Abstract
Abstract
In Western countries, thyrotoxic periodic paralysis (TPP) is a rare condition with only sporadic cases described so far. Here, we describe a 29-year-old Italian man who presented with leg weakness and hypokalaemia. Treatment with intravenous potassium resulted in a rapid resolution of symptoms. TPP as the underlying cause was suggested by suppressed thyroid-stimulating hormone (TSH), elevated free T3 and free T4, and the presence of TSH-receptor antibodies (TRAB). Genetic analysis showed no mutations in the candidate exons of calcium (CACN1AS), potassium (KCNE3) and sodium (SCN4A) channel genes. However, we identified the presence of two single nucleotide polymorphisms (SNPs), 1491C
Introduction
Thyrotoxic periodic paralysis (TPP) is a neuromuscular disorder characterized by episodic attacks of severe muscle weakness, low serum potassium and thyrotoxicosis. 1,2 Periodic paralysis is a complication of thyrotoxicosis occurring for the most part in Asian populations 3,4 with only sporadic cases described in Western countries. 5–7 Paralytic attacks may range from mild weakness of a particular group of muscles to a generalized weakness. 8 Usually, proximal muscles are affected more severely than the distal ones and meals rich in carbohydrate may precipitate these episodes. 4 Hypokalaemia is a common finding during the attacks, resulting from the shift of serum potassium into the intracellular space which is exacerbated by the hyperthyroidism. 9 The clinical features of TPP are similar to those occurring in hypokalaemic periodic paralysis (HPP), an autosomal dominant hereditary disorder. 10 In HPP, mutations have been found in the genes encoding for the dihydropyridine receptor calcium channel (CACN1AS), the voltage-gated potassium channel (KCNE3) and the voltage-gated sodium channel (SCN4A), which influence the functions of skeletal muscle. 11–13 These mutations have not been found in Asian patients with TPP. There are three reported cases in the Italian population which were not tested for mutations. 14–16 Here, we describe an Italian man affected by TPP who underwent genetic analysis.
Case report
The patient was a 29-year-old Italian man who was taken to the emergency room because of lack of strength in his legs and moderate weakness of the upper limbs. At the admission, the patient was unable to stand up. Muscle weakness had begun the night before. Initially moderate, weakness became progressively worse. Two other episodes occurred within the same month, both characterized by discomfort in the legs before the development of severe weakness that resolved spontaneously after 5 and 10 h, respectively. The patient reported a weight loss of 10 kg in the previous nine months. He had been taking sport supplement bags containing potassium and magnesium since he experienced leg weakness. All episodes occurred in the evening after meals rich in carbohydrates. Family history was positive for thyroid diseases with one sister affected by Graves' disease.
Physical examination revealed a body temperature of 37.8°C, a blood pressure of 180/90 mmHg and a heart rate of 115 beats/min. On neurological examination, the patient was alert and oriented. Proximal weakness was present in all extremities. Deep tendon reflexes were hypoactive. Neither rigour nucalis, nor sensory or cranial nerves deficit were observed. A magnetic resonance imaging of the spinal cord revealed no significant findings. Initial laboratory tests reported serum potassium concentrations of 2.6 mmol/L; other electrolytes were normal. The patient was treated with an infusion of 40 mmoL of potassium chloride in 500 mL of normal saline and sent to the Neurology Department 6 h later. Here, the neurological examination disclosed normal sensorium, nonetheless attention was drawn to the presence of tachycardia, tremor and recent weight loss. Further laboratory tests revealed the following values: free T3 20.0 pmol/L (3.4–7.8), free T4 73.3 pmol/l (9.0–21.9), thyroid-stimulating hormone <0.01 mU/L (0.4–4), thyroid receptors antibodies 17.60 UI/L (<10). Overall, such findings supported the diagnosis of TPP and Graves' disease as the underlying cause. Treatment with methimazole 30 mg/day and propranalol 40 mg/day was started.
