Abstract
Abstract
Background
We have examined the effect of delayed sample separation on serum creatinine measurement using the kinetic Jaffe method and estimated glomerular filtration rate (eGFR), and the impact this can have on chronic kidney disease (CKD) staging.
Methods
In a preliminary study clotted samples were left at room temperature for up to 48 h prior to centrifugation and serum creatinine measurement on a Roche modular analyser. A larger study then used paired clotted samples separated at baseline (30 min) and after 24 h at room temperature. To determine the effect on CKD staging, eGFR was calculated using the simplified [4-v] Modification of Diet in Renal Disease formula.
Results
Preliminary study. A significant increase in creatinine concentration (P < 0.001) occurred after a 16 h delay in sample separation. By 48 h the mean increase in creatinine was 29% (range 21–63%).
Twenty-four hour delay study. The mean increase in creatinine for 113 outpatient samples after delaying sample separation by 24 h was 11%, and exhibited considerable individual variation (range 2–87%). CKD staging for 32% of the group changed, 26% from stage 1 to 2 and 6% from stage 2 to 3.
Conclusions
Delay in sample separation affects the creatinine measurement using the Roche kinetic Jaffe reaction, and can result in the misclassification of CKD staging. Laboratories using the kinetic Jaffe method should not report creatinine results where there has been a delay in separation. This advice should also be reflected in practice guidelines.
Introduction
In April 2006 in-line with the Department of Health recommendations for implementing the National Service Framework for renal services, 1 National Health Service (NHS) clinical biochemistry laboratories in England began routinely reporting estimated glomerular filtration rate (eGFR). This aimed to simplify the management of chronic kidney disease (CKD) by enabling individuals to be categorized according to their level of kidney function into five stages, stage 1, >90, stage 2, 89 to 60, stage 3, 59 to 30, stage 4, 29 to 15 and stage 5, <15 (units in mL/min/1.73 m 2 ). 1,2 Estimated GFR is calculated using an equation derived from the Modification of Diet in Renal Disease (MDRD) Study, for which serum creatinine concentration is a critical variable. 3–5
The most common methods in routine use for serum creatinine determination in the UK are based on the Jaffe reaction. 6 This is despite continued criticism of the Jaffe method, which is subject to analytical interferences from substances such as bilirubin, ascorbic acid, glucose, pyruvate, acetoacetate, certain antibiotics and proteins. 7–10 These interferences are well known and assay modifications have been incorporated to minimize them. 9,11,12 What is less well known is that creatinine measurement using Jaffe methods may also be affected by sample age, and more specifically the time elapsed before sample separation.
In 1992 a study was published showing that after 48 h creatinine concentration increased by nearly 40% in lithium heparin samples left un-separated at room temperature. 13 This increase was observed using a kinetic Jaffe method (Dimension, DU Pont Wilmington, DE, USA), but not when creatinine was measured enzymatically. It was therefore suggested that this increase was not as a result of a change in creatinine itself, but was due to a non-specific chromogen that increases in concentration over time and cross-reacts with the Jaffe assay. The majority of requests for eGFR originate from primary care, so the likelihood that there will be a significant delay between sample collection, separation and analysis is increased. Consequently, if sample age does have an effect on serum creatinine measurement using Jaffe methods, this could result in misclassification of the CKD stage.
In this study, we have re-investigated the effect delay in sample separation and analysis has on the measurement of serum creatinine using a widely used Jaffe method (Roche compensated kinetic Jaffe), and examined the impact this has on both the eGFR and CKD staging.
Patients' samples and methods
Preliminary studies
Un-separated blood
Seven clotted blood samples (Vacuette serum sep. clot activator tubes, 4 mL, Greiner Bio-one GmbH, Kremsmünster, Austria) were collected from 10 laboratory volunteers, nine Caucasian, one Asian, five male, five female, age range 27–55 years. The un-separated clots were left exposed to light at room temperature (22±2°C) and the serum separated by centrifugation at 4600 rpm for 10 min after 30 min (baseline), 4, 8, 16, 24, 36 and 48 h. The serum was transferred to 5 mL plastic tubes (Starstedt), capped and frozen at −20°C.
