Abstract
Abstract
Background
Patent Blue V is an inert dye increasingly being used during cancer surgery to identify the sentinel lymph node. We recently discovered three cases with falsely elevated lipaemic indices on the Roche Modular, following intramammary injection of Patent Blue. This, and other potential interferences by Patent Blue, is examined in this study.
Methods
Serum samples which visually appeared normal, haemolysed or icteric were spiked with Patent Blue to a concentration of 7.3 mg/L and the effect on the lipaemic, haemolysis and icteric indices and routine chemical analyses (sodium, potassium, urea, creatinine, bilirubin, alanine aminotransferase, gamma-glutamyltransferase, aspartate aminotransferase, magnesium, creatine kinase, lactate dehydrogenase, amylase, albumin, calcium and phosphate) undertaken. Dose-response curves in the range of 4.9 mg/L to 22.7 mg/L of Patent Blue were established for normal, icteric and haemolysed pooled serum samples.
Results
Significant positive interference by 7.3 mg/L of Patent Blue was observed in the lipaemic index, with an increase of 149 ± 6.3 (mean ± standard deviation [SD]; P < 0.001). Significant negative interference was observed in both the haemolysis and icteric indices, with decreases of 71 ± 9.7 (P < 0.001) and 46 ± 8.9 (P < 0.001), respectively. Patent Blue had a linear dose-response effect on the serum indices. No significant differences were observed in the chemical analyses assessed.
Conclusions
The effects observed on the Roche Modular serum index measurements with Patent Blue raises the novel concept of interference in methods routinely used to detect interference. Despite Patent Blue having no direct effect on chemical analysis, failure to reliably detect potential interference, particularly haemolysis, may lead to misleading results being issued.
Introduction
We have recently discovered three cases of interference in the serum indices measurement on the Roche Modular (F Hoffmann-La Roche, Basel, Switzerland). Serum indices can be used to detect any signs of haemolysis, lipaemia and icterus in a sample, and are particularly useful when using automated pre-analytical modules where visual inspection of the sample for such potential interference may be difficult. On the Roche Modular system the semi-quantitative unitless serum indices are calculated from the differences in absorption at three different wavelength ranges (480–505 nm, 570–600 nm and 660–700 nm) with various correction factors (see Appendix 1). A haemolysis index (H) of 50 is equivalent to a known haemoglobin concentration of approximately 500 mg/L, an icterus index (I) of 100 being equivalent to a known unconjugated bilirubin concentration of approximately 100 µmol/L and lipaemia index (L) of 60 equivalent to 600 mg/L Intralipid (Kabi-Pharmacia Inc, Stockholm, Sweden). Although the Roche assay data-sheets give information as to the level of these indices a particular assay is free from interference, different laboratories may have different index decision thresholds. 1 In our laboratory, we classify H = 50–120 as slightly haemolysed, H = 120–250 as haemolysed and H > 250 as grossly haemolysed, I > 100 as being icteric and L > 60 as lipaemic.
We have observed three cases that were lipaemic according to the serum indices (lipaemic index 140–160, normal <60), but all had low triglyceride concentrations (0.42–0.9 mmol/L). When the samples were subsequently visually inspected it was noted that the samples were not remotely turbid or lipaemic, but a quite distinct bright green colour. Investigation into the source of the samples revealed that all three were from patients who had undergone surgery for breast cancer, and who had received an intramammary injection (2 mL) of 2.5% Patent Blue V (Guerbet, France) during the preceding 4–6 h.
Patent Blue is an inert dye increasingly being used during cancer surgery to identify the sentinel lymph node, which can be distinguished from other lymph nodes by its blue staining following the direct injection of the Patent Blue into the site of the primary tumour. 2–5 In breast cancer, recent studies have suggested that if the sentinel lymph node is free from tumour, widespread axillary node dissection may be avoided. 2 Similar studies are emerging with regard to colorectal cancer, bladder cancer and malignant melanoma. 3–5 As these latest studies become incorporated into routine medical practice it is likely we will see an increase in the number of serum and urine samples contaminated by Patent Blue.
