Abstract
Abstract
Background
Rheumatoid arthritis (RA) is the most common inflammatory joint disease. The aetiology of RA remains unknown, but autoimmune responses are considered to play an important role in the disease pathophysiology. Currently available data suggests that the process of diagnosing RA may benefit from testing for anticyclic citrullinated peptides. Identification of the presence of citrullinated proteins in rheumatoid synovial fluids is important for the elucidation of the aetiology of RA as well as in the differential diagnosis of rheumatic-related diseases.
Methods
A proteomics-based approach using electrophoresis/mass spectrometry was applied to identify the citrullinated proteins in synovial fluids from patients with RA. Synovial fluids from patients with RA were subjected to sodium dodecyl sulfate–polyacrylamide gel electrophoresis and Western blot analysis to detect the citrullinated proteins. Identification bands were then subjected to mass spectrometry.
Results
Three proteins – citrullinated fibrinogen, citrullinated fibronectin and citrullinated vimentin – in synovial fluids from RA patients were identified by matrix-assisted laser desorption ionization time-of-flight mass spectrometry.
Conclusions
Proteomics-based analysis can be used to detect citrullinated proteins in synovial fluids from RA patients.
Introduction
Rheumatoid arthritis (RA) is a common systemic autoimmune disease characterized by persistent joint inflammation, resulting in loss of joint function, morbidity and premature mortality. Rheumatoid factor has been used as the best RA-associated antibody marker, but its sensitivity for RA is only 30–80%. 1–3 Recently, an anticyclic citrullinated protein (anti-CCP) antibody, with a sensitivity of up to 80% and a specificity of 98% in patients with RA, has attracted much attention. 4 The anti-CCP antibody specifically recognizes the proteins containing citrulline, which is generated by the post-translational modification of arginine residues by peptidylarginine deiminase (PADI). To date, five isoforms of PADI (PADI1, 2, 3, 4 and 6) have been identified on human chromosome 1p36, a region related to susceptibility to RA. 5 Suzuki et al. 6 analysed single nucleotide polymorphisms and reported that the PADI4 gene is closely associated with RA in the Japanese population.
In contrast to its well-established relevance in diagnostic procedures, relatively little is known about the physiological significance of protein citrullination, which interferes with the organized protein structure and contributes to protein unfolding. A hypothesis that citrullination is intimately involved in the pathogenesis of RA, resulting in the breakage of self-tolerance to this cartilage-specific autoantigen, has been presented. 7
In the present study, we identified citrullinated protein profiles of synovial fluids from RA patients by a proteomics-based analysis.
Materials and methods
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis and Western blot with anticitrulline antibody
In this study, we used 10 synovial fluid samples from patients with RA attending the Department of Orthopedic Surgery, Osaka Medical College. Our study was approved by the ethics committee of the Osaka Medical College.
Synovial fluids were stored at −80°C until use. After incubation with hyaluronidase at 37°C for 30 min, samples of 60 μg protein of synovial fluids per lane were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE). 2–15% gradient polyacrylamide gels (9 cm long; Daiichi Chemical Co., Ltd., Tokyo, Japan) were applied to SDS–PAGE. Separated proteins were transblotted to polyvinylidene fluoride (PVDF) membranes (Hybond P; Amersham Biosciences, Tokyo, Japan) for 1 h at 20 V in a Mini Trans-Blot System (Bio-Rad, Hercules, CA, USA) or visualized by silver staining of the gels. After transfer, citrullinated proteins on the membranes were detected by Western blot using anticitrulline (modified) detection kit (Upstate Biotechnology, Inc., NY, USA) according to the manufacturer's instructions. Briefly, citrulline residues of the proteins immobilized on the membranes were modified by 2, 3-butanedione monoxime and antipyrine in a strong acid solution. Then, the modified citrulline residues were detected by the rabbit polyclonal-modified anticitrulline antibodies and goat antirabbit IgG antibody conjugate with horseradish peroxidase (HRP). The bands of citrullinated proteins were developed with an enhanced chemiluminescence system (ECL Plus; Amersham Biosciences).
