Abstract
Background
Research has demonstrated that most laboratory errors occur in the preanalytical phase of testing. In view of the paucity of studies examining preanalytical errors, we evaluated our laboratory request forms for the frequency and impact of incomplete data.
Methods
This study examined all request forms received at our laboratory during a five-day period. The forms were scrutinized for the presence of specific parameters. The impact of abbreviated diagnoses was analysed, as well as how lack of ward or telephone details affects the communication of critical results to clinicians.
Results
A total of 2550 request forms were analysed. Medication(s) used by the patient (89.6%) and doctor’s contact number (61.2%) were the most incomplete parameters. No diagnosis was provided on 19.1% of forms, and when a diagnosis was present it was an abbreviated form in 37.3%. This resulted in 35.5% of diagnoses not being recorded by reception staff. Incomplete ward information was found on 4.9% of forms. In a separate search, the impact of 151 request forms (collected over a period of eight months), with incomplete ward location information and corresponding to critical results was assessed. Critical results were not communicated by telephone to clinicians in 19.9% of cases.
Conclusion
As laboratory data influences 70% of medical diagnoses, incorrect or incomplete data provided to the laboratory could significantly impact the success and cost of overall treatment.
Introduction
Historically, laboratory quality was determined by the accuracy of the analytical phase. However, following the development of high-quality analytical techniques, analytical error is no longer the main reason for error in the laboratory testing process. 1 Furthermore, research has demonstrated that 68.2% of laboratory errors occur in the preanalytical phase. 2 This is primarily a result of the lack of standardized protocols for defining and measuring preanalytical variables. 3
Insufficient, incorrect or illegible data on laboratory request forms can delay communication of important clinical information, such as life-threatening results, and cause resource wastage. A previous study has shown that 43% of request forms lacked complete information. The specific items of information that were missing included the requesting physician’s name or pager number. Often, there was a misidentification of both the patient and requested test(s). 4
The National Health Laboratories Service (NHLS) provides laboratory services to hospitals in the public sector in the Republic of South Africa. In April 2007, the Department of Health in the Western Cape province of South Africa instructed all laboratories within the NHLS to comply with certain requirements regarding the processing of specimens accompanied by incomplete laboratory request forms. In accordance with the National Public Finance Management Act (Act 1 of 1999), which governs all procurement transactions of government, the following mandatory information on request forms is required before laboratories will be reimbursed: patient identification by name as well as hospital identification number, hospital ward or cost centre and the name of the clinician or nurse authorized to request the test(s).
We reviewed the compliance of laboratory request forms received in the Chemical Pathology laboratory in a large tertiary hospital with the above-mentioned requirements. We also analysed separately the impact of the absence of ward data on forms in the communication of critical Chemical Pathology results to requesting clinicians.
Materials and methods
This was a retrospective study conducted by the Division of Chemical Pathology, NHLS, Tygerberg Hospital. Tygerberg Hospital is an academic, tertiary hospital in the Western Cape province of South Africa. The Chemical Pathology laboratory receives specimens from both in- and outpatient sections of the hospital.
Original laboratory request forms received at the Chemical Pathology division during a five-day period from 16 to 20 July 2007 were manually scrutinized for the presence of parameters provided in Box 1. For the purpose of this study, we assessed requests for routine core Clinical Biochemistry automated tests. Request forms received from the HIV-rollout clinic use a different request form and were therefore excluded, as were forms requesting only manual tests.
Preanalytical quality indicators examined on Chemical Pathology request forms
Identification by name and surname
Identification by hospital number
Date of birth
Gender
Ward/location where patient resides
Requesting physician’s identification
Requesting physician’s contact number/pager number
Clinical/diagnostic information
Diagnosis present in an abbreviated form
Medications that the patient is/was taking at the time of specimen collection
Identification of specimen
Date of specimen collection
Time of specimen collection
Illegible handwriting
In addition, 200 randomly selected laboratory forms where the diagnosis was provided in an abbreviated form were also scrutinized to determine whether the diagnosis had been recorded correctly by the reception staff.
The impact of incomplete forms was evaluated by performing a separate search and assessing all (n = 151) critical Chemical Pathology results with absent or incomplete ward data on request forms. The request forms were all received at the laboratory from 1 January to 31 August 2007. Critical results can be defined as results suggesting that the patient is in imminent danger, unless appropriate therapy is initiated promptly. 5 Conformance with good laboratory practice demand all results above critical limits to be telephoned to the respective ward or clinician to facilitate prompt and appropriate action.
