Abstract
Abstract
Background
It is important to explain target proteins for the understanding of the pathogenesis of vitreoretinal diseases. In a previous study, we identified more than 100 proteins including seven angiogenic-modulated factors in vitreous humours (VHs). Although there have been many reports of expressed protein profiles in VHs, only a few of these are modified proteins, such as those undergoing phosphorylation and oxidation.
Methods
We applied Western blotting (WB), selective staining of phosphoproteins and mass spectrometry to detect and identify phosphoproteins in VHs of patients with vitreoretinal diseases. After the removal of albumin and immunoglobulins A/G in VHs, the proteins were separated by sodium dodecyl sulphate–polyacrylamide gel electrophoresis and reacted with anti-PY-20 monoclonal antibody on transfer membranes, and treated proteins were visualized with Phos-tag™ and identified by matrix-assisted laser desorption time-of-flight mass spectrometry (MALDI-TOFMS).
Results
WB analysis detected four positive bands, 20, 30, 35 and 55 kDa, in VHs of patients with vitreoretinal diseases. One of them, 55 kDa, was frequently detected in VHs of patients with macular hole (MH) and retinal detachment (RD), but the band was not found in patients with proliferative diabetic retinopathy (PDR). α-1 antitrypsin (α-1 AT) was identified in excised gel pieces of this band.
Conclusions
We identified five phosphorylated proteins such as α-1 AT in VHs of patients with vitreoretinal diseases by MALDI-TOFMS and WB analysis. Phosphotyrosyl α-1 AT was neither detected in PDR patients nor in any plasma. Phosphotyrosyl α-1 AT may be a new biomarker of MH and RD.
Introduction
Vitreoretinal diseases were mainly separated into two categories: one was a proliferative and inflammatory disease such as proliferative diabetic retinopathy (PDR), the other consisted of non-proliferative and non-inflammatory diseases such as macular hole (MH) and retinal detachment (RD).
PDR occurs as a chronic complication of diabetes mellitus and is a major cause of blindness in people below 65 years of age. In PDR, the production of angiogenic and antiangiogenic factors by retinal cells may be altered; consequently, the concentration of these factors in vitreous humour (VH) may also change, as well as elevated glycosylated haemoglobin concentrations, high blood pressure and chronic hyperglycaemia. Currently, considerable effort is being focused on the identification of biomarkers such as pigment epithelium-derived factor and insulin-like growth factor for the early detection and diagnosis of PDR. On the other hand, there are many non-proliferative, non-inflammatory vitreoretinal diseases such as RD and MH. RD, resulting from retinal breaks, is mainly caused by vitreoretinal traction that occurs because of injury and ageing. MH may be caused by a contraction of the pre-macular membrane in the vitreous cortex caused by ageing of the eyes, but the aetiologies of MH are still unclear.
From our previous reports, we identified the expressed soluble protein profiles of VHs from patients with vitreoretinal diseases by sodium dodecyl sulphate–polyacrylamide gel electrophoresis (SDS–PAGE), Western blotting (WB) and mass spectrometry (MS). 1,2 Although other researchers have also reported the identification of proteins expressed in VHs, such as pigment epithelium-derived factor and vascular epithelium growth factor, 3–6 there have been only a few reports concerning the profiles of post-translational-modified proteins, such as phosphoprotein, in VHs.
In general, phosphoproteins and phosphopeptides play important roles in various biological functions, such as the regulation of gene expression, protein synthesis, ion transport as well as cell differentiation and degradation. 7 In ocular tissues of experimental animals, some expressed proteins are either phosphorylated to promote their activity or phosphorylated by some other proteins for their biological effect. 8 For example, low molecular weight phosphopolypeptides activated the differentiation and degradation of lens cell growth in the iris–ciliary complex, aqueous humour and VH derived from experimental animals. 8 In the retina, phosphorylations of proteins were accelerated under ischaemic conditions, such as in ischaemia–reperfusion injury of rats or branch vein occlusion in the cat. 9,10
Moreover, in the postmortem human retina, it was reported that the apoptosis-inducing phosphorylation of protein kinase C contributes to retinal ganglion cell loss in glaucoma, 11 and six isoforms of NRL (29–35 kDa) were expressed specifically and generated by phosphorylation. 12 However, there have been only a few reports on phosphopolypeptides or phosphoproteins in VH derived from the human eye. Therefore, it is important to further investigate the precise mechanism of the production of phosphopolypeptides in retinas and VHs derived from patients with vitreoretinal diseases.