Protein identification by mass spectrometry
Protein identification by mass spectrometry was performed as reported previously. 8 Gels were stained with silver nitrate and gel pieces corresponding to Western blot-positive bands were excised. Each gel piece was alkylated and digested in 0.1 mL trypsin solution (0.01 g trypsin/L in 40 mmol/L NH4HCO3, pH 8.5; Promega Corp., Madison, WI, USA).
After enzymatic digestion, measurements of extracted peptides were performed with an Ultraflex MALDI-TOF/TOF mass spectrometer (Bruker Daltonics, Bremen, Germany). Peptide mass fingerprints were used to identify the proteins according to tryptic fragment size with the Mascot Search engine based on the entire NCBInr and SwissProt protein databases, using the assumption that the peptides were monoisotopic, oxidized at methionine residues and carbamidomethylated at the cysteine residues. Probability-based MOWSE scores were estimated by comparing the search results against the estimated random match population and were reported as −10 × log10 (p), where p is the absolute probability.
Immunoprecipitation and Western blot with anticitrulline antibody
Rabbit polyclonal antihuman fibrinogen antibody (AssayPro, St Charles, MO, USA) was used to immunoprecipitate fibrinogen as described previously. 9 Purified fibrinogen from synovial fluids of RA patients was identified by Western blot analysis with anticitrulline antibody as described above. Bands were developed with ECL Plus.
Western blot with antivimentin antibody
Synovial fluids separated by SDS–PAGE were transferred to PVDF membranes and were incubated with antivimentin antibody (1:100; Lab Vision Corporation, CA, USA). After washing twice in phosphate-buffered saline, membranes were incubated with HRP-conjugated donkey antirabbit IgG (1:1000; Amersham Biosciences) and bands were visualized with ECL Plus.
Results
Figure 1a shows the silver stain profile of proteins isolated from mixed synovial fluids from 10 RA patients. Figure 1b shows the profile of Western blot with anticitrulline antibody; more than 10 bands were visible. Bands in silver-stained gels corresponding to three major bands identified by Western blot (nos 1–3) were analysed by matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF/MS). Figure 2 shows the mass spectrum (mass fingerprint) obtained from tryptic digests of proteins in band 1. Underlined numbers in Figure 2 were identified as m/z values of tryptic digests of fibrinogen. Table 1 shows the peptide sequences of the fibrinogen contained in band 1. The MALDI/MS spectrum showed 60% coverage of the fibrinogen sequence. Figure 3 shows the mass spectrum obtained from tryptic digests of proteins in band 2. The MALDI/MS spectrum showed 28% coverage of the fibronectin sequence. Peptide sequences of vimentin were partially characterized by MALDI-TOF/MS with tryptic digests in band 3 (unpublished data). The molecular weight of band 3 was the same as that in the Western blot of synovial fluids treated with the antivimentin antibody (data not shown).