The manufacturer's data-sheet warns that Patent Blue may interfere in pulse oximetry, but does not report of problems with biochemical analysis. 6 There is evidence that a similar dye, Disulphine Blue, significantly interferes in some colorimetric assays. 7 No studies are available as to what effect Patent Blue has on biochemical analysis or serum index measurements; this is an issue we have tried to examine in view of our case reports suggesting interference with the lipaemic index on the Roche Modular.
Methods
We visually identified 20 ‘normal’, 20 haemolysed and 20 icteric serum samples. The serum indices were measured on all the samples, and ‘routine’ chemical analyses (sodium, potassium, urea, creatinine, bilirubin, alanine aminotransferase, gamma-glutamyltransferase, aspartate aminotransferase, creatine kinase, lactate dehydrogenase, amylase, albumin, calcium, magnesium and phosphate) were performed on the ‘normal’ samples using the Roche Modular.
Following a 1:100 dilution with water of 2.5% Patent Blue (Guerbet, France), 30 µL was added to 1 mL of sample (giving a final concentration of 7.3 mg/L). The serum indices and chemical analysis were repeated and Student's t-test (SPSS statistics analysis software, Chicago, USA) was used to determine if there was any significant difference in the results following the addition of Patent Blue.
Dose response curves in the range of 4.9 mg/L to 22.7 mg/L were established by adding 20–100 µL of the diluted Patent Blue solution to 1 mL of pooled normal serum samples, pooled haemolysed serum samples and pooled icteric serum samples. Serum indices were measured on all the samples, plus the ‘routine’ chemical analysis was repeated on the pooled normal serum samples.
Spectrometric scanning of the diluted Patent Blue was performed over the range 320 nm to 680 nm using a Shimadzu UV1601 UV-visible spectrophotometer (Shimadzu UK Ltd, Milton Keynes, UK).
Results
A significant positive interference by 7.3 mg/L Patent Blue was observed in the lipaemic index, with an increase of 149 ± 6.3 (mean ± SD; P < 0.001). Significant negative interference was observed in both the haemolysis and icteric indices, with mean decreases of 71 ± 9.7 (P < 0.001) and 46 ± 8.9 (P < 0.001), respectively (Figures 1a–c). The positive interference in the lipaemic index occurred even if the original sample was haemolysed or icteric. As the dose of Patent Blue added to pooled serum samples increased, the serum indices showed a linear dose-response effect (Figure 2).
