Abstract
Abstract
Background
A cardiac troponin concentration above the 99th percentile limit of a reference population is a sensitive marker of myocardial necrosis. Current guidelines require troponin assays to have a total imprecision of ≤10% at the 99th percentile limit. In this study, the Advia Centaur second-generation TnI-Ultra assay was validated and compared with its predecessor the cardiac troponin I (cTnI) assay, with a focus on the current guidelines for diagnosis of acute myocardial damage.
Methods
An imprecision profile of the TnI-Ultra assay was evaluated by analysing different pools over 20 days. The imprecision of the cTnI assay was used as comparison. The reference range was established based on TnI-Ultra analysis in 221 individuals.
Results
The cTnI concentration that could be determined with a total imprecision of 10% was 0.05 µg/L for the TnI-Ultra assay and 0.3 µg/L for the cTnI assay. The 99th percentile limit in the distribution of a reference population was 0.06 µg/L as determined with the TnI-Ultra assay.
Conclusions
The TnI-Ultra assay provides significantly improved sensitivity when compared with the cTnI assay and a total imprecision of ≤10% is obtained at the 99th percentile limit of value distribution of a reference population. Using the TnI-Ultra assay, slightly increased cTnI concentration can be detected reliably following the current guidelines.
Introduction
Cardiac troponins I (cTnI) and T (cTnT) are the preferred diagnostic markers of acute myocardial infarction (AMI). Guidelines of the Joint European Society of Cardiology (ESC) and the American College of Cardiology (ACC) Committee state that a troponin concentration exceeding the 99th percentile of a reference population is a specific biochemical marker of AMI. 1 The ESC/ACC guidelines require troponin assays to have a total imprecision of <10% at the 99th percentile level. 1 At the time of publication of the guidelines, no assays met this requirement. For the Advia Centaur cTnI assay, the concentration with a total CV of 10%, as determined in imprecision profiles, ranges from 0.33 to 0.35 µg/L with a 99th percentile value of 0.1 µg/L. 2,3 To meet the ESC/ACC requirements, a next generation of cardiac troponin assays with improved reproducibility and analytical sensitivity was developed.
In this study, the second-generation cTnI-assay for the Advia Centaur, the TnI-Ultra assay, was evaluated regarding imprecision at different cTnI concentrations and reference range. Its predecessor, the first-generation cTnI assay, was used for comparison.
Materials and methods
Subjects
For evaluation of imprecision at low cTnI concentrations, heparinized blood from 30 non-cardiac patients with undetectable troponin concentration was pooled and spiked with samples containing high amounts of troponin.
The reference range was established with heparinized blood samples from 221 non-hospitalized patients who were not referred to the laboratory by a cardiologist. It was confirmed by means of a questionnaire that the patients had no known history of myocardial disease. The patient group consisted of 110 men and 111 women with a median age of 58 years (range 14–86 years). Non-cardiac conditions associated with minor troponin increases were not specifically excluded to obtain a representative patient population for a hospital setting.
Subjects volunteering for the study were recruited according to the hospital guidelines and informed consent was obtained.
Pre-analysis
Blood samples were collected in vacuum polypropylene tubes containing lithium heparin (BD Vacutainer™, 4 mL, LH 68 IU Plus, BD Vacutainer Systems Preanalytical Solutions). Samples were centrifuged at 3000
Methods
The TnI-Ultra assay (Siemens Medical Solutions Diagnostics, Tarrytown, NY, USA) applies a three-site sandwich immunoassay technique, instead of the two-site sandwich technique of the cTnI assay. At the time of validation, three lots of TnI-Ultra reagent were available. Troponin assays were calibrated according to the manufacturer's instructions.
For determination of imprecision, two different protocols were used: a 20-day protocol performing two runs per day in duplicate and a 5-day protocol, performing one run per day in triplicate. Imprecision was calculated at five different concentrations. For the TnI-Ultra assay, the 20-day protocol was used at all concentrations. For the cTnI assay, the 20-day protocol was used for the middle three concentrations and the 5-day protocol for the lowest and highest concentrations. The non-parametric reference interval was evaluated for the TnI-Ultra assay. Two different lots of TnI-Ultra reagent were used and two calibrations were performed. Data were analysed with Analyze-It version 1.72 (Analyze-It Software Ltd, UK).
Results and discussion
Of the 221 outpatients, cTnI was lower than the limit of detection in 113 individuals. The non-parametric 95% reference interval was calculated as <0.0085 (below limit of detection) to 0.04 µg/L. The upper 99th percentile of the studied population was 0.06 µg/L. This value is at the high end of the range as reported by Siemens, with their product sleeve being 0.02–0.06 µg/L, depending upon sample type, instrument and reagent lot.
The results of the imprecision analysis are shown in Figure 1. The imprecision profile of TnI-Ultra shows ≤16.7% total imprecision at cTnI concentrations of ≥0.03 µg/L. The concentration which is measured with a total imprecision of 10% was estimated as 0.05 µg/L, which is in agreement with the 0.07 µg/L reported by Prontera et al. 4 The overall imprecision of the TnI-Ultra assay was clearly lower than the imprecision of the cTnI assay, which reached an imprecision of 10% at a troponin concentration of 0.3 µg/L.
