Abstract
Abstract
We report a case of IgG paraprotein cross-reactivity with IgM measurement in a patient with IgGκ multiple myeloma and associated IgA and IgM immune paresis. IgM measurements using a Beckman-Coulter Synchron LX IgM reagent initially gave unmeasurably low IgM concentrations (<0.3 g/L) but IgM quantitation using later batches of reagent gave IgM concentrations of 13–42 g/L. Immunofixation confirmed the continued presence of the IgGκ paraprotein band with suppression of IgA and IgM and analysis on the Behring BN2 and Beckman-Coulter Immage instruments showed IgM concentrations of <0.2 g/L. Immunofixation using the Synchron LX IgM reagent as antiserum confirmed binding of the Synchron LX IgM antiserum to the IgGκ paraprotein. This case highlights the importance of checking all unexpected immunoglobulin measurements with previous results and immunofixation findings to avoid mistakes.
Introduction
Paraprotein identification and immunoglobulin quantitation are key elements in the diagnosis and management of multiple myeloma. Immunoglobulin quantitation is usually performed using immunonephelometric and immunoturbidometric methods and requires the use of heavy chain-specific antisera to differentiate the immunoglobulin class. We present a case of IgGκ multiple myeloma in which a falsely elevated IgM concentration was reported due to IgG paraprotein cross-reactivity with the IgM immunoturbidometric reagent.
Case report
A 69-year-old patient was admitted in February 2006 with high output cardiac failure due to severe anaemia with a haemoglobin concentration of 4.8 g/dL. Multiple myeloma was subsequently diagnosed based on the presence of a serum IgGκ paraparotein (total IgG concentration of 78 g/L) and 65% bone marrow plasma cells infiltration. IgA and IgM concentrations were both noted to be suppressed (Table 1). No monoclonal free light chains were detected in either serum or urine immunofixation. Serum urea, creatinine and calcium concentrations were unremarkable, but the skeletal survey revealed compression fractures from T11 to L2 vertebrae. The β2-microglobulin and albumin concentrations were 6.4 mg/L and 27 g/L, respectively indicating stage III disease by the International Staging System for Multiple Myeloma. 1
Immunoglobulin measurements on samples A–F using Synchron LX reagents
*Run in Over Range Detection and Correction (ORDAC) mode
The patient was treated with monthly oral melphalan, prednisolone and monthly pamidronate infusions, and disease response was monitored by serial total IgG measurements. IgA and IgM concentrations were checked occasionally.
Partial remission was achieved after 12 courses with a nadir in total IgG concentration of 34 g/L. Treatment was discontinued, but within two months his IgG concentration increased to 42 g/L and he was recommenced on chemotherapy with melphalan, prednisolone and thalidomide.
It was noted that the IgM concentration had increased from <0.3 g/L to 22 g/L nine months after diagnosis. The patient had no symptoms or signs suggestive of other lymphoproliferative disorders and there was no population of cells with lymphoplasmacytoid morphology on review of the initial bone marrow aspirate.
Discussion
This report describes a case of IgGκ multiple myeloma with IgA and IgM immune suppression in which an unexpectedly raised IgM concentration was reported. The original electrophoresis and immunofixation on the first sample (sample A) had been performed on the Beckman-Coulter Paragon high resolution electrophoresis (HRE) and immunofixation electrophoresis (IFE) systems using manufacturer-supplied goat-derived antisera. Immunoglobulin quantitation by immunoturbidometry was performed on the Beckman-Coulter DXc-800 clinical chemistry analyser using Synchron LX reagents, which use goat-derived monospecific antisera. The laboratory routinely stores samples submitted for protein studies at –70°C for two years for follow-up work if required. Following doubts raised by the clinician concerning the reported IgM measurements, the two frozen specimens (samples A and D – samples B and C were unavailable) were thawed and re-run for immunoglobulin quantitation (see Table 1). Aliquots of sample E were also sent to neighbouring laboratories for immunoglobulin measurement using the Beckman-Coulter Immage and Behring BN2 analytical systems (see Table 2). Finally, immunofixation on sample E was performed (using the Paragon system) with an additional immunofixation lane using Synchron LX IgM Reagent (goat antibody monospecific for human IgM, cartridge B, lot 610303) as the antiserum (see Figure 1).