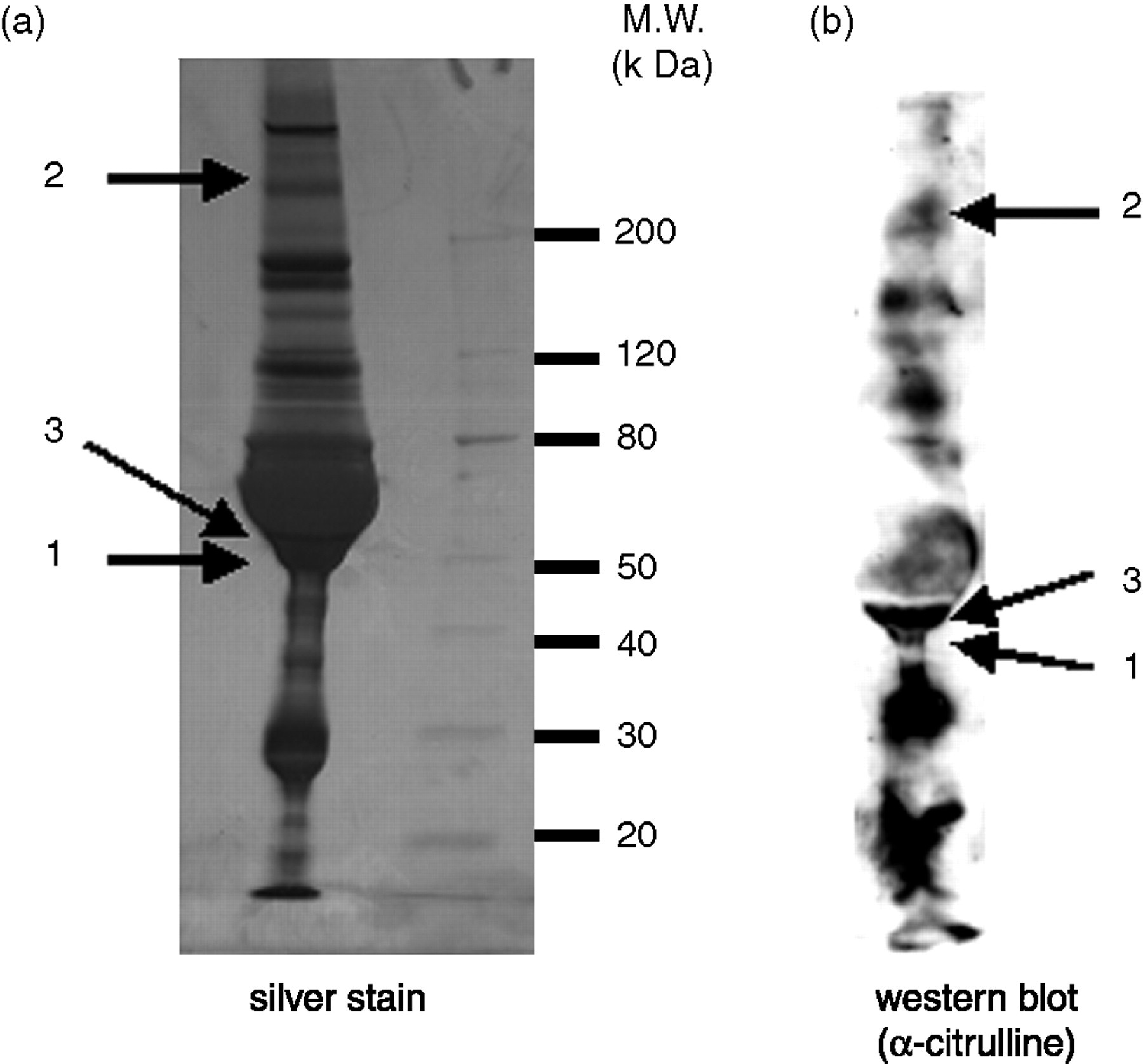
(a) Silver staining of mixed synovial fluids from 10 rheumatoid arthritis (RA) patients separated by sodium dodecyl sulphate–polyacrylamide gel electrophoresis. (b) Western blot analysis of synovial fluids from 10 RA patients with anticitrulline antibody. Numbered bands were identified by matrix-assisted laser desorption ionization time-of-flight mass spectrometry with tryptic digests of extracted protein, and data are listed in Table 2
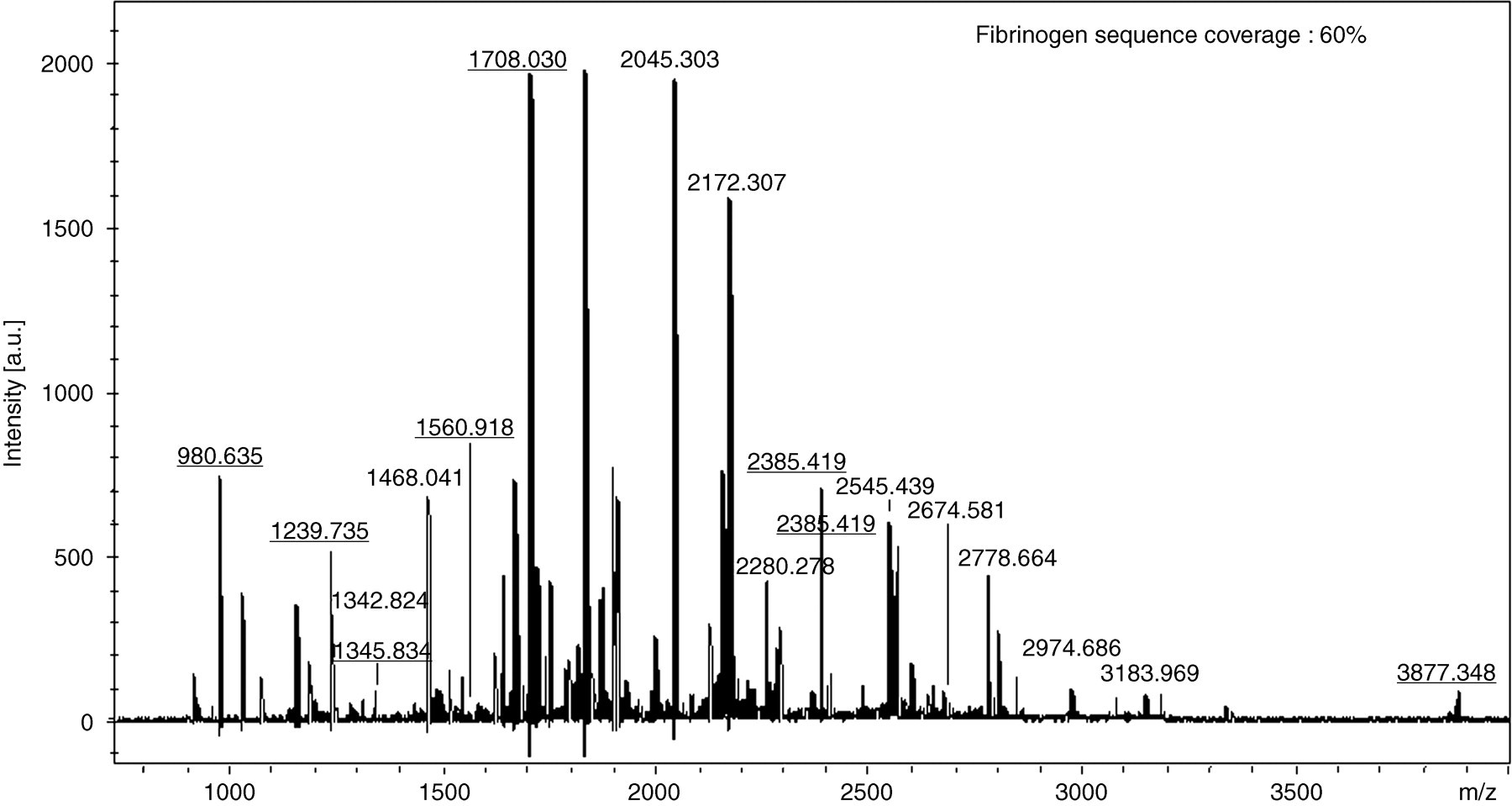