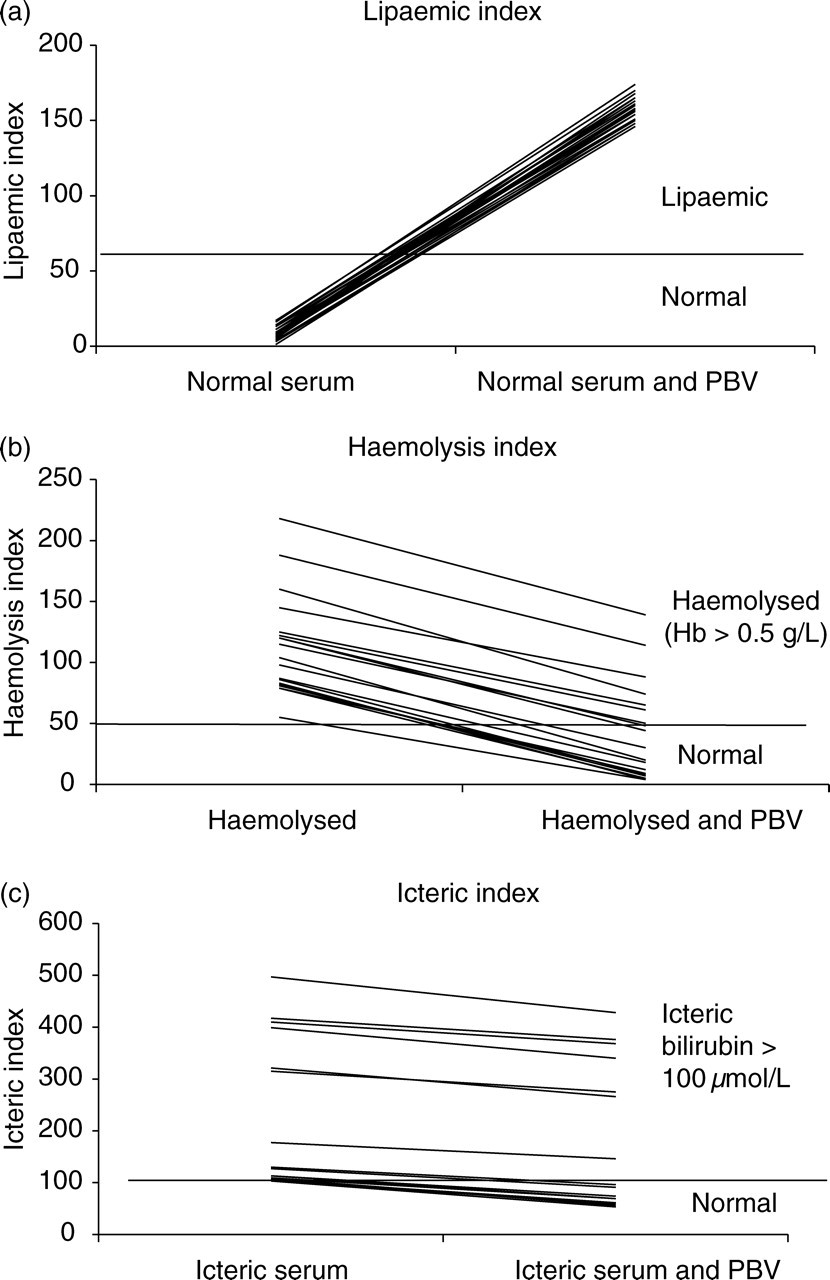
The effect of 7.3 mg/L Patent Blue V on the lipaemic index (a), the haemolysis index (b) and the icteric index (c) on the Roche Modular PBV, Patent Blue V; Hb, haemoglobin
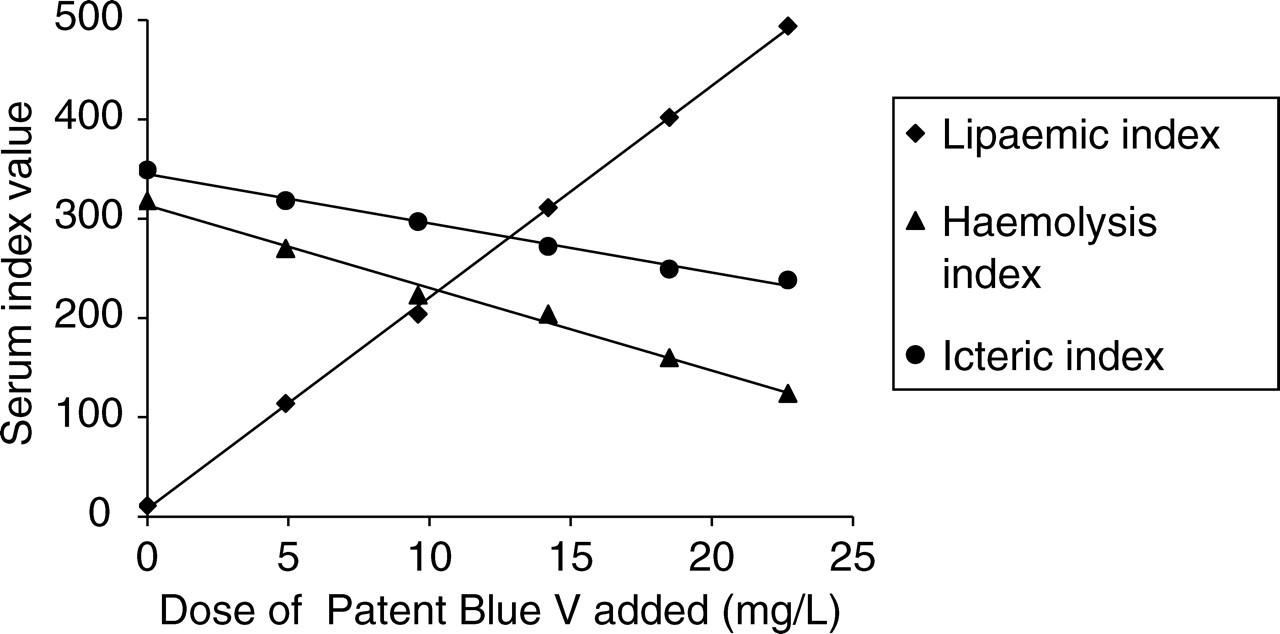
The effect of increasing concentration of Patent Blue V on the serum indices on the Roche Modular
No significant difference was observed for any of the routine chemical analyses following addition of 7.3 mg/L Patent Blue to the 20 ‘normal’ samples (Table 1).
The effect of Patent Blue V (PBV) on routine chemical analysis of normal serum on the Roche Modular
*Not significant
Spectrophotometric scanning revealed a peak absorbance of the Patent Blue at 640 nm (Figure 3)
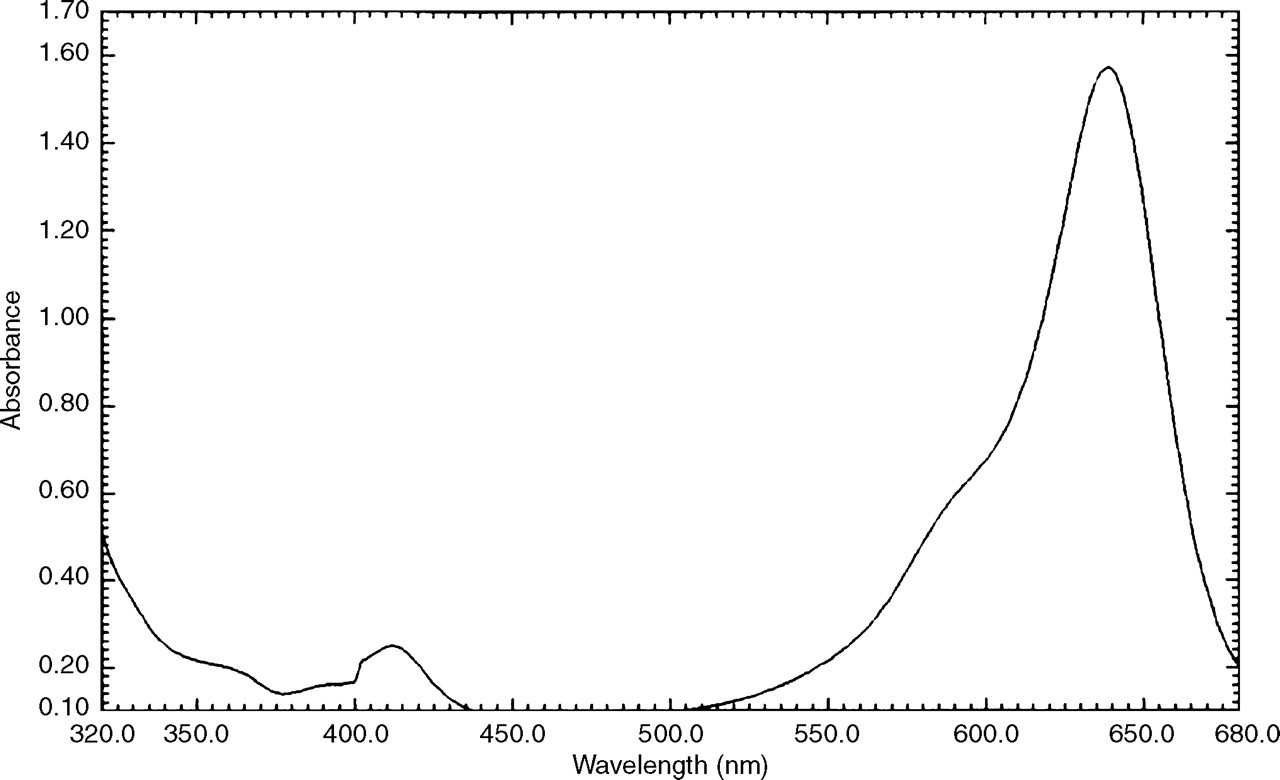
Spectrophotometric scan of Patent Blue V diluted in water
Discussion
Patent Blue V significantly interfered with the haemolysis, lipaemic and icteric index measurements on the Roche Modular, but did not affect the routine biochemical analyses assessed.