In this study, we applied SDS–PAGE, WB and MS analyses to discover phosphoproteins that can be used as biomarkers for understanding the aetiology of non-proliferative, non-inflammatory vitreoretinal disease. We identified five phosphotyrosyl proteins, such as α-1 antitrypsin (α-1 AT), in VHs derived from patients with MH and RD; phosphotyrosyl α-1 AT was neither detected in PDR patients nor in any plasma. Phosphotyrosyl α-1 AT may be a new biomarker of MH and RD.
Materials and methods
Chemicals and materials
Urea, glycine, SDS, iodoacetamide, 3-[(3-cholamidopropyl)dimethylammonio]-1-propanesulphonate (CHAPS), acrylamide, N, N-methylene bisacrylamide, tris (hydroxymethyl)- aminomethane (Tris), ammonium bicarbonate, glycerol, 2-mercaptoethanol, dithiothreitol, Tween 20, ammonium persulphate, phenylmethylsulphonyl fluoride, Nonidet P-40, silver nitrate and EDTA were purchased from Nakalai Tesque (Kyoto, Japan). Methanol, ethanol, acetonitrile, acetone, acetic acid, trifluoroacetic acid (TFA) and distilled water were from Merck (Darmstadt, Germany). Gradient slub-gel of 10–20% (9 cm long) was obtained from Daiichi Chemical Co, Ltd (Tokyo, Japan). The Phos-tag™ 300/460 phosphoprotein gel stain kit was purchased from Perkin-Elmer (Boston, MA, USA).
After informed consent was obtained by Dr Mukai and Dr Ikeda, we used 44 VHs in total (23 patients with PDR, 5 with RD and 16 with MH) from patients with vitreoretinal diseases (Amersham Biosciences) indicating vitreous surgery.
We have already dealt with procedures in this study, and received approval from the Ethical Review Board of Osaka Medical College and collected each sample after informed consent. Dr Mukai and Prof Ikeda are specialists in vitreoretinal diseases, and are the chief doctors of each case. They fully explained the purpose of this study to each patient and/or their family.
Sample preparation and sodium dodecyl sulphate–polyacrylamide gel electrophoresis/Western blotting
The VHs were obtained from patients with vitreoretinal diseases. Before the two-dimensional electrophoresis of VHs, we treated them with an Affi-Gel Blue gel (Bio-Rad, Hercules, CA, USA) and protein A/G PLUS-Agarose (Santa Cruz Biotech Inc, Santa Cruz, CA, USA) to remove albumin and immunoglobulin A/G according to the manufacturers' instructions. A 10 µg sample of protein in each of the treated VHs was separated by SDS–PAGE. The samples were run on a 10–20% gradient slub-gel at a constant current of 30 mA per gel. After electrophoresis, gels were electroblotted onto a polyvinylidene fluoride (PVDF) membrane (Hybond P, Amersham Biosciences, UK) for 1 h at a constant voltage of 20 V, using a Mini Trans-Blot system (Bio-Rad). Following transfer, PVDF membranes were incubated with a blocking solution consisting of washing buffer (10 mmol/L Tris–HCl/100 mmol/L NaCl/0.1% Tween 20) and 5% bovine serum albumin overnight at 4ºC, washed with washing buffer and then incubated with horseradish peroxidase-conjugated monoclonal anti-PY-20 antibody (Amersham Biosciences) at a 1:2000 dilution for 2 h at room temperature. After four washings, the PVDF membranes were developed with an enhanced chemiluminescence system (ECL plus, Amersham Biosciences), and the membranes were scanned with a Luminoimaging Analyzer LAS-3000 (Fuji Film, Osaka, Japan).
