Abstract
Background
Faeces must be sampled directly onto guaiac-based faecal occult blood test (FOBT) cards since analysis of specimens collected in traditional faeces containers is inappropriate because degradation of haemoglobin continues after faeces have been passed. Newer faecal immunochemical tests (FIT) are replacing FOBT, but it is likely that the practice of obtaining specimens in traditional faeces collection containers for later analysis will continue. The aim of this study was to assess the effect of delay in stool sampling on FIT.
Methods
Five specimens of faeces from healthy volunteers, all qualitatively FIT negative, were supplemented with whole blood haemolysate to three different FIT positive concentrations. Each sample was analysed daily after 1–14 days delay using a quantitative latex immunoturbidimetric-based FIT and also after five and ten days delay using a qualitative FIT.
Results
Haemoglobin concentrations fell each day, the rate being generally proportional to the original haemoglobin concentration. After eight days delay, no sample had a haemoglobin concentration >100 ng/mL and, after nine days, no sample had a haemoglobin concentration >50 ng/mL. After five days delay, five of the 15 supplemented faeces with initially positive qualitative FIT had negative FIT; after 10 days, none had positive FIT.
Conclusion
False-negative results will occur if sampling of fresh faeces into or onto FIT collection devices is delayed. Laboratories that undertake FIT analyses on faeces collected into traditional containers are likely to miss significant neoplasia. FIT collection devices must be used for sampling fresh faeces.
Introduction
There is evidence that degradation of haemoglobin continues after faeces have been passed. 1 This has relevance for traditional guaiac-based faecal occult blood tests (FOBT), which detect the pseudoperoxidase activity of haem. Using samples from volunteers spiked with whole blood haemolysate, Young et al. 2 examined the effect of delayed versus immediate application of faecal samples onto FOBT, and showed that false-negative results would occur when sampling of faeces onto the test card was delayed: it was emphasized that FOBT should be prepared immediately after passing of faeces and transport of moist samples avoided. However, clinical laboratories do not follow these recommendations and still receive most samples for FOBT in the traditional faecal collection containers. 3
It has been suggested that FOBT be ceased in all clinical settings except screening programmes and also proposed that FOBT be replaced by faecal immunochemical tests (FIT), as they undoubtedly have many advantages and fewer problems in both performance and interpretation. 4 These are based upon a different principle to FOBT, in that FIT use monoclonal or polyclonal antibodies and detect intact human haemoglobin or very early degradation products. Since current practices might continue and FIT analyses performed in laboratories, wards, clinics and general practices on samples of faeces collected in the traditional faecal containers, the effect of delay in sampling on faecal haemoglobin concentration, as assessed with both quantitative and qualitative FIT, was investigated.
Methods
Five fresh specimens of faeces from apparently healthy volunteers were tested with hema-screen SPECIFIC (Immunostics Inc., Ocean, NJ, USA, supplied by Alpha Labs Ltd., Eastleigh, Hants, UK). After thorough mixing, one portion of each was saved; another was supplemented with whole blood lysate prepared by freezing and thawing a specimen of venous blood collected into K2-EDTA vacutainers (Becton Dickinson, Oxford, UK) until the qualitative FIT result was weak-positive. Then, approximately twice and four times this amount of lysate was added to portions of each to give medium- and strong-positive FIT results. All were stored at between 18–21°C.
Samples were taken from the native and supplemented specimens after 24 hours and then at the same time each day for a further 13 days (14 days in total) into hema-screen SPECIFIC sample preparation tubes; these tubes have an integral sampling ‘stick’ on the lid to facilitate the safe transfer of a small amount of faeces into the tube containing phosphate-buffered saline with 0.5% sodium azide, which stabilizes the haemoglobin present.
The samples were then assayed using two methods. First, haemoglobin concentration was quantitatively measured in the material in the hema-screen SPECIFIC sample preparation tube using latex turbidimetry with FOB Gold reagents, calibrators and controls on a SENTiFOB fully automated photometric analyser (Sentinel Diagnostics, Milan, Italy). Secondly, after five and 10 days delay, qualitative FIT were also performed using hema-screen SPECIFIC test cassettes, which use a sandwich dye conjugate immunoassay using immunochromatography.
Results
The five native faeces had haemoglobin concentrations of 14.4, 14.4, 14.9, 30.1 and 33.2 ng/mL: all were negative by qualitative FIT analysis. The supplemented faeces had haemoglobin concentrations between 101.6 ng/mL and 2089.2 ng/mL, and all were positive by qualitative FIT. Weak-positive samples had mean haemoglobin concentration of 314.7 ng/mL, medium-positive had 561.6 ng/mL and strong-positive had 1430.5 ng/mL.
The haemoglobin concentration in the faeces fell with time. The rate of decrease was variable from one sample to the other, but generally proportional to the amount of haemoglobin as shown in Figure 1 for the series of faeces from one of the volunteers: all showed similar patterns. Mean haemoglobin (% of original concentration) in supplemented faeces (SEM) fell from 100% to 82.5% (5.7%), 60.3% (5.3%), 55.7% (6.0%), 46.5% (5.0%), 20.0% (3.1%), 14.2% (2.4%), 12.9% (2.6%), 8.1% (1.7%), 5.7% (2.0%), 7.3% (2.3%), 6.9% (1.9%), 5.7% (1.4%), 4.5% (1.3%) and 3.2% (0.9%) after 1–14 days delay.