Separated serum
Ten clotted blood samples (Vacuette serum sep. clot activator tubes, 5 mL) received from outpatients were selected at random (all Caucasian; 7 males, 3 females, aged 27–89 years). After 30 min the serum was separated by centrifugation, and a 200 μL aliquot of serum (baseline) transferred to a 5 mL tube, capped and frozen at −20°C. After all requests were complete the serum was left exposed to light at room temperature, and further 200 μL serum aliquots taken at 4, 8, 16, 24, 36 and 48 h and stored at −20°C.
Twenty-four hour delay study
Clotted blood samples were collected in duplicate from 113 individuals attending outpatient clinics and the sex, age and ethnicity were recorded (59 Caucasian, 44 Asian and 9 Afro-Caribbean; 50 male, 63 female, aged 18–88 years). After 30 min the serum was separated by centrifugation, and a 200 μL aliquot of serum (baseline) transferred to a 5 mL tube, capped and frozen at −20°C. The second clotted blood sample was left un-separated for 24 h at room temperature, then centrifuged and the serum frozen at −20°C.
At the end of the experiment all the serum samples were analysed at the same time to minimize the effect of method inter-batch variation. Samples were studied on the basis of assay validation for which local ethics committee approval was not required. The analysis of patients' samples was not delayed as a result of this study.
Creatinine kinetic Jaffe method
Creatinine measurement was performed using a Roche modular vertical P analyser with Roche creatinine Jaffe reagents (Roche, Diagnostics Limited, East Sussex, UK). This method is standardized against isotope dilution mass spectrometry. In accordance with the manufacturer's instructions to compensate for proteins that react non-specifically in the Jaffe method, all results were corrected down by 26 μmol/L. In our laboratory the between-batch coefficient of variation for creatinine concentrations of 63 and 175 μmol/L is 4.34 and 2.62%, respectively, (n = 176), and within-batch 2.20 and 1.22%, respectively (n = 10).
Creatinine enzymatic method