Methods
Genomic DNA was extracted from peripheral blood lymphocytes using the QIAamp NA blood Mini Kit (Qiagen, Valencia, CA, USA). Primers sequences used to detect the most common HPP mutations are reported in Table 1. Polymerase chain reaction (PCR) reactions were performed employing 100 ng of genomic DNA in a 50μL volume containing 0.2 mmol/L of each deoxy-NTP (Invitrogen, Paisley, UK), 10 pmol of each specific primer (Invitrogen), 5μL of PCR Gold Buffer 10× (Applied Biosystems, Foster City, CA, USA), 1.5 mmol/L of MgCl2 (Applied Biosystems) and 1.5 U of Amply Taq Gold (Applied Biosystems). Cycling conditions included an initial denaturation phase of 10 min at 94°C, followed by 40 cycles of 1 min of denaturation at 95°C, 1 min at annealing temperature, 1 min of extension at 72°C and a final extension of 10 min at 72°C. PCR products were resolved in a 1.5% agarose gel stained with ethidium bromide and purified using Montage PCR columns (Millipore, Billerica, MA, USA). Purified PCR products (3 μL) were sequenced using the ABI PRISM Big Dye terminator Cycle Sequencing Reaction Kit and the ABI PRISM 3100 Genetic Analyzer after purification with Centrisep columns (Applied Biosystems).
Primer sequences and polymerase chain reaction conditions used for DNA analysis
Results
Genetic analysis
DNA analysis revealed that no mutations were present in exons 11 and 30 of the CACN1AS gene, in exon 2 of the KCNE3 gene and in exon 12 of the SCN4A gene. However, we detected two different single nucleotide polymorphisms (SNPs) in exon 11 of the CACN1AS gene. The first one was a substitution 1491 C > T in the nucleotides encoding for asparagine 497 (Asn 497 Asn) (Figure 1a); the second one was a substitution 1551 T > C in the nucleotides for glycine 517 (Gly 517 Gly) (Figure 1b).
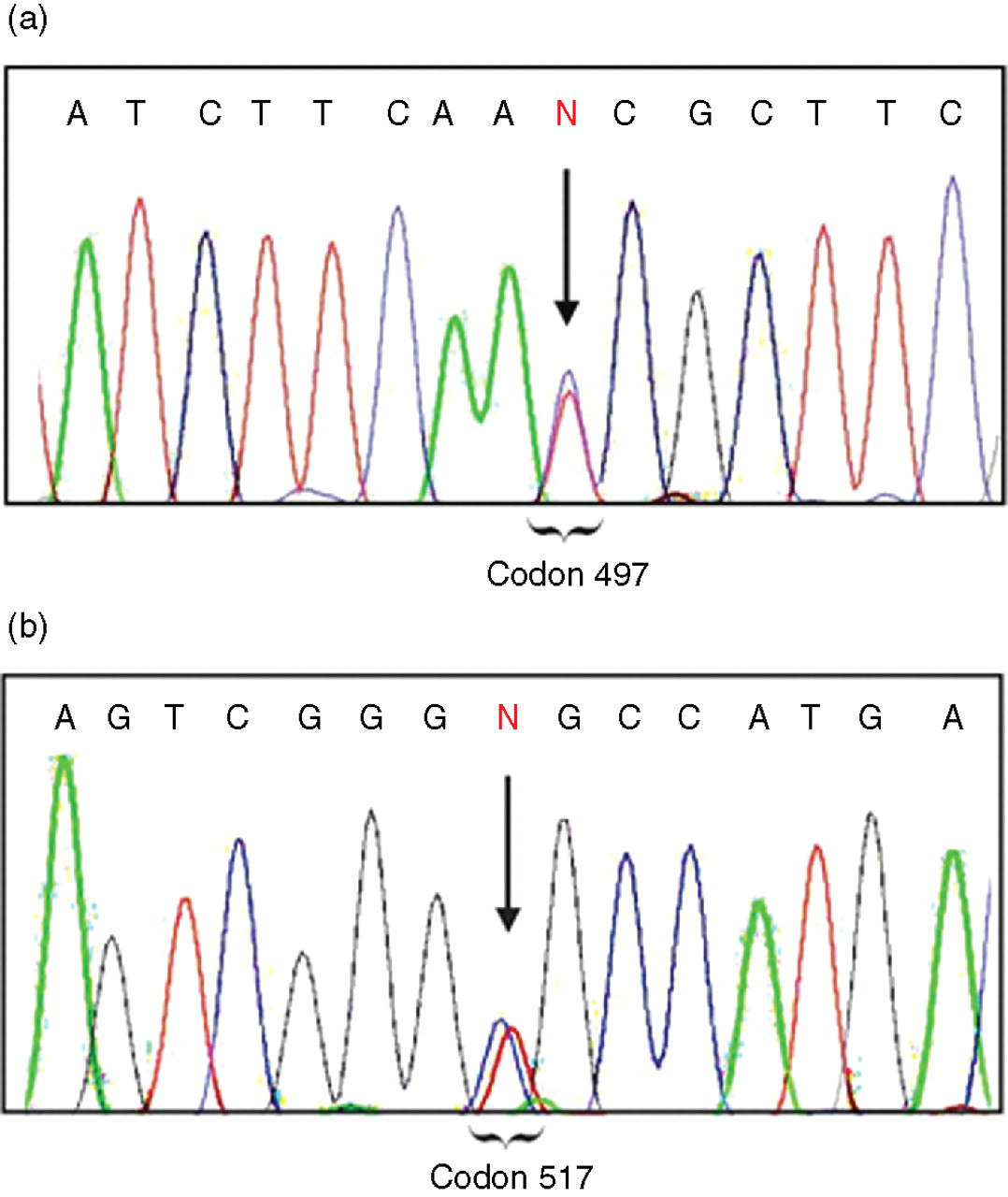
DNA sequence analysis of exon 11 CACN1AS gene. The arrows point at two heterozygous single nucleotide polymorphisms. (a) 1491C > T codon 497 (Asn 497 Asn) (b) 1551T > C codon 517 (Gly 517 Gly)
Discussion
TPP represents a life-threatening condition that in Asian countries affect up to 2% of individuals with hyperthyroidism. 1 The observation that hypokalaemic paralysis may occur as a manifestation of both HPP and TPP has led several groups to investigate whether the previously described HPP-associated mutations may be involved also in the development of TPP. 17–19 Kung et al. 17 examined 97 Chinese male subjects affected by TPP and found no association with mutations in the CACN1AS, KCNE3 and SCN4A genes associated with HPP. Ng et al. 18 analysed an additional 20 Chinese TPP patients and excluded any association with the CACN1AS and SCN4A genes. 