Digestion of phosphotyrosyl proteins
SDS–PAGE gel was stained with the Phos-tag™ kit, and the pieces of gel corresponding to WB-positive spots were excised. Each excised piece of the gel bands was reduced, alkylated and then digested with N-p-Tosyl-L-phenylalanine chloromethyl ketone (TPCK)-trypsin (Promega Bioscience Inc, Madison, WI, USA) in Eppendorf tubes at 37ºC for 16 h (all cases). After digestion, each piece was extracted with 0.5% TFA and 0.5% TFA/acetonitrile (1/1), respectively. Each extract was combined in a polyethylene tube and then concentrated under a vacuum. Protein identification was performed as previously reported. 1
Mass spectrometry
The purified phosphopeptides were analysed using matrix-assisted laser desorption time-of-flight mass spectrometry (MALDI-TOFMS) and nanoLC-ion trap MS. The extracted peptides were loaded onto the MALDI target plate by mixing 1 µL of each solution with the same volume of a matrix solution, prepared fresh every day by dissolving 0.3 g/mL of α-cyano-4-hydroxycinnamic acid (Wako Purified Reagent Co, Ltd, Kyoto, Japan) in acetone–ethanol (1:1, v/v) solvent. Measurements were performed using an Ultraflex MALDI-TOF/TOFMS (Bruker Daltonics, Germany) with an accelerating voltage of 20 kV. The laser wavelength was 337 nm and the laser pulse frequency was 25 Hz. Calibration was performed using external peptide standards (Bruker Daltonics) or the trypsin autodigestion peptide signal as an internal standard. The peptide mass fingerprint was used for protein identification from the tryptic fragment size using the Mascot Search Engine based on the entire NCBIn and SwissProt protein databases, employing the assumption that peptides are monoisotopic, oxidized at methionine residues and carbamidomethylated at cysteine residues.
Results
