Abstract
Background
The in vitro stability of N-terminal (NT)-proBNP (brain natriuretic peptide) at room temperature and at 4°C is excellent and has been well studied. However, less is known concerning its stability after a long-term frozen storage. This notion could be of major interest in the context of clinical evaluations.
Methods
NT-proBNP was measured on 97 heparinized samples before and after a two-year frozen storage (−20°C) using the Roche Elecsys system.
Results
There is a slight but significant decrease of NT-proBNP concentration after frozen storage. However, this decrease is <10% for more than 90% of the samples and the maximum decrease is 16%. Moreover, values on frozen samples are well correlated with values on fresh samples (r = 0.99) using the following equation: NT-proBNPstored sample = 0.93 NT-proBNPfresh sample + 81. A similar equation was found in lower concentrations (NT-proBNP ≤ 2000 ng/L).
Conclusions
These data show that NT-proBNP can be stored for at least two years at −20°C before measurement without a substantial loss of immunoreactivity.
Introduction
N-terminal (NT)-proBNP is co-secreted with brain natriuretic peptide (BNP) in response to myocardial stretch or cardiac overload. These two markers are often used in heart failure for diagnosis, prognosis and management. They are also powerful independent prognostic markers of mortality in acute coronary syndromes. The measurement of NT-proBNP is a sandwich immunoassay using two monoclonal antibodies: the capture antibody is against the N-terminal region (amino acid 1–21), whereas the detection antibody is against the middle part of the molecule (amino acid 39–50). The recommended specimen is heparinized plasma. Unlike BNP, the in vitro stability of NT-proBNP is optimal at room temperature as well as at 4°C on whole blood or plasma. The stability of the peptide is at least three days at room temperature and six days at 4°C when the sample is centrifuged at arrival in the laboratory. 1–4 As regards its stability in frozen samples, only few data have been reported in the literature. 5,6 Mueller et al. demonstrated that NT-proBNP is stable for at least four months at −20°C; other authors have reported a very good stability of this peptide for at least one year at −80°C. However, no data are available concerning the stability of NT-proBNP for more than four months at −20°C. The purpose of our study was to assess the in vitro stability of NT-proBNP after a −20°C storage for two years.
Materials and methods
To assess the stability, 97 samples with a routine NT-proBNP measurement (between January and February 2005) were frozen (−20°C) for 12 h after the blood sample was obtained. The samples used were heparinized plasma and were measured using an electrochemiluminescence immunoassay (Elecsys, Roche Diagnostics, Meylan, France). In our laboratory, the within and between runs of %CV (n = 10) at a mean of 150 ng/L were 1.7 and 2.7%, respectively, according to the manufacturer's data. Initial concentrations of NT-proBNP (fresh samples) ranged from 65 to 50,983 ng/L (5 concentrations exceeded the linearity limit, i.e. 35,000 ng/L). Two years later [mean (SD) time of storage, 800 (33) days], plasma samples were removed, thawed at room temperature and centrifuged prior to measurement in singlicate.
Results
Values on fresh samples compared with values on stored samples show the following equation: NT-proBNPstored sample = 0.93 (95% CI, 0.91–0.95) NT-proBNPfresh sample + 81 (95% CI −164 to +326) (r = 0.99). Excluding the five samples with a concentration > 35,000 ng/L, the equation is very close to the previous one: NT-proBNPstored sample = 0.93 NT-proBNPfresh sample + 73 (r = 0.99; n = 92). The recovery of NT-proBNP (repeat concentrationNT-proBNP/initial concentrationNT-proBNP) ranged from 84 to 122% with a mean recovery of 97%. For the unexpected 122% recovery, the initial value was very low: 71 ng/L and was unfortunately only measured in singlicate, as well as the repeated value. The recovery was between 90 and 110% for 86 of the 97 samples (87%). After storage, there is a slight but significant decrease (P = 0.001), which is more pronounced for high NT-proBNP values (P < 0.01). However, the maximum decrease is <20% and exceed 10% for only eight of the 97 samples (Figure 1). For these eight samples, age of the patients, sex ratio or the renal function are not significantly different from the other 89 samples.
