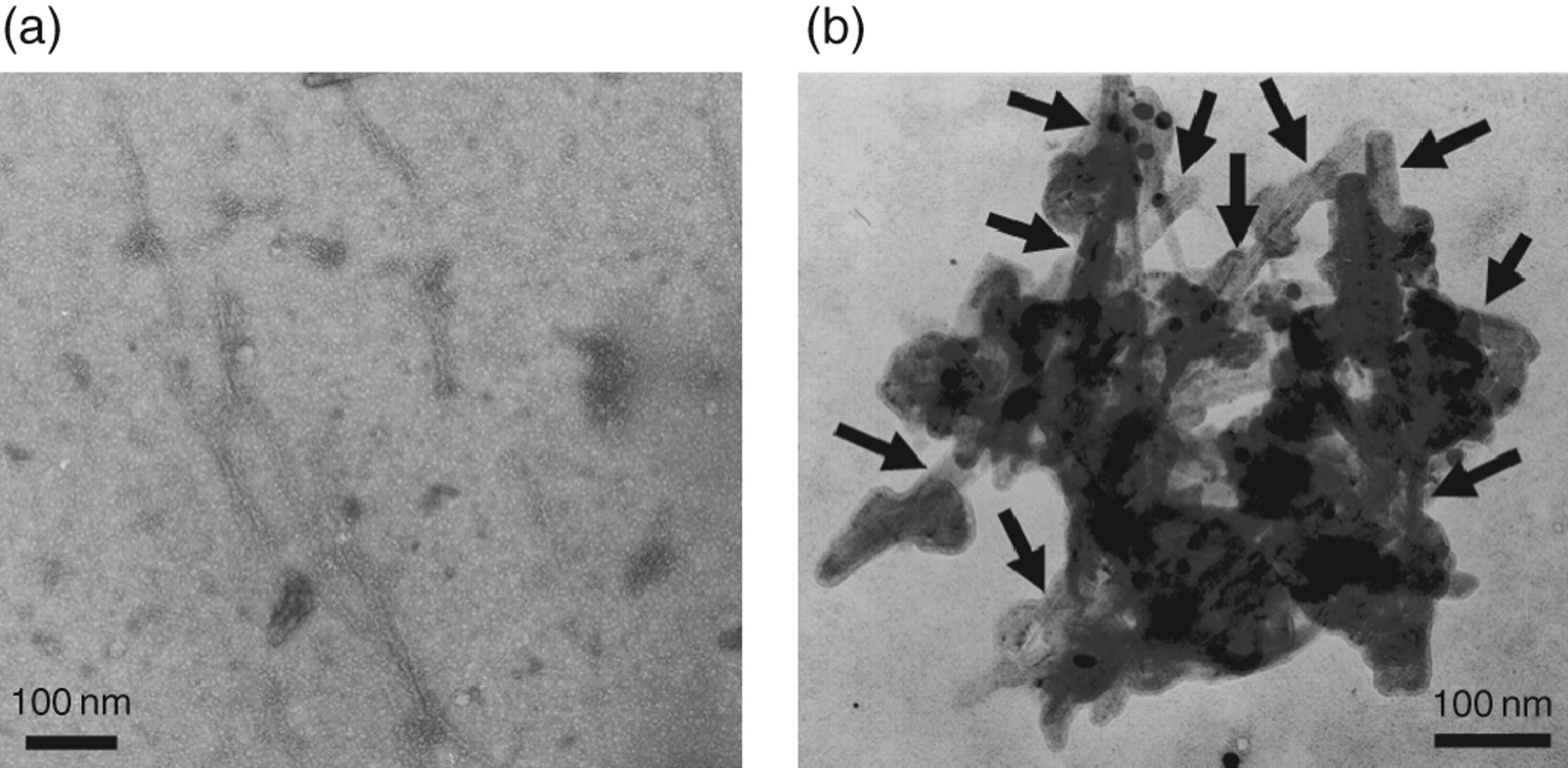
Negative-stain electron microscopic images of amyloid fibrils of β2m Arg97Ala incubated with seeds in 50 mmol/
Discussion
In this study, we demonstrated that the recombinant β2m Trp95Leu and Arg97Ala, with conformational changes in the C-terminus, formed more amyloid fibrils than did β2m WT. With ultrasonication to enhance destabilization of the protein and amyloidogenicity, β2m Trp95Leu and Arg97Ala generated amyloid fibrils even at physiological pH. Results of the present study provided direct evidence that C-terminal amino acids of β2m may play an important role in amyloid fibril formation.
Conformational analysis of β2m revealed that the Trp95 of β2m is hydrophobic and the most important residue in the C-terminus of β2m for stability of this globular protein. In our present study, we attempted to produce recombinant β2m with a mutation at Arg97 or Trp95. We were able to purify β2m WT, β2m Trp95Leu and β2m Arg97Ala. In contrast, the other four types of recombinant β2m with an amino acid substitution at the Trp95 residue formed aggregates and could not be purified. These results clearly suggest that Trp of β2m at position 95 may play a critical role in the stability of the molecule by influencing folding of the native form, and that β2m with those mutations at this amino acid may not be able to maintain stability.