All data were captured on Excel worksheets and patient confidentiality was maintained. No patient identifying information (name, hospital identification number) was included on the data record sheet and patients were identified by a study number only. Basic statistics on the Microsoft Excel software programme were used for data analysis.
The study was approved by the Ethics Committee of the University of Stellenbosch and performed according to the Helsinki Declaration (2000).
Results
During the study period, 2550 request forms were received. The results obtained are shown in Table 1.
Absence of parameters on laboratory request forms (n = 2550)
Patient and clinician information
No clinician’s name was entered on 189 (7.4%) forms and 1560 (61.2%) had no contact telephone or pager number. Both the clinician’s name and contact telephone or pager number was missing on 167 (6.5%) of the forms.
Gender detail was not recorded in 129 (5.1%), whereas 124 (4.9%) forms had no ward information. Twenty forms (0.8%) lacked both ward and requesting clinician information (name as well as pager number of the clinician). The date of birth was not recorded on 97 (3.7%) forms. Four forms (0.2%) had no patient names and seven (0.3%) had no hospital identification numbers. Only one form lacked both patient name and hospital identification number.
Clinical information
A diagnosis was not indicated on 487 (19.1%) forms, 2283 (89.6%) had no details of therapy, while 4% of all forms were illegible. Of the 2063 (80.9%) forms where a diagnosis was present, 770 (37.3%) had the diagnosis written in an abbreviated form.
We analysed separately 200 request forms with abbreviated diagnoses. In 69 (34.5%) of these the diagnosis was recorded in the abbreviated form; in 51 (25.5%) the abbreviation was correctly interpreted and recorded and in 71 (35.5%) no diagnosis was recorded by the reception staff. Nine (4.5%) abbreviations were misinterpreted and incorrectly recorded.
Specimen information
Two-hundred and eighty (11%) request forms did not specify the type of specimen collected, 83 (3.3%) did not state the date and 390 (15.3%) did not state time of collection.
Figure 1 shows the results obtained when 151 critical Chemical Pathology results without ward information on request forms were analysed. Of these critical results, 30 (19.9%) were not telephoned to the respective ward or clinician, because of the absence of ward data on the forms. It was possible to communicate the other 121 critical results after much effort in tracing the patient. This involved examining previous results on the same patient to ascertain a ward or clinician contact number, paging the attending clinician, whether this information was available and phoning hospital inquiries to determine the location of the patient. Most of the critical values not communicated were elevated potassium concentrations (n = 17; 57%). Of these, 11/17 (65%) had no signs of haemolysis. The highest non-haemolyzed potassium value was 8.7 mmol/L. Six (20%) of the 30 critical results not telephoned lacked both ward location and requesting clinician details on the associated request form.
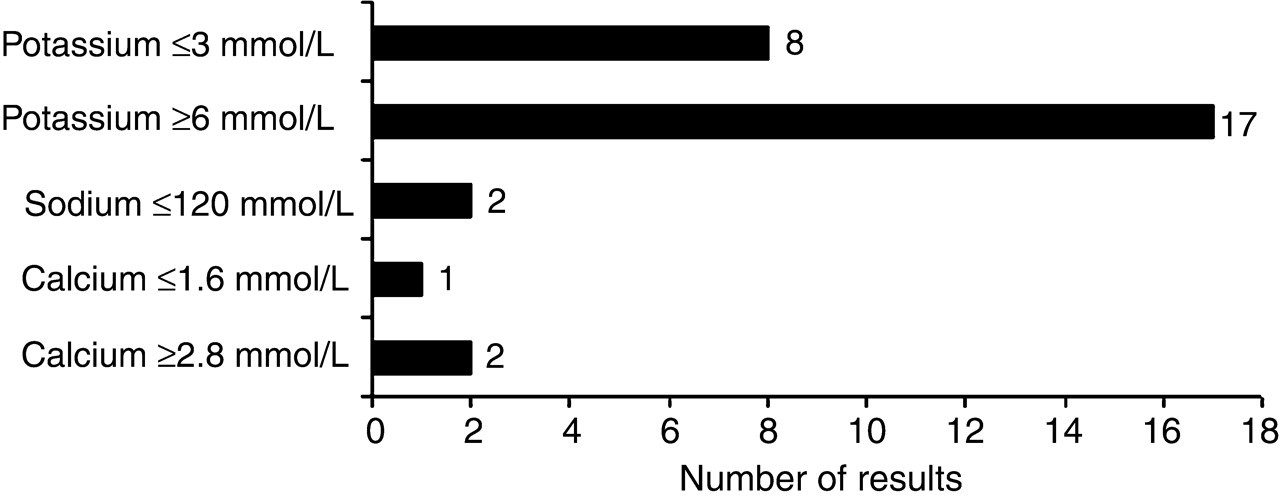
Critical results not phoned out because of absence of ward data on laboratory request forms (n = 30)
Discussion
Very few studies have examined the frequency and impact of incomplete laboratory request forms. Laboratory errors are of utmost importance, as laboratory data influence 70% of medical diagnoses and can impact significantly on the success and cost of patient treatment. 6 Previous studies have shown that the analytical error rate has decreased significantly over time as a result of better training and qualification of staff, better rules for detecting the errors early in internal quality control programmes, more effective external quality assessment schemes and improvement in analytical techniques. 1,3 Plebani and Carraro 2 ascribed 68.2% of errors in their laboratory to the preanalytical phase. Plebani 7 subsequently emphasized the importance of errors in the pre-preanalytical phase of testing (i.e. procedures not performed in the clinical laboratory or not under the control of laboratory personnel) and the need to reduce them.
