Abstract
Abstract
Background
Internal standards are essential in quantitative mass spectrometry (MS) assays to correct for variability in sample extraction and ionization at the source. In liquid chromatography MS assays, analogues of the analyte with several atoms replaced by their stable isotopes, e.g. 2H (D, deuterium) are often used as internal standards.
Methods
Possible interference by naturally occurring isotopes of an analyte in the internal standard channel in a liquid chromatography tandem MS assay was assessed using cortisol and its deuterated internal standard, D2-cortisol, as an example. Mass spectra were analysed and standard curves were prepared with varying concentrations of internal standard to determine the extent of any interference.
Results
The mass spectra showed that a naturally occurring isotope of cortisol at m/z 365 acts in the same way as D2-cortisol and fragments to give a daughter ion of the same m/z. Cortisol-365 can therefore falsely, but significantly, increase the amount of internal standard detected, and this will concomitantly decrease the relative response for cortisol. The standard curves with varying concentrations of internal standard showed that this phenomenon can affect the linearity of an assay.
Conclusions
Our results show that care is needed in assay development when doubly deuterated internal standards are used. Interference by naturally occurring isotopes of the analyte of interest in the internal standard transition is possible and it is important to ensure that an appropriate internal standard concentration is chosen that permits linearity of the assay over the required range.
Introduction
Internal standards (IS) are essential in quantitative mass spectrometry (MS) assays to correct for variability in sample extraction and ionization at the source. A fixed amount of IS is added to the sample early in the extraction procedure and is thus taken through the same steps of sample preparation as the analyte of interest. The analyser detects both the analyte and IS and the response is recorded as a ratio of the two, so that any variation in extraction or ionization between samples is compensated for. 1
It is important that the analyte and IS have sufficiently similar chemical and physical properties such that they behave in the same way and are subject to the same matrix effects. IS in quantitative liquid chromatography-MS assays tend to be either structural analogues or stable isotopically labelled (SIL) analogues of the analyte. SIL–IS are compounds in which several atoms in the analyte are replaced by their stable isotopes, e.g. 2 H (D, deuterium), 13C, 15N or 17O. SIL–IS are preferred for MS detection since they are chemically identical to the analyte and therefore chromatograph in a similar manner. 1
The aim of this project was to assess possible interference by naturally occurring isotopes of the analyte of interest in the IS transition.
Methods
The measurement of cortisol and its deuterated IS, D2-cortisol, by liquid chromatography tandem mass spectrometry (LC-MS/MS) was used as an example. A WatersTM Quattro Micro tandem mass spectrometer (Waters, Manchester, UK) was used in electrospray positive ionization mode. 2 To ascertain how isotopes of cortisol could interfere in D2-cortisol measurement, solutions of cortisol and D2-cortisol in methanol at a concentration of 1 mg/L were infused into the mass spectrometer and the mass spectra of both the parent and daughter ions were acquired.
The extent of any interference was determined by preparing four different standard curves over the cortisol concentration range 0–1500 nmol/L, each with a different concentration of D2-cortisol IS; 4, 10, 41 and 205 nmol/L. The standard curves were prepared by adding 25 μL D2-cortisol IS to 100 μL calibrator in a 96 deep well plate, followed by 100 μL trichloroacetic acid 5% (w/v). The plate was then thermosealed, vortex-mixed and centrifuged at 8000
Results
The parent ion scans show that the mass to charge ratio (m/z) of the most abundant parent ion of cortisol is 363. Since nominal mass data were used, isotopes of cortisol are also seen with decreasing abundance in the spectrum at m/z 364 and m/z 365 due to the naturally occurring 13C content (Figure 1a). The isotope of cortisol at m/z 365 has the same m/z as the most abundant parent ion of the IS D2-cortisol (Figure 1b).