18 Finally, 11 Thai TPP patients, analysed for the presence of channel mutations, confirmed the results of the above studies. 19 In Caucasians, a R83H mutation in the KCNE3 gene has been reported in a Brazilian man with TPP, 20 but others have not confirmed such findings. 21 In our case, genetic analysis revealed no mutations in the exons of CACN1AS, KCNE3 and SCN4A genes analysed, suggesting that, as with Asians, the mechanisms underlying the pathogenesis of HPP and TPP are different. It should be noted, however, that we observed two silent SNPs in exon 11 of the CACNA1S gene including SNPs 1491 C > T and 1551 T > C. In the Thai population, a higher but not statistically significant frequency of the 1491 C > T SNP has been reported in TPP individuals compared with the normal subjects. 19 As for the SNP 1551 T > C, a study on Brazilian subjects affected by TPP reported a higher frequency of such SNP in TPP individuals compared with controls, 22 and another study in Thai TPP subjects confirmed such data. 19 In Western populations over the last years, an increasing number of TPP case reports have been published, but a genetic analysis has been rarely performed. In Europeans, the SNP 1551 T > C has been reported only in one patient, 23 with our patient being the second one. The exact role of these SNPs in the pathogenesis of TPP is unclear. Kung et al. 17 reported an association of some SNPs in the CACNA1S gene with TPP in Chinese patients. The authors suggested that since these SNPs are proximally located to the thyroid responsive element (TRE), they may affect the binding affinity of TRE and then modulate the stimulation of thyroid hormone on the CACNA1S gene. Although the extreme rarity of this disease among Europeans makes it difficult to conduct proper association studies, our case report shed more light on the genetic features of TPP in Caucasians. The observation that few data are available on the genetic characteristics in such patients suggests that more efforts are necessary to evaluate the role of genetics in the pathogenesis of TPP in Western countries.
Footnotes
ACKNOWLEDGEMENTS
This study was supported by grants from the Italian Ministry of Health, the Italian Ministry of Research and from the Fondazione Umberto Di Mario ONLUS.