Matrix-assisted laser desorption ionization mass spectrum obtained from the protein in band 1 after trypsin digestion. Underlined numbers indicate the m/z of tryptic digests of fibrinogen
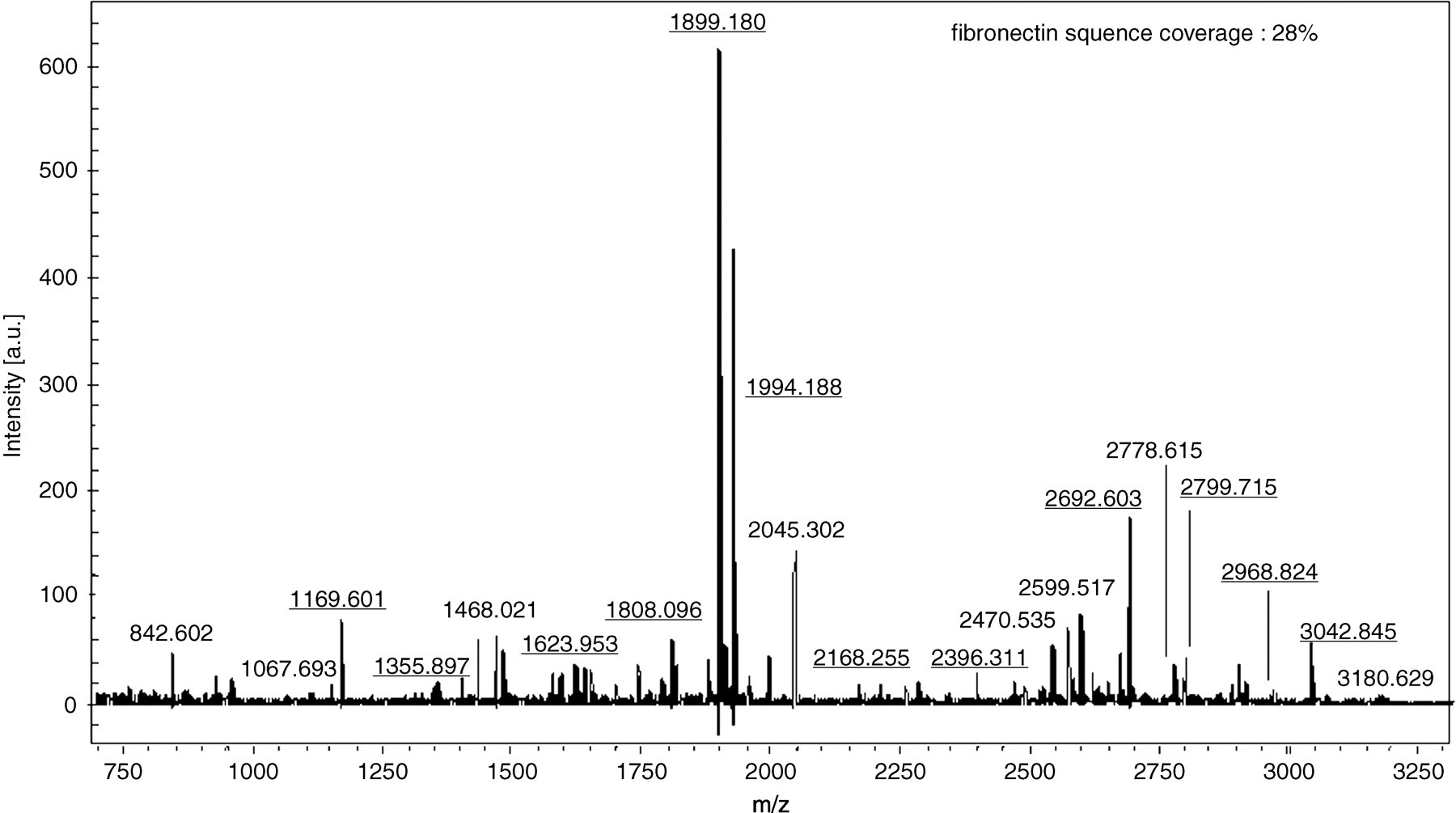
Matrix-assisted laser desorption ionization mass spectrum obtained from the protein in band 2 after trypsin digestion. Underlined numbers indicate the m/z of tryptic digests of fibronectin
Peptide sequences of proteins contained in band 1 as determined by mass spectrometry
Synovial fluid immunoprecipitated with polyclonal antihuman fibrinogen antibody showed the presence of citrullinated fibrinogen by immunoblotting with antibody to citrullinated proteins (Figure 4). Figure 4a shows a Coomassie blue stain of material immunoprecipitated by the polyclonal antihuman fibrinogen antibody. Figures 4b and c show Western blots of the immunoprecipitates blotted with antifibrinogen antibody and anticitrulline antibody, respectively. The three citrullinated proteins are listed in Table 2.