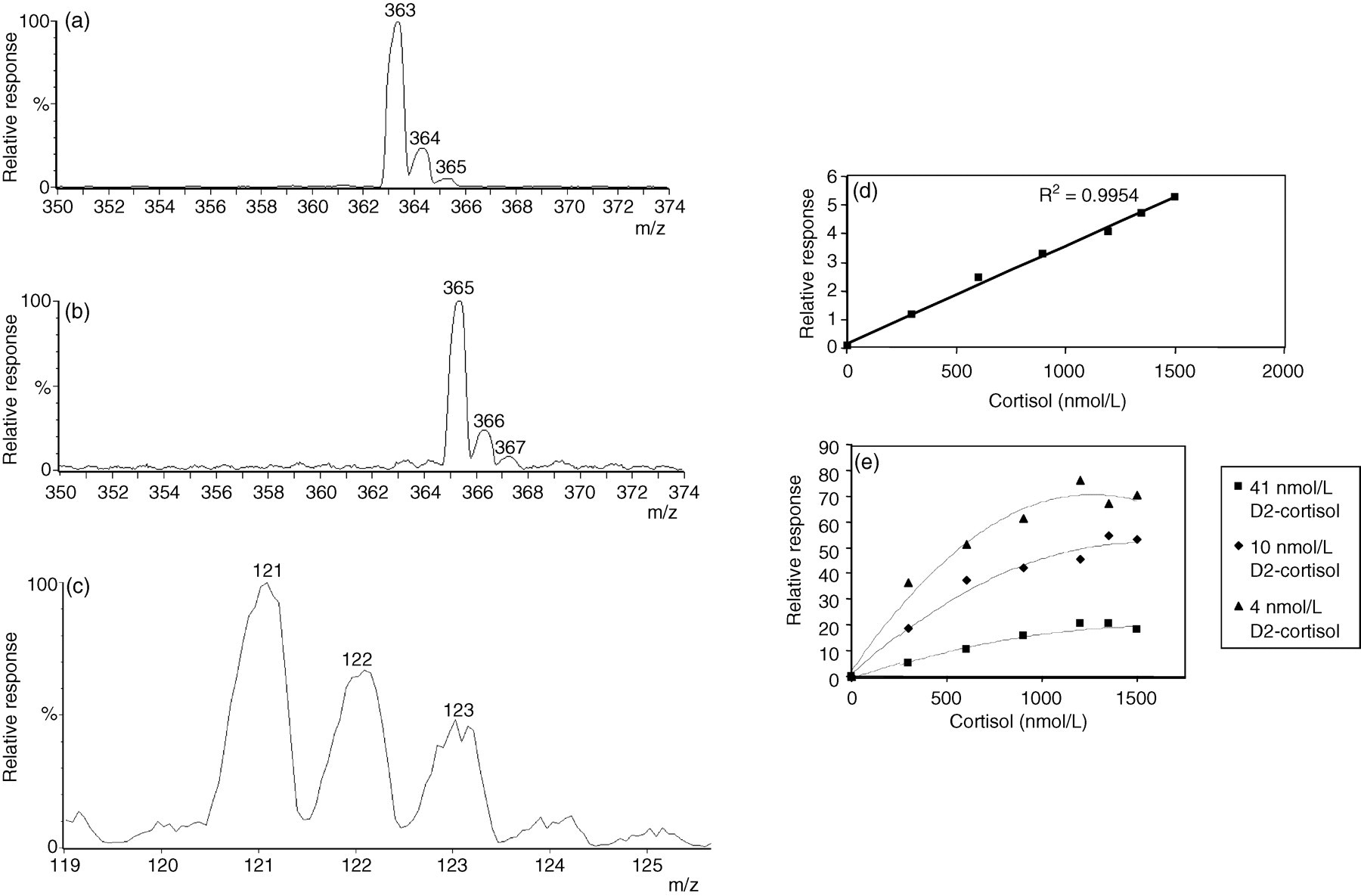
(a) Mass spectrum of the parent ions of cortisol; (b) Mass spectrum of the parent ions of D2-cortisol; (c) The daughter ions produced when cortisol-365 fragments; (d) Cortisol standard curve with internal standard (IS) concentration 205 nmol/L; (e) Cortisol standard curve with IS concentrations 41, 10 and 4 nmol/L
The most abundant daughter ion of cortisol has m/z 121 and that of D2-cortisol has m/z 122. Thus the transitions monitored are 363 > 121 for cortisol and 365 > 122 for D2-cortisol. The isotope cortisol-365 will fragment to give daughter ions of m/z 121, 122 and 123 (Figure 1c). Thus cortisol-365 may be measured by the IS transition and falsely increase the ion count of this transition.
Standard curves for the cortisol assay were repeatedly prepared with varying IS concentration. When the highest concentration of IS was used a linear standard curve was produced using all of the standards. However, as the IS concentration was decreased, although the standard curves remain linear at the low end, they become increasingly less linear when the high standards are included (Figures 1d and e).
Discussion
The mass spectra of the parent and daughter ions of cortisol and D2-cortisol show how the naturally occurring isotope cortisol-365 can falsely increase the amount of IS detected, and concomitantly decrease the relative response for cortisol. The standard curves prepared with varying concentrations of IS show that this effect is most pronounced when high concentrations of cortisol are present with low concentrations of D2-cortisol that are not sufficient to mask the contribution from cortisol-365.
We have seen this same effect in other cases where a D2 deuterated IS is used, for example, testosterone and D2-testosterone. However, as the effect is most pronounced when high concentrations of the analyte are measured, whether or not non-linearity of the standard curve occurs depends on the analytical range of the assay.
Use of an IS with greater than 2 deuteriums can overcome this phenomenon since interference in the measurement of an IS of this nature could only occur if a natural isotope of the analyte >2 mass units heavier than the most abundant parent ion was found. Methods have been published which use D3-cortisol as an IS, suggesting awareness of this problem. 3
This work does not detract from the fact that deuterated IS are highly appropriate for use in LC-MS/MS assays. However, caution must be used in assay development to ensure that an appropriate IS concentration is chosen that permits linearity of the assay over the required range.