From the 24 h study the 10 duplicate paired samples with the largest difference between creatinine concentrations for samples separated after 30 min (baseline) and a delay of 24 h creatinine were selected. These were then analysed using a VITROS 5, 1 FS Chemistry System with VITROS enzymatic creatinine slides (Ortho-Clinical Diagnostics, Johnson-Johnson Company, High Wycombe, Bucks, UK).
Statistics and calculations
Estimated GFR was calculated using the simplified [4-v] MDRD formula, multiplied by a correction factor of 1.212 for Afro-Caribbeans and 0.742 for women. As for routine clinical reports in our laboratory, Asians were treated as Caucasians with respect to GFR estimation.
Results
Preliminary study
The effect of leaving clotted blood un-separated on creatinine measurement and eGFR for the 10 volunteers is shown in Figure 1. Analysis of the results for each volunteer using repeated measures ANOVA with Tukey's multiple comparisons showed that the increase in creatinine concentration was significant after 16 h (P < 0.001). By 48 h creatinine concentrations had increased above the baseline measurement (samples separated after 30 min) on average by 29% (range 21–63%). The mean coefficient of variation for the seven creatinine measurements performed for each volunteer was 11.3% (range 8.1–16.2%) and was over five times greater than that found for the within-batch variation of the assay.
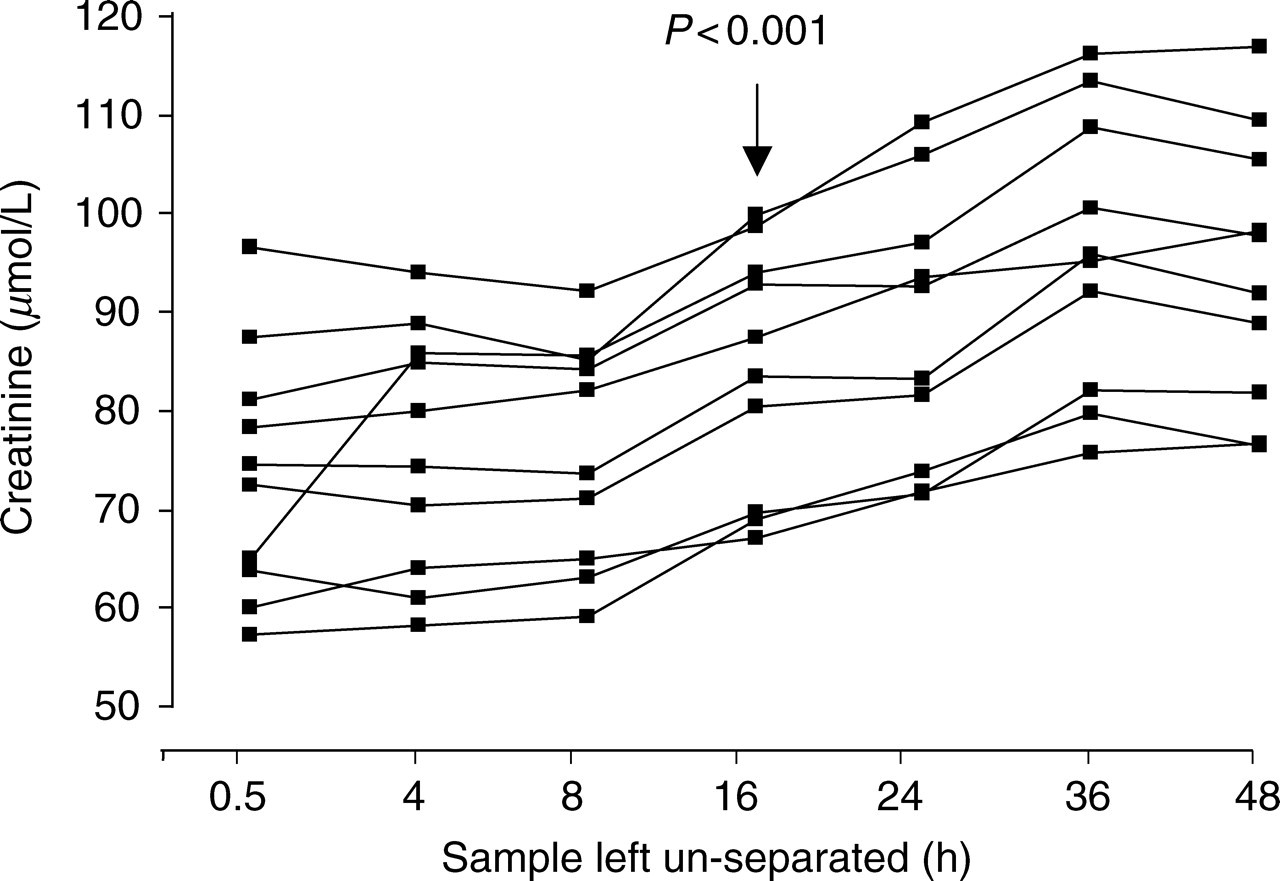
Effect of delaying clotted sample separation for up to 48 h on creatinine concentration determined using the Roche kinetic Jaffe method for 10 volunteers. The time at which the increase in creatinine concentration is significant compared with baseline is indicated on the figure and was determined using a repeated measures ANOVA with Tukey's multiple comparisons test
Figure 2 shows the effect this increase in creatinine has on the CKD staging for the 10 volunteers. Baseline creatinine measurement revealed that three of the volunteers had an eGFR in the CKD stage 2 category and the remaining seven, CKD stage 1. Based on the changes in eGFR alone, by 16 h the staging of four of the seven volunteers with a baseline CKD stage 1 had changed to stage 2, and this was seen in all seven by 36 h. Despite changes in creatinine and eGFR the staging of the three individuals with a baseline CKD stage 2 did not alter during the course of the study.