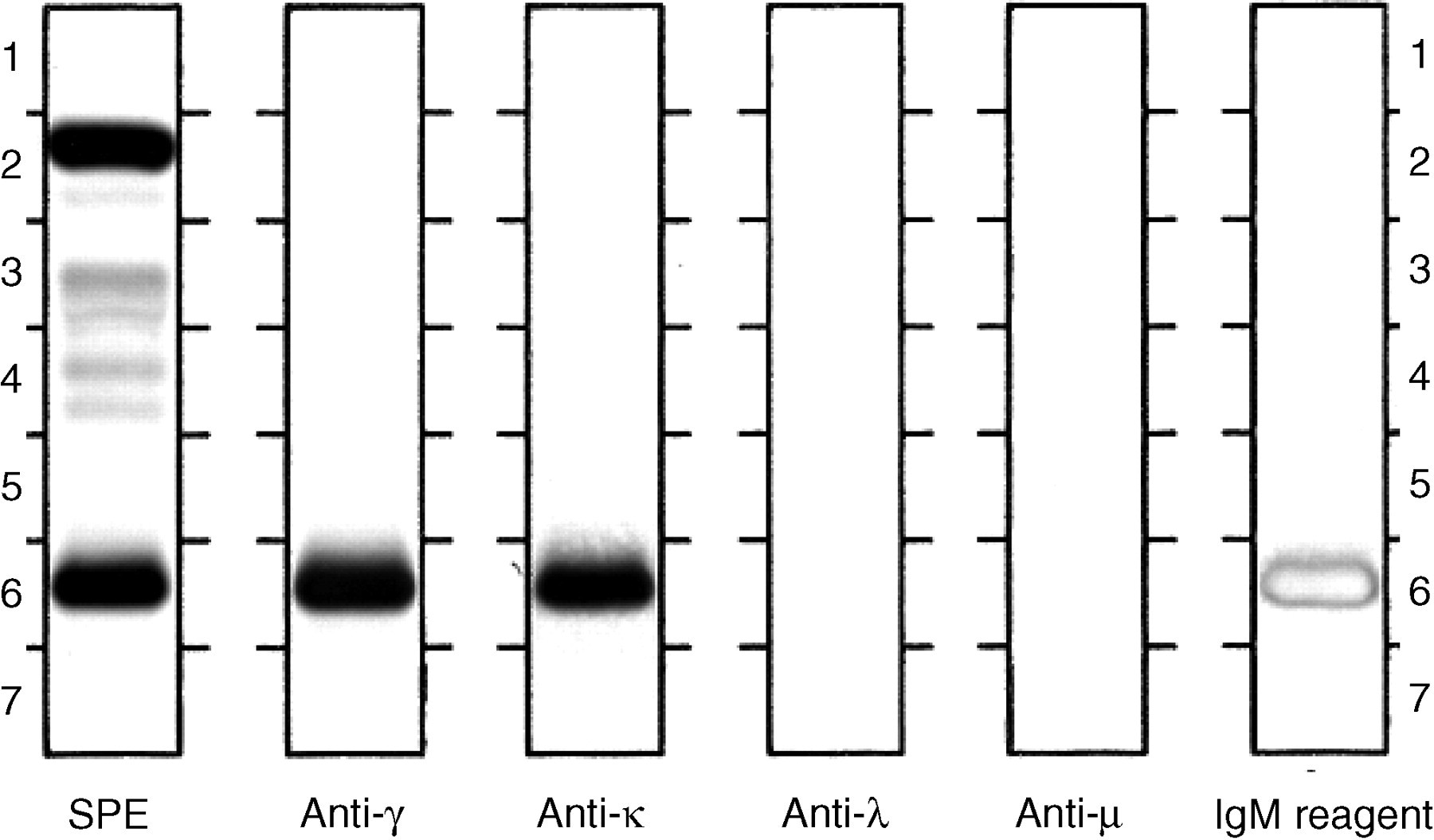
Immunofixation of sample E using the Paragon system and Paragon antisera (lanes 2–5) and Synchron LX IgM reagent (lot no 610303) as antiserum (lane 6)
Immunoglobulin measurements on sample E
The presence of a raised IgM concentration in an IgG myeloma patient with immune suppression was puzzling and warranted further study and explanation. A shift in immunoglobulin production from IgG to IgM under treatment was a biologically plausible explanation for this finding, with cases of switching from IgG to IgD and IgGλ to IgGκ reported. 2,3 Other possibilities include the presence of IgG and IgM paraproteins with common variable regions in the heavy chains or the development of disufide-bonded IgG–IgM fragment complexes. 4,5 However, the failure to identify any IgM paraprotein in either the original or follow-up immunofixation, the identical electrophoretic mobility of the ‘IgM’ band identified with the Synchron reagent with the original IgGκ paraprotein band and the low IgM concentrations measured using the Immage and BN2 assays points to an methodological rather than biological cause for this phenomenon. Three different lots of Synchron LX IgM reagent have given raised IgM measurements for samples from this patient (Table 1). Cross-reactivity of the IgM antiserum used in the Synchron assay with the patient's IgGκ paraprotein is the most likely explanation and is supported by the positive immunofixation staining at the position of the IgGκ paraprotein with use of the Synchron IgM reagent (Figure 1). There are reports of heavy chain antisera cross-reactivity with other serum proteins (fibrinogen, C3, C4 and transferrin), but few of cross-reactivity between different heavy chain antisera. 6,7 Reports generally describe antisera used for immunofixation studies rather than immunochemical-based immunoglobulin quantitation. Cases affecting immunoglobulin measurement could go unnoticed when immunoglobulin quantitation alone (without electrophoretic and immunofixation studies) is requested, especially in the absence of analytical error flags as in this report. This case highlights the importance of checking all unexpected immunoglobulin measurements with previous results and immunofixation findings to avoid mistakes.