The absorbance peak of Patent Blue at 640 nm overlaps with two of the bichromatic wavelength pairs used for serum index measurement (570–600 nm and 660–700 nm). It is evident from the formulae used to derive the index value from the absorbance measurements, how Patent Blue would cause a positive interference in the lipaemia index, and different magnitudes of negative interference in the haemolysis and icterus indices (Appendix 1).
The negative interference on the haemolysis index is of greatest concern. Electrolyte disorders are not uncommon in the postoperative period, especially in patients on intravenous fluids. Potentially serious hypokalaemia may be completely masked by haemolysis; if the haemolysis remains undetected due to the negative interference by Patent Blue on the index measurement, an inaccurate and unreliable result may be issued to the clinician.
The negative interference in the icteric index, although significant, is not as likely to have such a major impact on clinical care, especially considering the actual measurement of bilirubin is unaffected by Patent Blue.
The positive interference in the lipaemic index confirms the findings that we observed in the three clinical cases. In our laboratory we routinely request a lipid profile on all lipaemic specimens. The discrepancy between the lipaemic index and the lipid profile results indicates the possibility of Patent Blue contamination and should prompt visual inspection of the sample, particularly to exclude haemolysis which may have been missed by the index measurements.
The chemical analyses in this study were chosen as they represent the most commonly requested tests on the Roche Modular in the acute setting, not necessarily because interference was expected. The concentration of Patent Blue used in the study (7.3 mg/L) was that observed to give an increase in the lipaemic index of approximately 150, comparable to the level of interference we observed in the three clinical cases. The finding that at this level there is no interference observed in any of the analyses assessed was very reassuring. It can probably be explained by the fact, that unlike the serum indices, the chemical analyses use a sample blank in the assays.
Following on from this preliminary report there are a number of areas where further research is required. It may be predicted that interference is possible in assays that do not blank and where the primary detection wavelengths coincide with the absorbance wavelengths of Patent Blue. Therefore serum index measurements and chemical analysis using different analysers and methods, in addition to the full repertoire of tests on the Roche Modular, need to be examined for potential interference. The dose-response curve on the pooled serum samples suggested that there was no interference at over three times the concentration observed in our patients, however the literature regarding the use of Patent Blue in surgical oncology is rapidly expanding and it is possible that the different doses and techniques described may give quite different serum concentrations to those observed in our clinical scenarios. Further research is therefore also required to determine the effect of much greater concentrations of Patent Blue.
In summary, this study has raised the novel concept of interference in the methods routinely used by the Roche Modular to detect interference by haemolysis, icterus and lipaemia. Despite Patent Blue having no direct effect on many chemical analyses, failure to reliably detect other forms of interference, particularly haemolysis, may lead to misleading results being issued to the clinician with potentially adverse clinical consequences.
Footnotes
Appendix 1