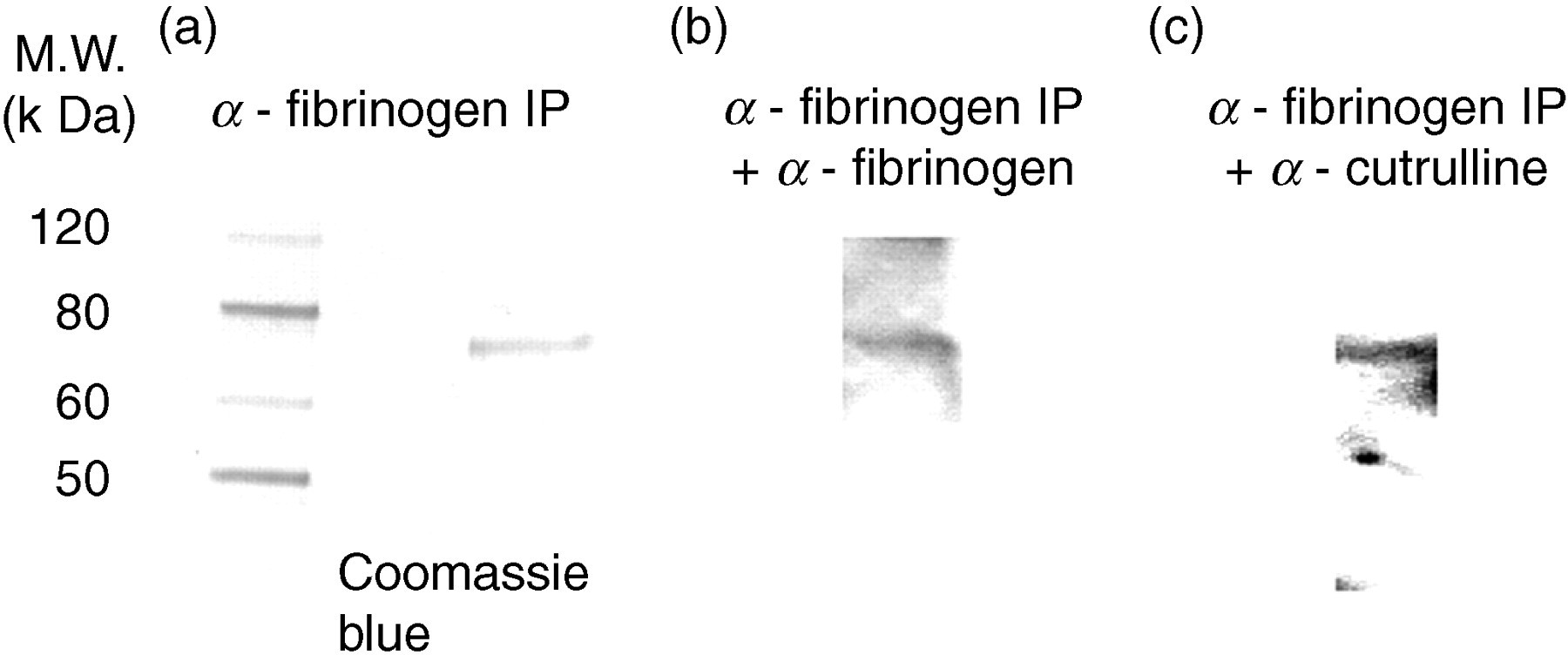
Coomassie blue stain of proteins, immunoprecipitated from synovial fluids with antihuman fibrinogen antibody and separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis. (a) Western blot analysis of material immunoprecipitated with antifibrinogen antibody (b) and anticitrulline antibody (c)
Identified citrullinated proteins in synovial fluids
Discussion
It has become clear that there is a close relation between citrullinated proteins and the aetiology of RA. In the present study, we analysed synovial fluids from 10 RA patients with a proteomics-based approach and detected three citrullinated proteins, those of fibrinogen, fibronectin and vimentin. Peptide sequences of fibrinogen and fibronectin are extensively characterized. Although we detected a band corresponding to vimentin that reacted with anticitrulline antibody, the peptide sequence was only partially characterized by MALDI-TOF/MS.