To investigate conformational changes of β2m Trp95Leu and β2m Arg97Ala, we utilized CD and Trp fluorescence spectrum analyses. In the far-UV CD spectra, a protein with well-defined β-sheets shows a negative peak at 220 nm. 25 Our data indicated that the signal intensity of the β2m Trp95Leu and Arg97Ala spectrum decreased compared with that of β2m WT, although the β2m Trp95Leu and Arg97Ala spectra had negative peaks at about 220 nm. These data suggest that β2m Trp95Leu and Arg97Ala may possess a different conformation, and as the result, it may reduce β-sheet content because of the change of the C-terminal amino acids. The positive peaks of the Trp fluorescence spectrum for β2m Trp95Leu and Arg97Ala shifted from 334 to 349 and 342 nm, respectively, which indicates conformational changes around the Trp residues. In general, the red shift and decrease in the peak fluorescence intensity indicate the exposure of Trp residues. β2M has two Trp residues at positions 60 and 95. In native β2m, Trp60 connects the D and E strands and is exposed to the outside of the molecule from the beginning, whereas Trp95 lies initially in the middle of the molecule. 26 Thus, the change in the Trp fluorescence spectrum (Figure 2b) may indicate the exposure of Trp95, the partial unfolding of the C-terminus.
Other researchers have also elucidated various aspects of the mechanisms of amyloid fibril formation. McParland et al. 27 reported that an unfolding of both the C-terminal and N-terminal regions of β2m is a crucial step in the initiation phase of amyloid fibril formation. Hoshino et al. 28 reported that the hydrogen molecule in the A and G strands is most likely to be exchanged with deuterium. Stoppini et al. 8 reported that a monoclonal antibody specific for C-terminal peptides could inhibit β2m amyloid formation. We also previously reported that a monoclonal antibody against the C-terminal residues of β2m at positions 92–99 could detect the conformational intermediate of β2m in amyloid deposits obtained from DRA patients. 14
Our results clearly indicated that β2m Trp95Leu and Arg97Ala were unstable and had a greater amyloidogenic potential than did β2m WT, although mutated β2m had previously been investigated along with 12 other mutated types of β2m and the authors concluded that β2m Arg97Ala had little effect on protein stability. 25,29 Structural prediction suggests that β2m Arg97Ala cannot maintain the hydrogen bond with both Asn17 and Thr73, and thus hydrophobicity of the G strand is reduced (data not shown). This destabilization of the C-terminus may expose the G strand to the outside of the molecule and may produce a state of the β2m that allows assembly of amyloid fibrils.
In this study, we also demonstrated that ultrasonication enhanced amyloid formation of β2m at acidic and physiological pH. With ultrasonication, β2m Trp95Leu and Arg97Ala generated more amyloid fibrils than did β2m WT; similar relationship of the result on the two proteins was also obtained without ultrasonication. These findings confirmed that β2m Trp95Leu and Arg97Ala had more amyloidogenic potential than did β2m WT. Stathopulos et al. 18 previously reported that ultrasonication induced amyloid fibril formation in various proteins. More recently, Ohhashi et al. 30 demonstrated that ultrasonication enhanced WT-β2m amyloid fibril formation. Ultrasonication produces gas bubbles, high temperature and mechanical forces. 31 Those factors may induce destabilization of β2m and may thus enhance amyloid fibril formation.
For the diagnosis of prion diseases, one of the amyloid forming disorders, ultrasonication was employed in PMCA in which cycles of ultrasonication broke small amount of prion fibrils into smaller pieces acting as seeds for the formation of new fibrils from soluble prion protein. 19 By the same token, using ultrasonication, we could make the diagnosis for β2m amyloidosis earlier, if we could amplify and detect small amount of β2m fibrils in the early stage of DRA patients. Further investigations are needed.
It is interesting that ThT fluorescence intensity gradually decreased with or without ultrasonication after 6 and 12 hours, respectively. At 24 hours, when the ThT fluorescence intensity of β2m was three-fold lower than that at six hours, EM images of β2m incubated with ultrasonication showed amorphous debris containing long, straight fibrils, that is, typical amyloid fibrils (Figure 6b). This debris was not observed at six hours (Figure 6a). These results suggest that ThT may have difficulty to bind amyloid fibrils in debris and that the decreasing ThT fluorescence intensity was caused simply by this binding difficulty.
In conclusion, conformational changes in the C-terminus of β2m may play an important role in amyloid fibril formation of DRA. Ultrasonication is a useful technique to enhance β2m amyloid fibril formation even in the physiological solution.
Footnotes
ACKNOWLEDGEMENTS
This work is supported by the Japan Society for the Promotion of Science, the Amyloidosis Research Committee, the Pathogenesis, Therapy of Hereditary Neuropathy Research Committee, the Surveys and Research on Specific Disease, the Ministry of Health and Welfare of Japan, Charitable Trust Clinical Pathology Research Foundation of Japan and Grants-in-Aid for Scientific Research (B) 17390254 from the Ministry of Education, Science, Sports and Culture of Japan.
