Abstract
Background
The analytical performance characteristics of an enzyme-linked immunosorbent assay for the receptor for advanced glycation end-products (RAGE) were evaluated. The within- and between-subject components of biological variation were also estimated.
Methods
Blood was sampled from healthy volunteers into K2-ethylenediamine tetraacetic acid (EDTA) and serum separator tubes (SST) and the stability of RAGE in whole blood, plasma and serum examined. Performance characteristics of the assay were assessed using quality control materials. Three samples were obtained from each of 21 healthy volunteers one-week apart, RAGE measured and components of biological variation estimated.
Results
RAGE concentrations in blood specimens collected into K2-EDTA and SST were stable for at least 6 hours and, after centrifugation, both plasma and serum were stable for at least 24 hours. The RAGE assay had the following characteristics: inter-assay imprecision: coefficient of variation ≤7.3%, working range: 26–5000 ng/L, linearity: r = 0.9977 and detection limit: 26 ng/L. Overall within- and between-subject biological variations were 14.6% and 56.5%, the index of individuality was 0.31 and the reference change value was 49.0% at P < 0.05.
Conclusion
Samples for RAGE analyses in serum or plasma can be collected without significant difficulties with the assay showing acceptable analytical performance characteristics. Conventional population-based reference values are of limited utility in diagnosis, indicating that RAGE is likely to be more useful in monitoring disease.
Introduction
Microvascular changes in diabetes mellitus may be accelerated by formation of advanced glycation end-products (AGE) 1 that subsequently bind to the receptor for advanced glycation end-products (RAGE). 2 Studies have shown elevated RAGE expression on peripheral blood monocytes in non-diabetic patients with chronic kidney disease (CKD), 3 and more recently, increasing serum RAGE in patients with type 2 diabetes mellitus as CKD progresses. 4 Future research is required to assess whether RAGE provides an improved indicator of disease progression or severity. Our aims were to assess the characteristics of a commercially available RAGE enzyme-linked immunosorbent assay (ELISA) method and to estimate and apply the components of biological variation of RAGE. 5
Subjects and methods
A series of venous blood samples was taken from each of five apparently healthy volunteers into K2-ethylenediamine tetraacetic acid (EDTA) and serum separator tubes (SST) (Becton Dickinson, Oxford, UK). Samples were allowed to stand at room temperature for 30 minutes, 2, 4 and 6 hours before subsequent centrifugation and analysis. Two further specimens were sampled from each volunteer; one into K2-EDTA and the other into SST. Both were centrifuged at 30 minutes and aliquots were removed after 30 minutes, 2, 4, 6 and 24 hours following centrifugation. Stability of RAGE was investigated using a one-way analysis of variance to compare the plasma and serum RAGE concentrations at different time-points. A student’s unpaired t-test was used to compare the differences in mean concentrations between plasma and serum. All statistics were carried out using GraphPad Prism, version 3, (GraphPad Software Inc. San Diego, USA). P < 0.05 was considered significant.
Inter-assay imprecision for the RAGE ELISA (R&D Systems, Europe, Ltd., Abingdon, UK), was estimated by analysis of duplicates of three different quality control (QC) materials corresponding to low, medium and high RAGE concentrations, in 15 independent assays. Linearity was assessed by analysis of serial dilutions of three different patient samples with high RAGE concentrations (1:2, 1:4, 1:8, 1:16) and subsequent determination of corresponding final RAGE concentrations. Simple regression analysis was used to determine the correlation coefficient (r). Finally, detection limit of the assay was also assessed.
Three samples were obtained from each of 21 apparently healthy volunteers one week apart and RAGE concentrations measured to estimate the within- and between-subject components of biological variation. Procedures were adopted to minimize both preanalytical and analytical variation. Venous blood was sampled into SST at the same time of day each week, under the same conditions of venepuncture, by a single phlebotomist. Samples were centrifuged after 30 minutes and stored under the same conditions at −80°C until analysis by a single analyst. Data obtained were used to set quality specifications, determine the index of individuality and calculate the reference change value (RCV).
Results and discussion
The concentrations of RAGE in each of the whole blood samples showed no significant differences (P > 0.05) over the time period studied for both plasma and serum after centrifugation, indicating that RAGE concentrations remain stable in whole blood for up to 6 hours. Furthermore, there were no significant differences between plasma and serum, so that either specimen type could be used in routine practice. Analysis of RAGE in plasma and serum post centrifugation showed no significant differences in RAGE concentrations (P > 0.05): RAGE remains stable in plasma and serum for a period of at least 24 hours.
Inter-assay imprecision (coefficient of variation, CV%) was relatively uniform across the analytical range: 7.3%, 5.7%, and 5.6% at RAGE concentrations of 684, 1838 and 3276 ng/L, respectively. These results represent commercially prepared QC materials and some work in the future to assess whether sample matrix affects imprecision would be desirable. Linearity studies on three samples gave r = 0.9977, 0.9875 and 0.9873: the excellent linear correlation demonstrated supports the validity of sample dilution for those samples outside the working range of the assay. Zero standard replicates ranged from 12 to 26 ng/L, mean = 18 ng/L, and standard deviation = 4 ng/L setting the analytical detection limit as 26 ng/L.
Overall within- (CVI) and between-subject (CVG) biological variations were 14.6% and 56.5%. Mean serum RAGE concentration was not significantly different between genders. Comparison of CVI in the population studied and the two groups stratified according to gender showed no significant differences. CVG was significantly higher in females compared with males (Figure 1).