These findings have led to an agreement on the definition of a laboratory error as a defect occurring at any part of the laboratory cycle 8 and this definition has been incorporated in the ISO Technical Report 22367. 9 There is a paucity of literature evaluating the preanalytical phase of laboratory testing, as highlighted in a recent review on quality in the extra-analytical phases in clinical laboratory management by Ricós et al. 10
Our hospital does not operate ward-based electronic requesting 4 and therefore processes completed request forms manually, following bar coding on arrival at the laboratory. A previous study has shown that manually completed forms can lead to insufficient, incorrect or illegible data on request forms. 4 Incomplete forms also impact on other divisions of Pathology, as they may adversely affect not only the diagnostic processing, but also infection control surveillance (e.g. Microbiology). 11 Incomplete forms may not only complicate billing purposes, but can also affect the interpretation of results by pathologists and make the communication of critical laboratory results to the physician or ward difficult.
Previous studies have shown that if laboratory forms are improved and made more user friendly, errors and inappropriate testing may be reduced. 12–15 The layout of our forms was updated in 2003 and is in our opinion simple to use.
A drug history and provisional diagnosis were found to be incomplete on the majority of request forms. Many drugs can affect the interpretation of results and some may even interfere with assays. 16,17 In some instances, the correct interpretation of results may depend upon the provisional diagnosis indicated on the request form. For example, in those instances where a tumour marker result is falsely low because of a high-dose hook effect.
The use of abbreviated diagnoses was prevalent in our study and resulted in errors in the data recording process. Clinicians should be aware that even commonly used medical abbreviations may be misinterpreted, as the data input staff may not be familiar with the meaning of the abbreviations.
The time of specimen collection is important in therapeutic drug monitoring. The reference values for certain analytes also differ according to the time of day. The type of specimen obtained is important where bloody taps of other body fluids can be confused with blood and where it may result in the use of inappropriate reference ranges.
Where no ward information was provided on request forms, 30 (19.9%) critical results were not phoned out. Of this number, one-fifth had no physician’s name stated on the form. Our experience suggests that many physicians do not answer their pagers (when the number was stated on the request form) after hours when not on call. This complicates the task of the laboratory personnel when trying to communicate important information.
The Royal College of Pathologists and the Royal College of General Practitioners have become aware of the fact that laboratory staff are often unable to communicate life-threatening or severely abnormal results to primary care general practitioners after hours. Although this does not directly apply to our setting, it is of interest that they emphasize the need for correctly filled in laboratory forms. 18
Our results are probably not unique, although they may not reflect the experience of those laboratories that operate ward-based electronic requesting. In those instances, ward requesting can be set up so that the requests are not transmitted to the laboratory unless all appropriate data are provided by the requesting clinician. This will almost certainly lead to better quality information being received by the laboratory.
In conclusion, this study demonstrates that processing incomplete laboratory request forms may lead to the misinterpretation of results and have a serious impact on patient care. Although this study was limited to Chemical Pathology requests received within an academic tertiary hospital, it is likely that similar results would have been obtained in other settings where ward-based electronic requesting is not in place. One way to improve the quality of data provided with each request would be to introduce ward-based electronic requesting.
Footnotes
ACKNOWLEDGEMENTS
We thank Comfort Asoogo and Miriam Owusu Sekyere for their help in processing the forms. We would also like to thank Dr Mia le Riche and Dr Robert Wicomb for proofreading our manuscript.