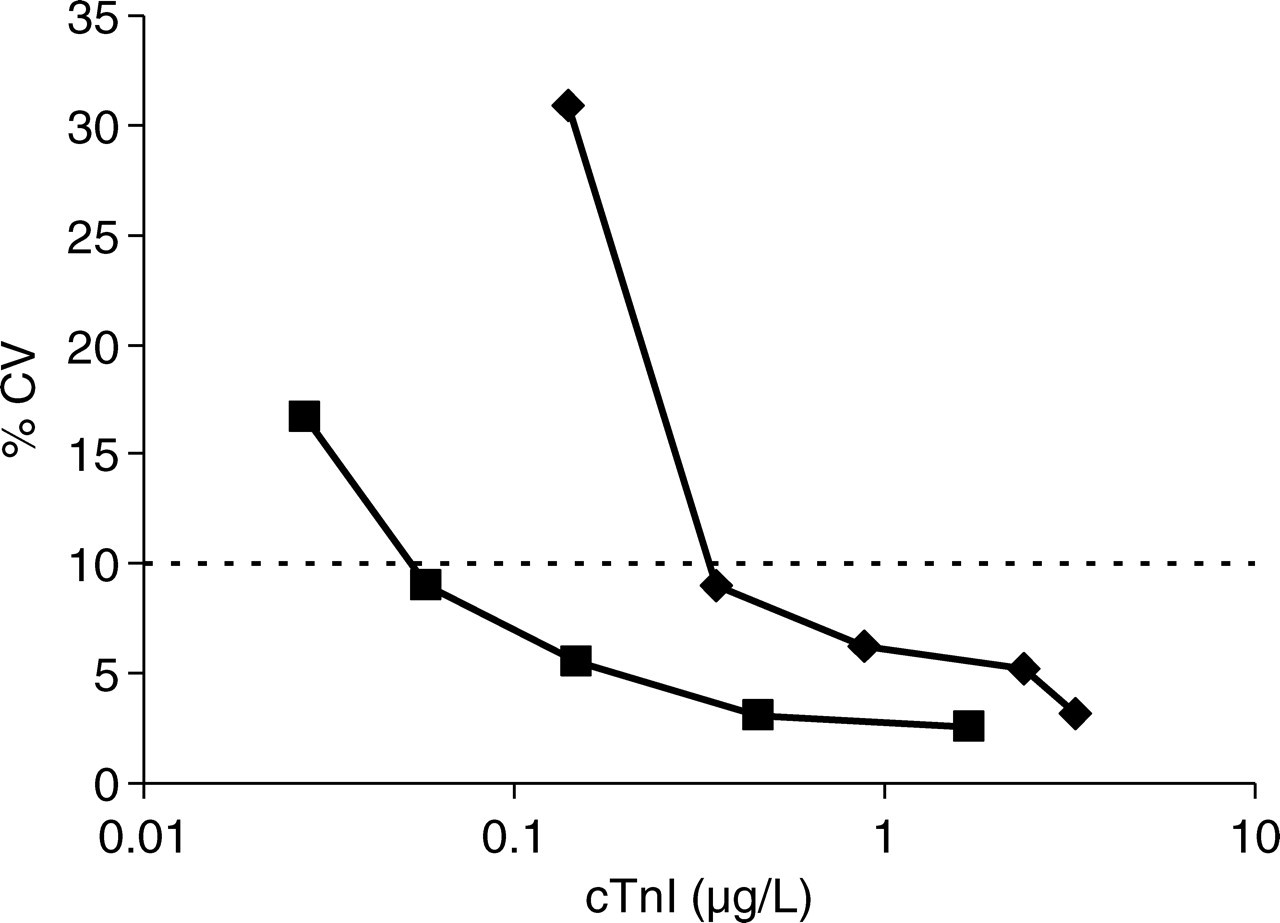
Comparison of the precision profiles of the TnI-Ultra assay (squares: 10% CV at 0.05 µg/L) and the cTnI assay (diamonds: 10% CV at 0.3 µg/L) in a semi-logarithmic graph. Dashed line indicates 10% total imprecision
In this study, the 99th percentile (0.06 µg/L) of a reference population could be determined with <10% CV. Thus, the TnI-Ultra assay enables laboratories to adhere to current recommendations 1 for the detection of acute myocardial necrosis. As discussed by Melanson et al., 5 the improved assay precision enables earlier detection of myocardial damage. However, the number of patients with elevated TnI concentration and atypical clinical presentation of acute myocardial necrosis will increase. It is still debated whether and which therapeutic action should be taken with slightly elevated troponin concentrations, as mortality in patients with minimally increased cTnI concentrations will be lower than in patients with clearly elevated values. 6 The TnI-Ultra assay, or another cTnI assay with equal performance, should be used in clinical studies into the optimization of diagnosis and treatment of minor myocardial necrosis.
Footnotes
Acknowledgement
Siemens Medical Solutions Diagnostics (formerly Bayer Healthcare, Diagnostics Division, Breda, The Netherlands) supplied the reagents used in this study free of charge.