Fibrinogen and fibrin play important roles in blood clotting, fibrinolysis, cellular and matrix interactions, inflammation, wound healing, angiogenesis and neoplasia. Masson-Bessiere et al. 10 suggested that autoimmunization against deiminated fibrin is a critical step in RA pathogenesis. In a recent study, citrullinated fibrinogen was detected as a soluble citrullinated autoantigen in synovial fluids of RA patients by immunoblotting. 11 Matsuo et al. 12 confirmed the significance of fibrinogen as a major citrullinated autoantigen in synovium of RA patients by a proteomic approach. 12 However, citrullinated fibrinogen in synovial fluids was not identified by proteomics-based analysis.
Fibronectin is expressed at high concentrations in arthritic joints, and plays important roles in pro-inflammation, pannus invasion and other events associated with disease progression. 13 In synovium, Chang et al. 14 reported that the fibronectin citrullination is a specific event in RA that it alters interactions between fibronectin and its receptors and growth factors contributing to such mechanisms of RA pathogenesis as perturbed angiogenesis and apotosis. 14 However, citrullinated fibronectin has not been detected in synovial fluids in previous studies.
Vimentin is an intermediate filament that is expressed widely in mesenchymal cells and macrophages and is readily detectable in synovium and fibroblast-like synoviocytes. 15 Anticitrullinated vimentin autoantibodies are known to possess high specificity (92–98%) and to show positive predictive value (84–99%) for RA. 16 It is reported that immune reactants are plentiful in the rheumatoid joint; citrullinated vimentin as antigen is present in the synovium, but not in synovial fluids. 17 Citrullinated fibronectin and vimentin have been detected not in synovial fluids, but in the synovium and serum, respectively, of RA patients. It has been unclear whether these proteins are citrullinated in synovial fluids. We identified citrullinated forms of these proteins in synovial fluids with a proteomics-based approach in the present study.
In previous studies, citrullinated proteins have been identified by immunoblotting with antibodies against specific deiminated proteins. The identification of candidate proteins is necessary for this method, therefore the detected proteins are necessarily limited. For proteomics-based analysis, the identification of candidate proteins is not necessary.
Proteomics is a powerful tool for protein discovery; however, citrullinated proteins were detected by the proteomics-based analysis only in the synovium, not in the synovial fluids. Synovial fluids can be collected more easily than synovium and more directly reflect joint inflammation than serum. Therefore, we analysed synovial fluids using proteomics in the present study.
In conclusion, we analysed synovial fluids from 10 RA patients by a proteomics-based approach and detected three citrullinated proteins, those of fibrinogen, fibronectin and vimentin. Our proteomics-based analysis is easier and more useful for screening than conventional immunoblotting. This method provides a new approach for the detection of citrullinated proteins.
Footnotes
ACKNOWLEDGEMENTS
We thank Dr Akihito Ishigami (Tokyo Metropolitan Institute of Gerontology, Tokyo, Japan) for suggestions and Mrs Kyoko Fukamoto (Osaka Medical College, Osaka, Japan) for technical help.