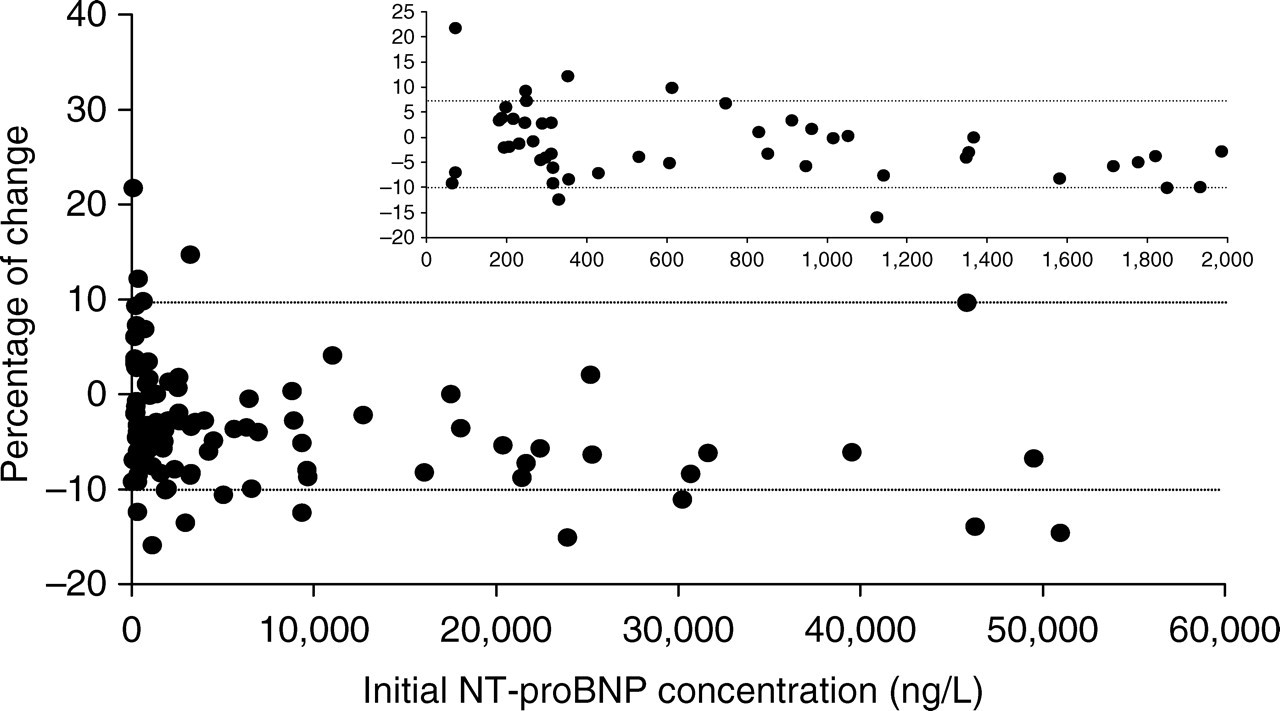
Percentage of change between the repeat value (stored sample) and the initial value (fresh sample). Inset, NT-proBNP in the range of 0–2000 ng/L. Percentage of change = (repeat value − initial value)/initial value × 100
In a second stage, we more specifically evaluated the correlation for lower values. As the different cut-off values were 125 and 450 ng/L for the diagnosis of ambulatory heart failure and range from 300 to 1800 ng/L in the context of dyspnoea, we decided to study the correlation in the range of 0–1000 ng/L (n = 34) and 0–2000 ng/L (n = 48). In this latter range of values, the correlation comparing repeated (frozen samples) and initial (fresh samples) values was considered excellent with nearly the same equation than the previously described (slope 0.934; intercept 22). For values between 0 and 1000 ng/L, the correlation was even better (slope 1.00; intercept −0.84; r = 0.99).
Conclusion
These data show, for the first time to our knowledge, that NT-proBNP can be stored for at least two years at −20°C before measurement without a substantial loss of immunoreactivity using the Roche Elecsys system. Our results suggest that, as the different NT-proBNP immunoassays use the same antibodies developed by Roche Diagnostics, this procedure could also be applied to other NT-proBNP immunoassays. When a clinical protocol is launched, it is of major importance for physicians and researchers to know the conditions of storage of blood samples for the measurement of different markers. In the case of NT-proBNP, samples can be simply stored at −20°C for at least two years.
Footnotes
Acknowledgements
The authors are grateful to Vincent Masson (Roche Diagnostics) for providing reagents. We also thank Richard Medeiros, Rouen University Hospital Medical Editor, for editing the manuscript.