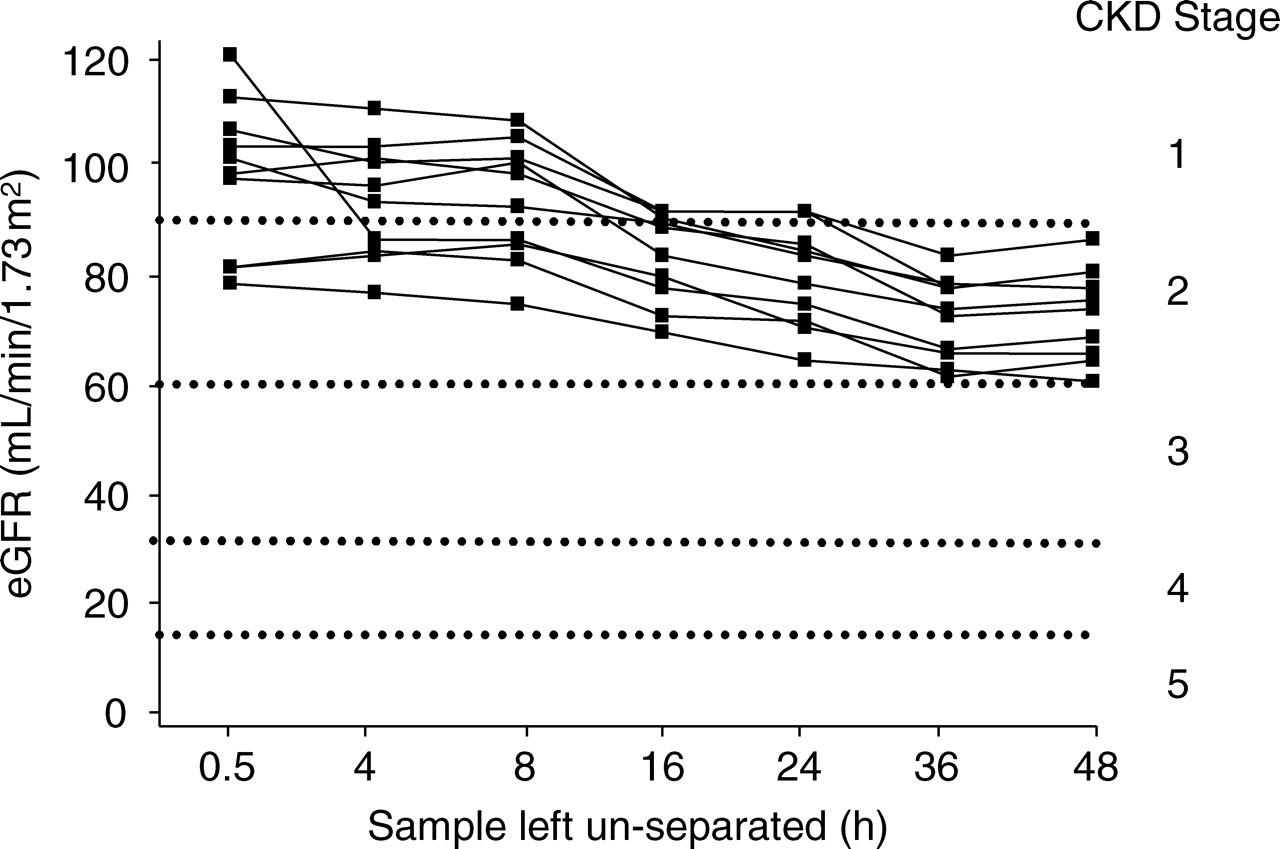
Effect of delaying clotted sample separation for up to 48 h on chronic kidney disease (CKD) staging for 10 volunteers.
When the experiment was repeated using 10 randomly selected separated serum samples sent to the laboratory from outpatient departments, no increase in measured creatinine concentration over time was observed. In fact, after 48 h a slight drop in creatinine concentration was found. The mean coefficient of variation for the seven creatinine measurements performed for each serum sample was 4.87% (range 2.38–7.81%) and is closer to that found for the within-batch variation of the assay.
Twenty-four hour delay study
Table 1 shows the effect leaving clotted blood samples un-separated for 24 h at room temperature has on creatinine measurement and eGFR for 113 patients attending outpatient clinics. Using the Wilcoxon matched pairs signed ranks test, the difference in mean creatinine between the baseline and 24 h samples was found to be highly significant (P < 0.0004), as was the difference in eGFR (P < 0.0001). As would be expected, the mean baseline creatinine concentration was higher for men compared with women, although the mean eGFR was similar. There were slight differences in the mean baseline creatinine and eGFR between the different ethnic groups, but the number of subjects studied was not high enough for the results to be significant. Neither sex nor ethnicity changed the effect of the 24 h delay in sample separation on creatinine measurement, with increases of approximately 11% seen in the male, female and the three different ethnic groups.
Effect of delaying clotted sample separation for 24 h on creatinine concentration using Roche kinetic Jaffe reaction and the estimated glomerular filtration rate (eGFR) for 113 randomly selected individuals attending outpatient clinics
P values were calculated using the Wilcoxon matched pairs signed ranks test
Figure 3 shows the baseline CKD staging for the 113 study population and the change as a result of delaying sample separation for 24 h. The staging of 32% of the study population changed after the 24 h delay, 26% went from CKD stage 1 to stage 2, and 6% from stage 2 to stage 3.