The phosphotyrosyl proteins in treated VHs of patients with PDR, MH and RD were separated by SDS–PAGE and visualized using the Phos-tag™ kit. As shown in Figure 1a, many positive bands in VHs were found at ranges from 40 to 120 kDa, as well as those of plasma samples. Figure 1b shows comparisons of the WB patterns using an anti-PY-20 monoclonal antibody for the detection of phosphotyrosyl proteins in the plasma and VHs derived from patients with vitreoretinal disease. From the results of the WB patterns, several positive bands, 20, 30, 35 and 55 kDa, were found in VHs of RD and MH on the PVDF membrane using the ECL detection kit. In VHs of PDR, three bands of 20, 30 and 35 kDa bands were found, but not the 55 kDa band. On the other hand, the 30 kDa band was only found in plasma samples. The 55 kDa band was only detected in seven of eight patients with MH and three of five patients with RD, being present in none of the 19 patients with PDR.
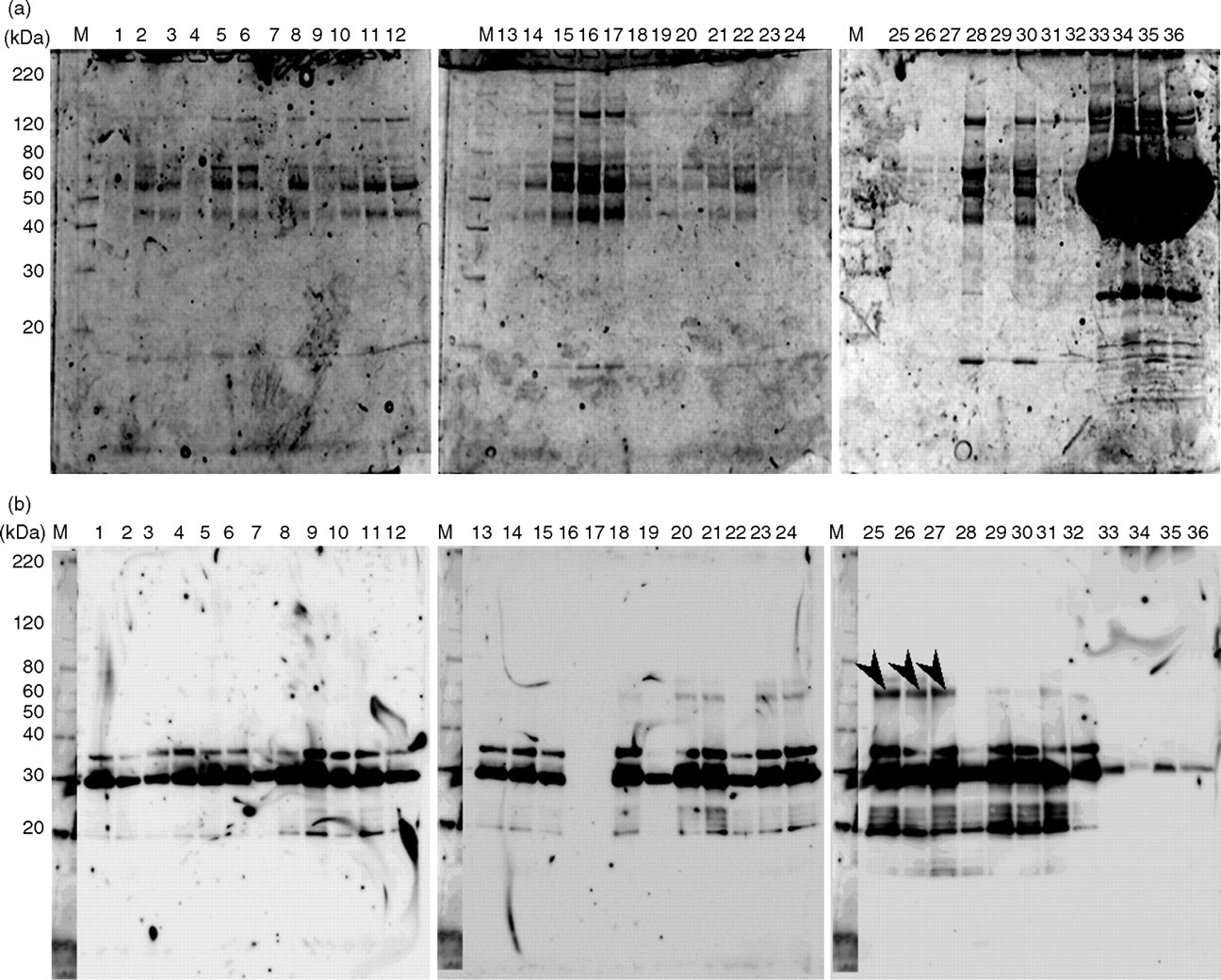
(a) The phosphoprotein profiles of treated vitreous humours (VHs) and plasma of patients with vitreoretinal diseases using the Phos-tag™ kit. 1–19: proliferative diabetic retinopathy (PDR); 20–27: macular hole (MH); 28–32: retinal detachment (RD); 33–36: plasma; M, molecular marker. (b) Western blot analyses of proteins in the unbound fractions of plasma and VHs using an anti-PY-20 antibody. The arrows indicate specific bands present only in VHs of MH. 1–19: PDR; 20–27: MH; 28–32: RD; 33–36: plasma; M, molecular marker
These four bands, 20, 30, 35 and 55 kDa, corresponding to the WB-positive bands in VHs were analysed using MALDI-TOFMS. The MALDI-TOFMS spectra (peptide mass fingerprint) obtained from the protein in positive band no. 5, 55 kDa, are shown in Figure 2a. The peaks numbered in the figure are identified as the m/z of tryptic peptides with their high sequence coverage and score. As shown in Figure 2b, the MALDI/TOF-TOF MS of the peak, MH + =1642.38 in Figure 3, revealed a sequence of α-1 AT from 50 to 63 amino acid residues. The MS/MS spectra of the peak, MH+=2186.74 in Figure 2a, also coincided with the sequence of α-1 AT from 161 to 179 amino acid residues (data not shown).