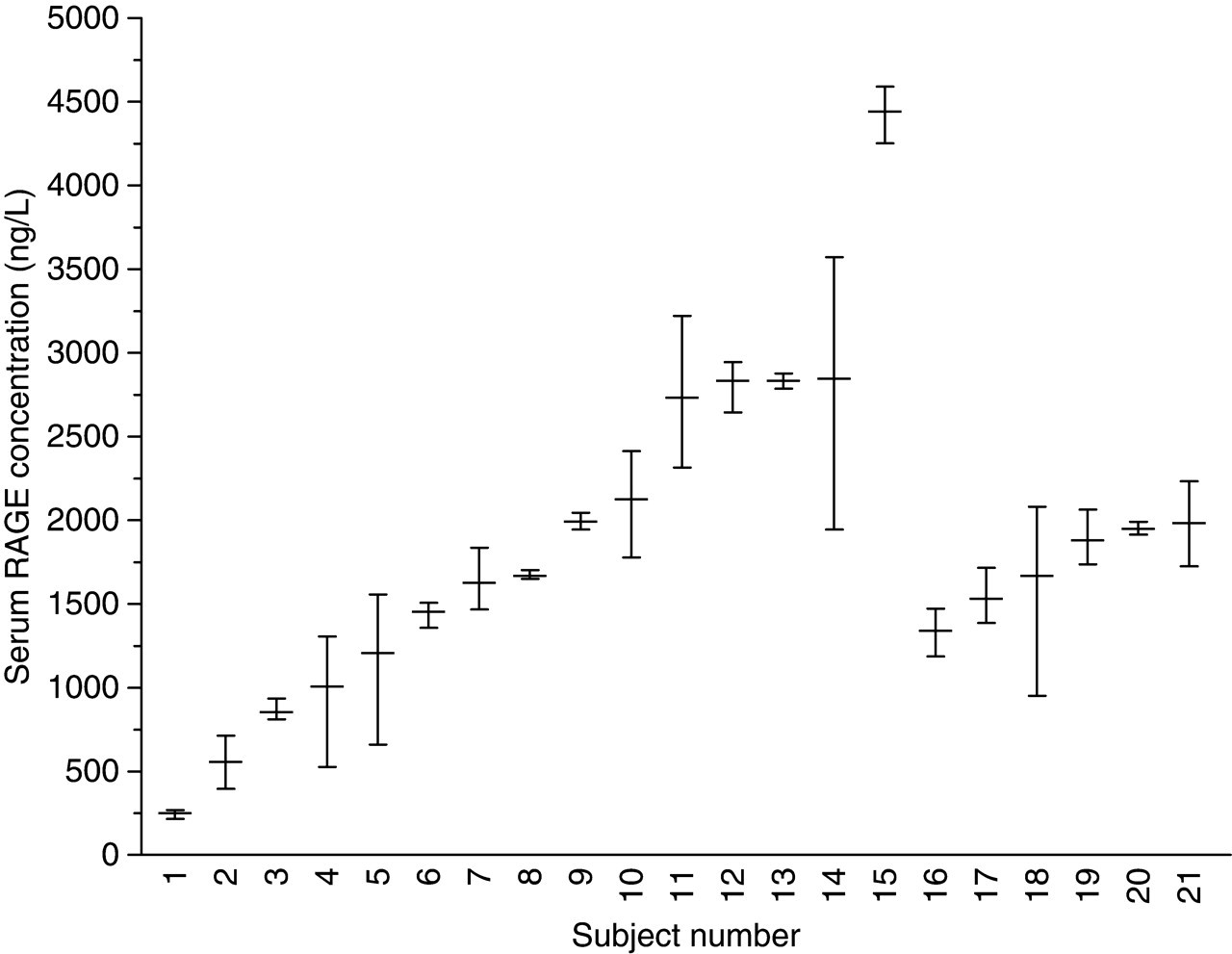
Variation (mean and absolute range) of serum receptor for advanced glycation end-products in three specimens collected from each of 21 healthy subjects over a three-week period. Female: 1–15, male: 16–21
For the whole group, the, quality specification for analytical imprecision was calculated as CVA < 7.3%. When this desirable assay performance is achieved (CVA < 0.50 CVI), the overall variability of every test result will not be confounded significantly by the analytical component of the test result variability, thereby improving overall reliability of the results. The observed inter-assay imprecision meets the quality specification. This is important with respect to monitoring patients over time since minimizing analytical variation results in a smaller RCV.
The index of individuality overall was 0.31, indicating that serum RAGE has marked individuality and that conventional population-based reference values would be of little utility in identification of unusual results or in assessing the significance of changes in serial results. However, quantities with low index of individuality are of significant value in monitoring disease if RCV are used. 6
Calculation of RCV for a significant (P < 0.05) change in serial results in an individual gave RCV of 49.0% (50.7% in males and 48.3% in females). Relatively large differences between the results of sequential samples are required for them to be significantly different. However, use of RCV, as opposed to conventional reference values, ensures a significant difference has occurred in serial results prior to clinical decisions being made.
RAGE assays could become part of the repertoire of the clinical laboratory as specimen collection, transport and handling pose few problems. Moreover, a simple RAGE assay, easily performed with standard ELISA methodology, has acceptable imprecision, working range, linearity and detection limit. Quality specifications for the desirable imprecision of RAGE have shown these are easily surpassed with this methodology. Data on biological variation suggest the more useful role for RAGE measurement would be in monitoring of disease, given the marked individuality. Such monitoring should be performed using RCV.