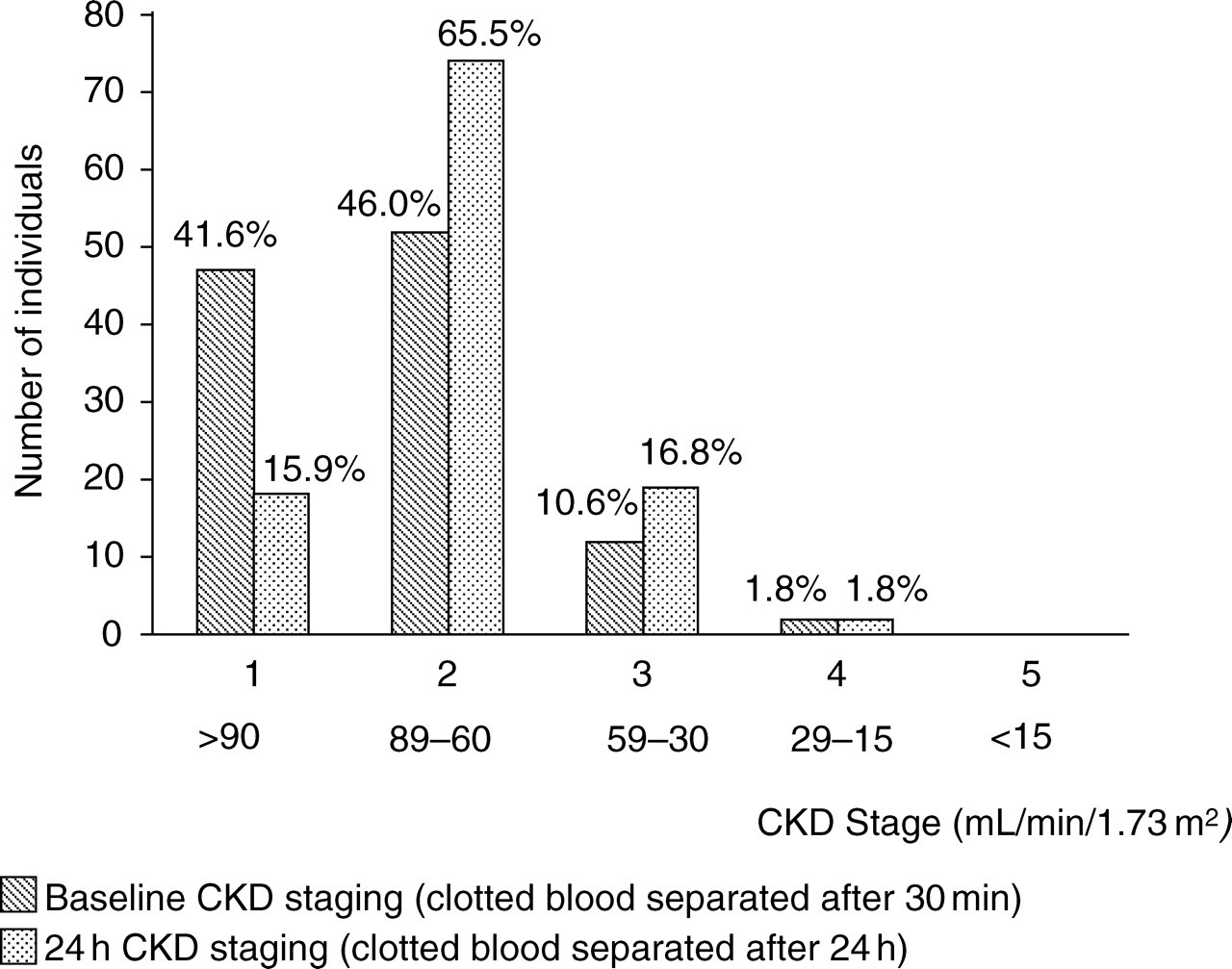
Effect of 24 h delay in separation of clotted samples on chronic kidney disease staging for 113 randomly selected individuals attending outpatient clinics
Ten duplicate samples with the greatest difference in creatinine concentrations between the baseline measurement and after a delay in separation of 24 h were selected (mean % increase in creatinine concentration 29.4, range 19.7–86.6%), and analysed using an enzymatic method on the VITROS 5. The results are compared in Table 2. In contrast to the kinetic Jaffe method, little change in creatinine was observed using this enzymatic method, with a mean decrease in creatinine concentration of 2.7% (range –13.8 to +8.6%) between the baseline and samples separated after a 24 h delay.