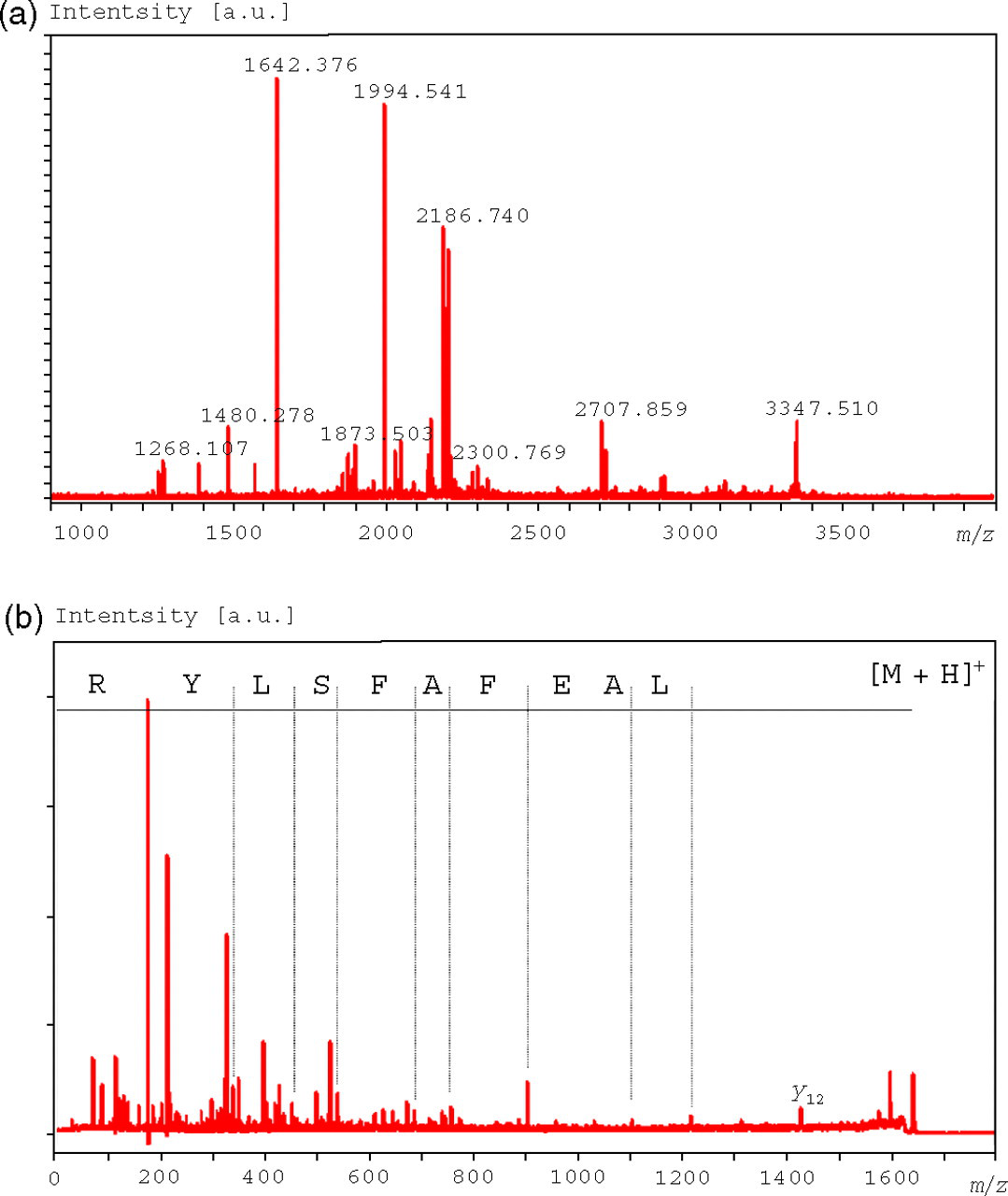
(a) Matrix-assisted laser desorption time-of-flight mass spectrometry (MALDI/TOFMS) mass spectrum (mass fingerprint) obtained from the proteins in band no. 5 treated with a Zip Tip C18 after tryptic digestion. Marked numbers indicate the m/z of tryptic digests of α-1 AT. (b) MALDI-TOF/TOF mass spectra obtained from a peak, MH+=1642.38, which coincided with the sequence of α-1 AT from 50 to 63 amino acid residues

Western blot analyses of proteins in the unbounded and desalting fractions of vitreous humours (VHs) derived from macular hole (MH) patients and a normal rabbit using an anti-PY-20 antibody. The arrows indicate specific bands present only in VHs of MH
Proteins identified from gel bands, tryptic peptides identified by matrix-assisted laser desorption time-of-flight mass spectrometry (MALDI-TOFMS) and time-of-flight mass spectrometry (TOF/TOFMS) and a database search showing the molecular mass
†[M+H]+, m/z used for TOF/TOFMS, numbers from N-terminal cited from database
Bold letters identified the sequences of tryptic peptides by TOF/TOFMS. Asterisks indicated oxidized methionine
Discussion
In this study, we used SDS–PAGE, WB and MS for the identification of five phosphotyrosyl proteins: α-1 AT, collagen α-2, catenin β-1, acyl CoA-binding domain containing protein IV and calcium-binding protein in VHs derived from patients with vitreoretinal diseases.