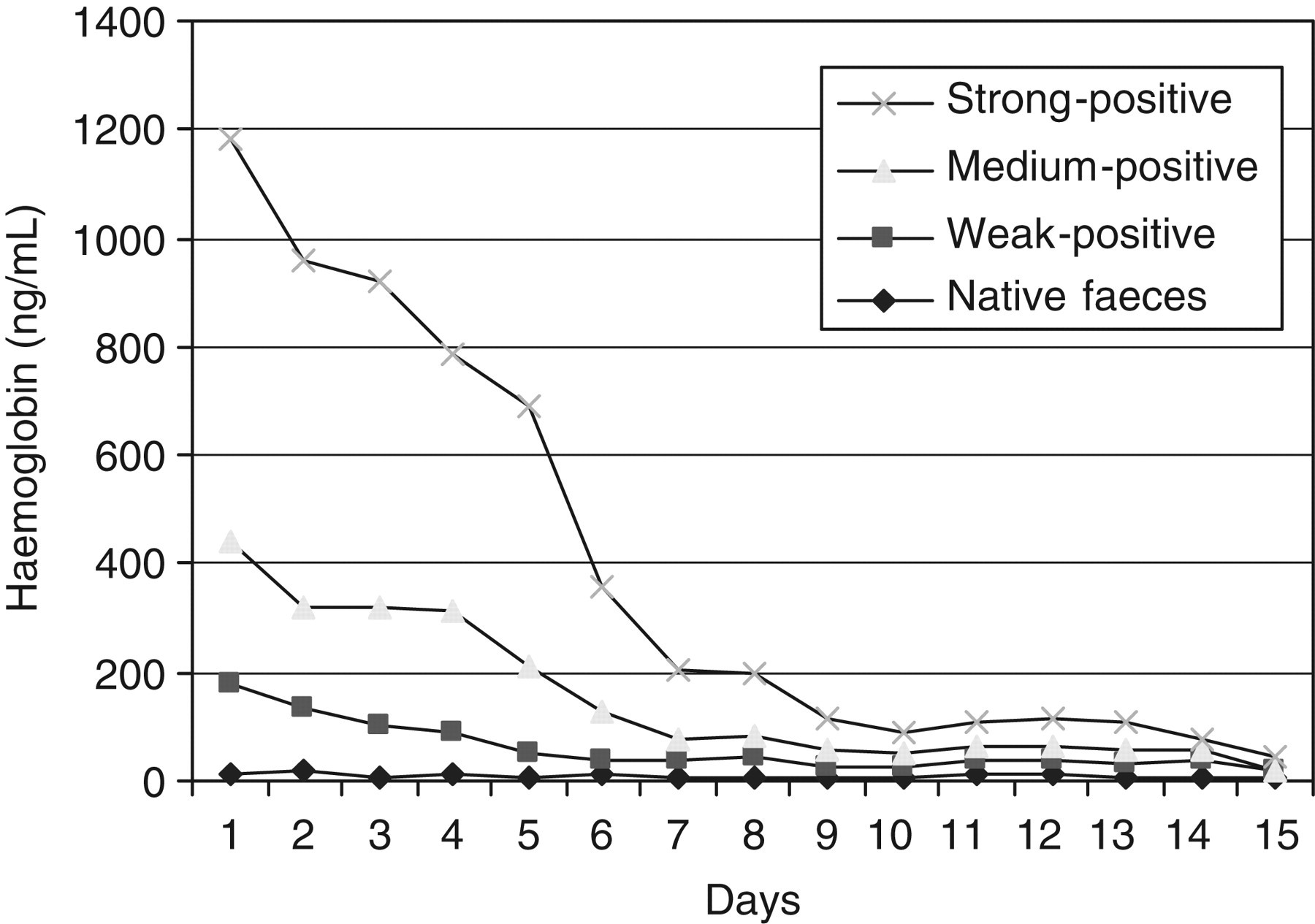
Haemoglobin concentration (ng/mL) in native faeces and faeces supplemented with whole blood lysate to give weak, medium and strong qualitative faecal immunochemical test results
Faecal specimens with haemoglobin near to the cut-off concentration became negative after short periods of delay. For example, the haemoglobin concentration in the weak-positive samples fell below 50 ng/mL after three to six days. After eight days, no sample had a haemoglobin concentration >100 ng/mL, a commonly used cut-off concentration for the declaration of positive or negative results in quantitative FIT analysis. 5 After nine days, no sample had a haemoglobin concentration >50 ng/mL, which is the usual cut-off concentration of commercially available qualitative FIT such as that used in the Scottish Bowel Screening Programme. 6
To verify these results with a different FIT, the samples were assayed with the qualitative FIT after five days, when five of the 15 supplemented faeces now had negative FIT, and after 10 days, when none had positive FIT.
Discussion
In conclusion, false-negative results for faecal haemoglobin will occur if sampling of fresh faeces into the tubes or onto the cards of FIT collection devices is delayed. Clinical laboratories and other sites that undertake FIT analyses on faeces collected into traditional containers (e.g. ‘pots’ with integral spoons in the lids) are likely to miss significant neoplasia. This will lead to adverse outcomes for patients since the earlier the diagnosis of colorectal (bowel) cancer is made, the better the prognosis. The actual collection devices supplied with the particular FIT must be used for sampling fresh faeces.
Footnotes
ACKNOWLEDGEMENTS
We thank Sentinel Diagnostics for the loan of the SENTiFOB analyser and Alpha Labs Ltd. for the loan of pipettes. NHS Tayside has a consultancy agreement with Immunostics Inc.