Comparison of creatinine concentrations measured using Roche kinetic Jaffe reaction and Ortho enzymatic reaction for clotted samples separated after 30 min (baseline) and a delay of 24 h
Discussion
Our preliminary study of 10 laboratory volunteers showed that a delay in the separation of clotted blood samples does affect the creatinine measurement using the kinetic Jaffe assay, thus confirming the findings of previous studies. 13,14 We also found that the increase in creatinine is significant after only a 16 h delay in sample separation, and exhibits wide variation, for example, in one case the creatinine value went from 64 to 109 μmol/L after delaying sample separation by 36 h, an increase of 68%. CKD staging was affected as a result, with 50% of the individuals studied changing from stage 1 to stage 2 after 24 h and 70% by 36 h. No significant change in creatinine was observed using separated serum samples or when the analysis was performed using an enzymatic method.
We extended the study to look at the impact delay in sample separation and creatinine analysis would have on outpatient samples. Although in the preliminary study the greatest effect on creatinine measurement was observed after 36 h, it is more realistic that the delivery of samples to the laboratory from general practitioners (GPs) may be delayed by up to 24 h. Therefore, for 113 random individuals attending outpatient clinics a comparison of creatinine results was made between samples separated immediately and a second sample left un-separated at room temperature for 24 h. On average, creatinine results increased by 11% after delaying sample separation by 24 h. Again this increase was not consistent, varying from 2 to 87%. For one individual, a 70-year-old man, the creatinine changed from a baseline concentration of 55 to 103 μmol/L. We could identify no obvious reason for this intra-individual variability in the effect delay in sample separation has on creatinine concentration. No correlation was found with gender, ethnicity or the serum concentrations of albumin, total protein or bilirubin, and further investigations are required to establish the cause.
The CKD staging for 32% of the study group changed, and in particular 6% went from stage 2 to stage 3 (<60 mL/min/1.73 m 2 ). Just how such results would impact on patient care is a matter for debate. The guidelines for CKD clearly state that an eGFR between 60 and 90 mL/min/1.73 m 2 does not, on its own, indicate CKD, and other evidence of kidney disease (e.g. proteinuria) or structural abnormality is required. 1,2 However, it is our experience that many clinicians in primary care do put considerable emphasis on the eGFR result alone. At the very least, if such results are reported this could result in unnecessary repeat phlebotomy and anxiety for those individuals affected.
With aspects of GFR estimation using the MDRD equation coming under scrutiny, including the measurement of creatinine by the Jaffe reaction, it is of concern that such a considerable pre-analytical contribution to variation has been overlooked. Neither recent national and international guidelines on CKD nor extensive reviews on the subject mention the effect of sample age on creatinine measurement. 1,4,5,9–11 Even the Roche kit insert for creatinine fails to mention the effect of un-separated clotted samples. 15 In the UK requests from GPs make up 40–70% of an NHS laboratory's workload and since all requests for creatinine automatically generate an eGFR result, misclassification of CKD staging due to delayed sample separation has the potential to affect many results. An eGFR <60 mL/min/1.73 m 2 , is associated with adverse outcomes such as cardiovascular disease, and intense management of these patients is recommended. 2 Consequently, the misidentification of stage 3 CKD due to sample age in particular could result in unnecessary drug treatment, and referral to specialist renal clinics.
This study has shown that delayed sample separation results in a significant and unpredictable increase in creatinine concentration determined using the kinetic Jaffe reaction after 16 h, and this can lead to changes in eGFR and CKD staging. Although many laboratories may already have rejection criteria for old un-separated samples in place, it is important that all laboratories introduce this. Unfortunately, it is not always easy to ascertain the age of samples taken in primary care and sent to a central laboratory for analysis. Hopefully, the introduction of more advanced information technology systems into primary care phlebotomy will mean that timing of samples is much more clearly defined. Future CKD guidelines should therefore include information warning of the possible impact of delaying separation on creatinine measurement using kinetic Jaffe methods, eGFR and CKD staging.
Footnotes
Acknowledgements
Thanks to City Hospital laboratory staff and phlebotomists who helped with this study. We are grateful to Samantha Clay who performed the creatinine analysis. We thank Dr David Vallance of the Department of Clinical Chemistry, Russell's Hall Hospital, Dudley, West Midlands DY1 2HQ for arranging creatinine analysis on the VITROS analyser.