α-1 AT (55 kDa) is one of the acute-phase proteins in plasma as well as the other extracellular fluids, and it plays an important role in immunoresponse processes. Nakanishi et al. 1,2 previously reported that, by proteomics-based analysis, the expression of α-1 AT in VH of patients with PDR was more greatly increased than that of other vitreoretinal diseases. This suggested that the high concentrations of native α-1 AT in VHs were correlated with the state of inflammation or the severity of oxidative stress in the retina. So, we concluded that α-1 AT in VHs would be a candidate biomarker for the diagnosis of PDR in our previous report. On the other hand, there are few reports on modified α-1 AT, such as via phosphorylation, oxidation and β-elimination, in VHs. In this study, we first identified phosphotyrosylated α-1 AT in VHs of vitreoretinal diseases by MALDI-TOFMS and WB analysis. Yukitake et al. 13 reported that α-1 AT not only stimulated the phosphorylation activity of cyclic AMP-dependent protein kinase (PKA) in whole cultured testis cell extracts, but also α-1 AT itself was phosphorylated by PKA in vivo and in vitro. This suggested that the α-1 AT phosphorylated by PKA is related to spermatogenesis in human and animal testes. 13 Generally, phosphorylation of the protein plays an important role in a signal transmission cascade and also controls gene expression, cell proliferation, differentiation and various cell multifunctions such as apoptosis. When the activities of PKA in phosphorylation abnormally increase, it causes the failure of homeostasis, such as via excessive angiogenesis, mutagenesis or inflammation.
In general, the expressions of angiogenic factors and acute-phase reactant proteins were increased in the states of angiogenesis and/or inflammatory and then the rate of phosphorylation by ATP-dependent protein kinases was increased in endothelium. On the other hand, Lopes-Neblina et al. 14 reported that phosphorylated proteins decreased after ischaemia–reperfusion injury using a hepatic model of partial ischaemia and reperfusion. In his report, hypoxia initially activates the kinase system, which also initiates a cascade of phosphoregulatory events. One of these protein kinases, Akt (a 60 kDa protein kinase) phosphorylates and/or modulates various proteins and thereby plays a critical role in the activation of other modulators of the inflammatory state. They concluded that the phosphorylation of the Akt was decreased after ischaemia–reperfusion injury, but that phosphorylated Akt increased after treatment with an antiselectin blocker, TBC-1269. Our data in this study showed that the phosphorylated α-1 AT was only detected in VHs derived from MH and RD, although the conditions in the retina of PDR were not identical to those in hepatic cells under ischaemia or reperfusion. We speculated that the activities of the dephosphorylation reaction in the inflammatory state might be more increased than those of the non-inflammatory state, such as in MH and RD.
Collagen α-2 is a component of the extracellular matrix that is able to form structural links with cells. The concentrations of type IV collagen (IV-C) in vitreous fluids of PDR were higher than those in other vitreoretinal diseases in response to the progression of diabetic retinopathy. 15 Although the collagen was bound with discoidin domain receptor 1 and activated a tyrosine kinase-mediated signalling cascade, the collagen itself was not phosphorylated by protein kinases.
Catenin β-1 plays essential roles in both cell–cell adhesion and signal transductions, and catenins link some cytoplasmic domains of classic cadherins to the cytoskeleton. 16 One correlation of the cytoskeleton association is the phosphorylation of tyrosine residues of catenin β. The phosphorylation of catenin β is regulated by the combined activities of the tyrosine kinase Fer and the tyrosine phosphatase PTP1B. Under an elevated tyrosine kinase activity, catenin β is tyrosine phosphorylated, cadherin-mediated adhesions are compromised and the metastatic potential is increased. This condition might well be the case among many tumour cells. 17
Of course, it requires further research to identify more phosphotyrosyl proteins from WB-positive spots. We believe that the identification of phosphotyrosyl proteins in VHs may be an important initial step to investigate the pathological changes in vitreoretinal disease, and phosphotyrosyl protein will be an important diagnostic biomarker.
In conclusion, we identified α-1 AT in VHs derived from patients with MH and RD. Phosphotyrosyl α-1 AT was neither detected in VHs derived from patients with PDR, nor in the plasma. Phosphotyrosyl α-1 AT may be a new biomarker of MH and RD.
Footnotes
Acknowledgements
This work was supported by a 2005–2006 Grant-in-Aid for Scientific Research (17590501) from the Ministry of Education, Science and Culture of Japan.
